Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Gla domain AI simulator
(@Gla domain_simulator)
Hub AI
Gla domain AI simulator
(@Gla domain_simulator)
Gla domain
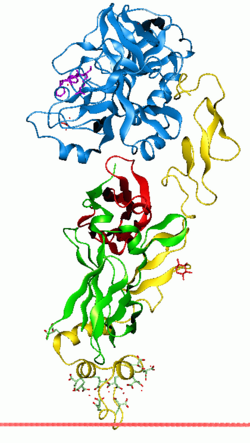
Vitamin K-dependent carboxylation/gamma-carboxyglutamic (GLA) domain is a protein domain that contains post-translational modifications of many glutamate residues by vitamin K-dependent carboxylation to form γ-carboxyglutamate (Gla). Proteins with this domain are known informally as Gla proteins. The Gla residues are responsible for the high-affinity binding of calcium ions.
The GLA domain binds calcium ions by chelating them between two carboxylic acid residues. These residues are part of a region that starts at the N-terminal extremity of the mature form of Gla proteins, and that ends with a conserved aromatic residue. This results in a conserved Gla-x(3)-Gla-x-Cys motif that is found in the middle of the domain, and which seems to be important for substrate recognition by the carboxylase.
The 3D structures of several Gla domains have been solved. Calcium ions induce conformational changes in the Gla domain and are necessary for the Gla domain to fold properly. A common structural feature of functional Gla domains is the clustering of N-terminal hydrophobic residues into a hydrophobic patch that mediates interaction with the cell surface membrane.
At present, the following human Gla-containing proteins (Gla proteins) have been characterized to the level of primary structure:
In all cases in which their function was known, the presence of the Gla residues in these proteins turned out to be essential for functional activity.
Gla domains are found in vertebrate coagulation proteins. There seems to be a single origin of these Gla domains.
The Gla domain causes a binding affinity to phosphatidylserine, a membrane phospholipid, in most Gla coagulation proteins. The exceptions are FVII and protein C, which instead bind phosphatidic acid. In any case, in vertebrate coagulation proteins, the Gla domain anchor the proteins to a membrane, allowing the coagulation complexes to form.
The bone Gla protein (osteocalcin) and the matrix Gla protein have diverged in a jawed vertebrate ancestor. In humans, the bone Gla protein mainly helps bone mineralization by collecting calcium ions and the matrix Gla protein prevents soft tissue calcification by binding away calcium ions.
Gla domain
Vitamin K-dependent carboxylation/gamma-carboxyglutamic (GLA) domain is a protein domain that contains post-translational modifications of many glutamate residues by vitamin K-dependent carboxylation to form γ-carboxyglutamate (Gla). Proteins with this domain are known informally as Gla proteins. The Gla residues are responsible for the high-affinity binding of calcium ions.
The GLA domain binds calcium ions by chelating them between two carboxylic acid residues. These residues are part of a region that starts at the N-terminal extremity of the mature form of Gla proteins, and that ends with a conserved aromatic residue. This results in a conserved Gla-x(3)-Gla-x-Cys motif that is found in the middle of the domain, and which seems to be important for substrate recognition by the carboxylase.
The 3D structures of several Gla domains have been solved. Calcium ions induce conformational changes in the Gla domain and are necessary for the Gla domain to fold properly. A common structural feature of functional Gla domains is the clustering of N-terminal hydrophobic residues into a hydrophobic patch that mediates interaction with the cell surface membrane.
At present, the following human Gla-containing proteins (Gla proteins) have been characterized to the level of primary structure:
In all cases in which their function was known, the presence of the Gla residues in these proteins turned out to be essential for functional activity.
Gla domains are found in vertebrate coagulation proteins. There seems to be a single origin of these Gla domains.
The Gla domain causes a binding affinity to phosphatidylserine, a membrane phospholipid, in most Gla coagulation proteins. The exceptions are FVII and protein C, which instead bind phosphatidic acid. In any case, in vertebrate coagulation proteins, the Gla domain anchor the proteins to a membrane, allowing the coagulation complexes to form.
The bone Gla protein (osteocalcin) and the matrix Gla protein have diverged in a jawed vertebrate ancestor. In humans, the bone Gla protein mainly helps bone mineralization by collecting calcium ions and the matrix Gla protein prevents soft tissue calcification by binding away calcium ions.