Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Radioactive decay AI simulator
(@Radioactive decay_simulator)
Hub AI
Radioactive decay AI simulator
(@Radioactive decay_simulator)
Radioactive decay
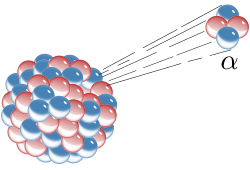
Radioactive decay (also known as nuclear decay, radioactivity, radioactive disintegration, or nuclear disintegration) is the process by which an unstable atomic nucleus loses energy by radiation. A material containing unstable nuclei is considered radioactive. Three of the most common types of decay are alpha, beta, and gamma decay. The weak force is the mechanism that is responsible for beta decay, while the other two are governed by the electromagnetic and nuclear forces.
Radioactive decay is a random process at the level of single atoms. According to quantum theory, it is impossible to predict when a particular atom will decay, regardless of how long the atom has existed. However, for a significant number of identical atoms, the overall decay rate can be expressed as a decay constant or as a half-life. The half-lives of radioactive atoms have a huge range: from nearly instantaneous to far longer than the age of the universe.
The decaying nucleus is called the parent radionuclide (or parent radioisotope), and the process produces at least one daughter nuclide. Except for gamma decay or internal conversion from a nuclear excited state, the decay is a nuclear transmutation resulting in a daughter containing a different number of protons or neutrons (or both). When the number of protons changes, an atom of a different chemical element is created.
There are 28 naturally occurring chemical elements on Earth that are radioactive, consisting of 35 radionuclides (seven elements have two different radionuclides each) that date before the time of formation of the Solar System. These 35 are known as primordial radionuclides. Well-known examples are uranium and thorium, but also included are naturally occurring long-lived radioisotopes, such as potassium-40. Each of the heavy primordial radionuclides participates in one of the four decay chains.
Henri Poincaré laid the seeds for the discovery of radioactivity through his interest in and studies of X-rays, which significantly influenced physicist Henri Becquerel. Radioactivity was discovered in 1896 by Becquerel and independently by Marie Curie, while working with phosphorescent materials. These materials glow in the dark after exposure to light, and Becquerel suspected that the glow produced in cathode-ray tubes by X-rays might be associated with phosphorescence. He wrapped a photographic plate in black paper and placed various phosphorescent salts on it. All results were negative until he used uranium salts. The uranium salts caused a blackening of the plate in spite of the plate being wrapped in black paper. Curie named the radiation rayons de Becquerel, "Becquerel Rays" and showed these rays were a property of atoms.
While X-rays were produced using electrical energy, the source of energy for radiation was a mystery. In 1899, Julius Elster and Hans Geitel performed key experiments to find the energy source for radioactivity, excluding extraction of energy from air by measurements in a vacuum and extraction of energy from outer space by measurements 300m down a mine in the Harz mountains. If the atoms themselves were the source of energy, this meant the seemingly immutable atoms must be altered when emitting the rays. In 1900 Curie summarized the puzzle of radioactivity as a choice between two equally unlikely possibilities: either energy was not conserved or chemical elements could be transmuted.
Rutherford was the first to realize that all such elements decay in accordance with the same mathematical exponential formula. Rutherford and his student Frederick Soddy were the first to realize that many decay processes resulted in the transmutation of one element to another. Subsequently, the radioactive displacement law of Fajans and Soddy was formulated to describe the products of alpha and beta decay.
The early researchers also discovered that many other chemical elements, besides uranium, have radioactive isotopes. A systematic search for the total radioactivity in uranium ores also guided Pierre and Marie Curie to isolate two new elements: polonium and radium. Except for the radioactivity of radium, the chemical similarity of radium to barium made these two elements difficult to distinguish.
Radioactive decay
Radioactive decay (also known as nuclear decay, radioactivity, radioactive disintegration, or nuclear disintegration) is the process by which an unstable atomic nucleus loses energy by radiation. A material containing unstable nuclei is considered radioactive. Three of the most common types of decay are alpha, beta, and gamma decay. The weak force is the mechanism that is responsible for beta decay, while the other two are governed by the electromagnetic and nuclear forces.
Radioactive decay is a random process at the level of single atoms. According to quantum theory, it is impossible to predict when a particular atom will decay, regardless of how long the atom has existed. However, for a significant number of identical atoms, the overall decay rate can be expressed as a decay constant or as a half-life. The half-lives of radioactive atoms have a huge range: from nearly instantaneous to far longer than the age of the universe.
The decaying nucleus is called the parent radionuclide (or parent radioisotope), and the process produces at least one daughter nuclide. Except for gamma decay or internal conversion from a nuclear excited state, the decay is a nuclear transmutation resulting in a daughter containing a different number of protons or neutrons (or both). When the number of protons changes, an atom of a different chemical element is created.
There are 28 naturally occurring chemical elements on Earth that are radioactive, consisting of 35 radionuclides (seven elements have two different radionuclides each) that date before the time of formation of the Solar System. These 35 are known as primordial radionuclides. Well-known examples are uranium and thorium, but also included are naturally occurring long-lived radioisotopes, such as potassium-40. Each of the heavy primordial radionuclides participates in one of the four decay chains.
Henri Poincaré laid the seeds for the discovery of radioactivity through his interest in and studies of X-rays, which significantly influenced physicist Henri Becquerel. Radioactivity was discovered in 1896 by Becquerel and independently by Marie Curie, while working with phosphorescent materials. These materials glow in the dark after exposure to light, and Becquerel suspected that the glow produced in cathode-ray tubes by X-rays might be associated with phosphorescence. He wrapped a photographic plate in black paper and placed various phosphorescent salts on it. All results were negative until he used uranium salts. The uranium salts caused a blackening of the plate in spite of the plate being wrapped in black paper. Curie named the radiation rayons de Becquerel, "Becquerel Rays" and showed these rays were a property of atoms.
While X-rays were produced using electrical energy, the source of energy for radiation was a mystery. In 1899, Julius Elster and Hans Geitel performed key experiments to find the energy source for radioactivity, excluding extraction of energy from air by measurements in a vacuum and extraction of energy from outer space by measurements 300m down a mine in the Harz mountains. If the atoms themselves were the source of energy, this meant the seemingly immutable atoms must be altered when emitting the rays. In 1900 Curie summarized the puzzle of radioactivity as a choice between two equally unlikely possibilities: either energy was not conserved or chemical elements could be transmuted.
Rutherford was the first to realize that all such elements decay in accordance with the same mathematical exponential formula. Rutherford and his student Frederick Soddy were the first to realize that many decay processes resulted in the transmutation of one element to another. Subsequently, the radioactive displacement law of Fajans and Soddy was formulated to describe the products of alpha and beta decay.
The early researchers also discovered that many other chemical elements, besides uranium, have radioactive isotopes. A systematic search for the total radioactivity in uranium ores also guided Pierre and Marie Curie to isolate two new elements: polonium and radium. Except for the radioactivity of radium, the chemical similarity of radium to barium made these two elements difficult to distinguish.