Recent from talks
Knowledge base stats:
Talk channels stats:
Members stats:
ATP7A
ATP7A, also known as Menkes' protein (MNK), is a copper-transporting P-type ATPase which uses the energy arising from ATP hydrolysis to transport Cu(I) across cell membranes. The ATP7A protein is a transmembrane protein and is expressed in the intestine and all tissues except liver. In the intestine, ATP7A regulates Cu(I) absorption in the human body by transporting Cu(I) from the small intestine into the blood. In other tissues, ATP7A shuttles between the Golgi apparatus and the cell membrane to maintain proper Cu(I) concentrations (since there is no free Cu(I) in the cell, Cu(I) ions are all tightly bound) in the cell and provides certain enzymes with Cu(I) (e.g. peptidyl-α-monooxygenase, tyrosinase, and lysyl oxidase). The X-linked, inherited, lethal genetic disorder of the ATP7A gene causes Menkes disease, a copper deficiency resulting in early childhood death.
The ATP7A gene is located on the long (q) arm of the X chromosome at band Xq21.1. The encoded ATP7A protein has 1,500 amino acids. At least 12 disease-causing mutations in this gene have been discovered. Mutations/additions/deletions of this gene often cause copper deficiency, which leads to progressive neurodegeneration and death in children.
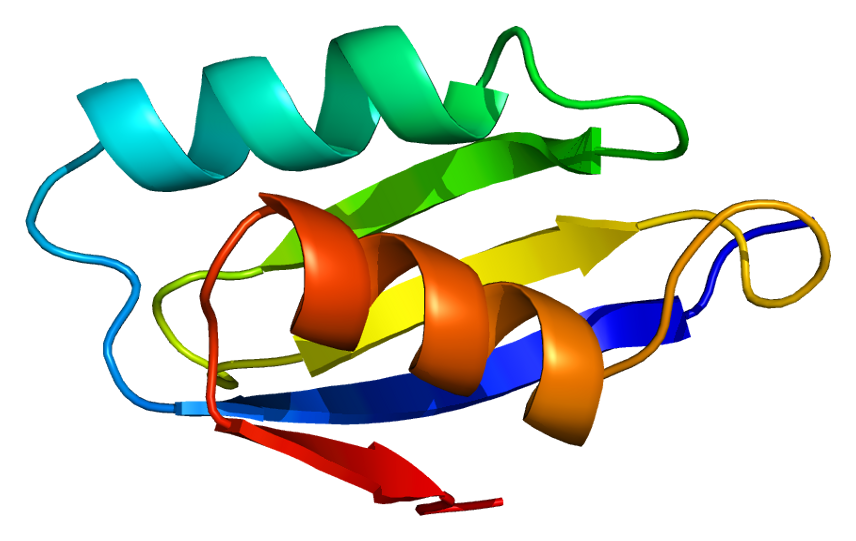
ATP7A is a transmembrane protein with the N- and C-termini both oriented towards the cytosol (see picture). It is highly homologous to protein ATP7B. ATP7A contains three major functional domains:
Many motifs in the ATP7A structure are conserved:
Between transmembrane segments 6 and 7 is a large cytoplasmic loop, where three motifs are located: DKTG, SEHPL, and GDGXND.
The six Cu(I)-binding sites at the N-terminal bind one Cu(I) each. This binding site is not specific for Cu(I) and can bind various transition metal ions. Cd(II), Au(III) and Hg(II) bind to the binding site more tightly than does Zn(II), whereas Mn(II) and Ni(II) have lower affinities relative to Zn(II). In the case of Cu(I), a possible cooperative-binding mechanism is observed. When the Cu(I) concentration is low, Cu(I) has a lower affinity for ATP7A compared to Zn(II); as the Cu(I) concentration increases, a dramatic increasing affinity of Cu(I) for the protein is observed.
The two cysteine (C) residues in each Cu(I)-binding site are coordinated to Cu(I) with a S-Cu(I)-S angle between 120 and 180° and a Cu-S distance of 2.16 Å. Experimental results from a homologous protein ATP7B suggests that reducing reagents are involved, and upon Cu(I) binding the disulfide bonding between the cysteine residues is broken as cysteine starts to bind to Cu(I), leading to a series of conformational changes at the N-terminal of the protein, and possibly activating the Cu(I)-transporting activity of other cytosolic loops.
Of the six copper(I)-binding sites, two are considered enough for the function of Cu(I) transport. The reason why there are six binding sites remains not fully understood. However, some scientists have proposed that the other four sites may serve as a Cu(I) concentration detector.
Hub AI
ATP7A AI simulator
(@ATP7A_simulator)
ATP7A
ATP7A, also known as Menkes' protein (MNK), is a copper-transporting P-type ATPase which uses the energy arising from ATP hydrolysis to transport Cu(I) across cell membranes. The ATP7A protein is a transmembrane protein and is expressed in the intestine and all tissues except liver. In the intestine, ATP7A regulates Cu(I) absorption in the human body by transporting Cu(I) from the small intestine into the blood. In other tissues, ATP7A shuttles between the Golgi apparatus and the cell membrane to maintain proper Cu(I) concentrations (since there is no free Cu(I) in the cell, Cu(I) ions are all tightly bound) in the cell and provides certain enzymes with Cu(I) (e.g. peptidyl-α-monooxygenase, tyrosinase, and lysyl oxidase). The X-linked, inherited, lethal genetic disorder of the ATP7A gene causes Menkes disease, a copper deficiency resulting in early childhood death.
The ATP7A gene is located on the long (q) arm of the X chromosome at band Xq21.1. The encoded ATP7A protein has 1,500 amino acids. At least 12 disease-causing mutations in this gene have been discovered. Mutations/additions/deletions of this gene often cause copper deficiency, which leads to progressive neurodegeneration and death in children.
ATP7A is a transmembrane protein with the N- and C-termini both oriented towards the cytosol (see picture). It is highly homologous to protein ATP7B. ATP7A contains three major functional domains:
Many motifs in the ATP7A structure are conserved:
Between transmembrane segments 6 and 7 is a large cytoplasmic loop, where three motifs are located: DKTG, SEHPL, and GDGXND.
The six Cu(I)-binding sites at the N-terminal bind one Cu(I) each. This binding site is not specific for Cu(I) and can bind various transition metal ions. Cd(II), Au(III) and Hg(II) bind to the binding site more tightly than does Zn(II), whereas Mn(II) and Ni(II) have lower affinities relative to Zn(II). In the case of Cu(I), a possible cooperative-binding mechanism is observed. When the Cu(I) concentration is low, Cu(I) has a lower affinity for ATP7A compared to Zn(II); as the Cu(I) concentration increases, a dramatic increasing affinity of Cu(I) for the protein is observed.
The two cysteine (C) residues in each Cu(I)-binding site are coordinated to Cu(I) with a S-Cu(I)-S angle between 120 and 180° and a Cu-S distance of 2.16 Å. Experimental results from a homologous protein ATP7B suggests that reducing reagents are involved, and upon Cu(I) binding the disulfide bonding between the cysteine residues is broken as cysteine starts to bind to Cu(I), leading to a series of conformational changes at the N-terminal of the protein, and possibly activating the Cu(I)-transporting activity of other cytosolic loops.
Of the six copper(I)-binding sites, two are considered enough for the function of Cu(I) transport. The reason why there are six binding sites remains not fully understood. However, some scientists have proposed that the other four sites may serve as a Cu(I) concentration detector.