Recent from talks
Knowledge base stats:
Talk channels stats:
Members stats:
Α-Bungarotoxin
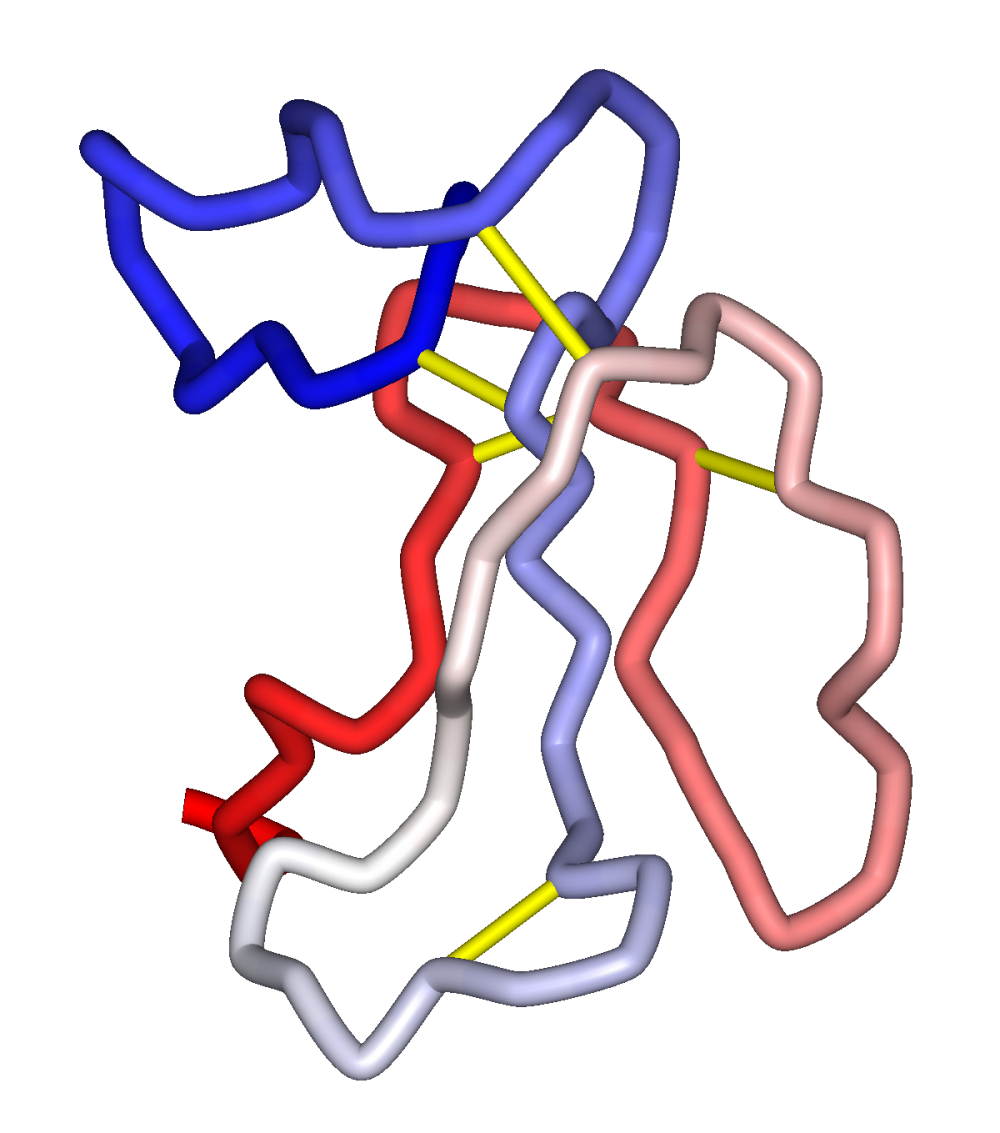
α-Bungarotoxin is one of the bungarotoxins, components of the venom of the elapid Taiwanese banded krait snake (Bungarus multicinctus). It is a type of α-neurotoxin, a neurotoxic protein that is known to bind competitively and in a relatively irreversible manner to the nicotinic acetylcholine receptor found at the neuromuscular junction, causing paralysis, respiratory failure, and death in the victim. It has also been shown to play an antagonistic role in the binding of the α7 nicotinic acetylcholine receptor in the brain, and as such has numerous applications in neuroscience research.
Bungarotoxins are a group of toxins that are closely related with the neurotoxic proteins predominantly present in the venom of kraits. These toxins are directly linked to the three-finger toxin superfamily. Among them, α-bungarotoxin (α-BTX) stands out, being a peptide toxin produced by the Taiwanese banded krait, also known as the many-banded krait or the Taiwanese or Chinese krait.
The venom of the many-banded krait, like the majority of the snake venoms, involves a combination of proteins that together lead to a remarkable range of neurologic consequences. The Elapid snake family is known for their potent α-neurotoxic venom, which has a postsynaptic mechanism of action.[citation needed] These neurotoxins primarily affect the nervous system, blocking the nerve impulse transmission, leading to paralysis and potentially death if untreated.
In South and Southeast Asia, envenomation from a many-banded krait bite is a common and life-threatening medical condition when not promptly treated. Nevertheless, krait bites usually take place at night and do not show any local symptoms, so victims are not aware of the bite. This can delay medical care, which makes it the major cause of mortality associated with krait venom.
The first time that the many-banded krait was described was in 1861 by the scientist Edward Blyth. It was characterized by its distinctive black-and-white banded pattern along its body, with a maximum length of 1.85 m. This very venomous species is found in central and southern China and Southeast Asia. Their venom contains various neurotoxins, including α-BTX.
According to later research on its mechanism of action, α-bungarotoxin binds irreversibly to the postsynaptic nicotinic acetylcholine receptor (nAChR) at the neuromuscular junction. It inhibits the action of acetylcholine competitively, leading to respiratory failure, paralysis and even death.
α-bungarotoxin specifically targets the nervous system, interfering with the nerve impulse transmission. The primary target of the toxin is the neuromuscular junction of skeletal muscles, where the motor nerve terminal and the nicotinic acetylcholine receptor are the major target sites. Their neurotoxic effect is often referred to as resistant neurotoxicity. This is because of the damage caused to nerve terminals that leads to acetylcholine depletion at the neuromuscular junction. The regeneration of the synapses can take days, which prolongs the paralysis and recovery process for the victim. In addition, the severity of the paralysis ranges from mild to life-threatening depending on the degree of envenomation, its composition and the early therapeutic intervention.
Antivenom therapy is the current standard treatment for snake envenoming. In China, the Bungarus multicinctus monovalent antivenom (BMMAV) is produced and, in Taiwan the Neuro bivalent antivenom (NBAV). Both antivenoms are immunoreactive to the neurotoxins found in the venom, including the α-BTX, which neutralize the venom lethality. BMMAV is specifically designed to neutralize the venom of the Bungarus multicinctus, therefore being more efficacious compared to NBAV. On the other hand, NBAV targets the venom from multiple species of snakes that produce neurotoxic effects, including the Bungarus multicinctus. The use of BMMAV or NBAV might differ based on availability, regional protocols and the specific venomous snake that is present in the area.
Hub AI
Α-Bungarotoxin AI simulator
(@Α-Bungarotoxin_simulator)
Α-Bungarotoxin
α-Bungarotoxin is one of the bungarotoxins, components of the venom of the elapid Taiwanese banded krait snake (Bungarus multicinctus). It is a type of α-neurotoxin, a neurotoxic protein that is known to bind competitively and in a relatively irreversible manner to the nicotinic acetylcholine receptor found at the neuromuscular junction, causing paralysis, respiratory failure, and death in the victim. It has also been shown to play an antagonistic role in the binding of the α7 nicotinic acetylcholine receptor in the brain, and as such has numerous applications in neuroscience research.
Bungarotoxins are a group of toxins that are closely related with the neurotoxic proteins predominantly present in the venom of kraits. These toxins are directly linked to the three-finger toxin superfamily. Among them, α-bungarotoxin (α-BTX) stands out, being a peptide toxin produced by the Taiwanese banded krait, also known as the many-banded krait or the Taiwanese or Chinese krait.
The venom of the many-banded krait, like the majority of the snake venoms, involves a combination of proteins that together lead to a remarkable range of neurologic consequences. The Elapid snake family is known for their potent α-neurotoxic venom, which has a postsynaptic mechanism of action.[citation needed] These neurotoxins primarily affect the nervous system, blocking the nerve impulse transmission, leading to paralysis and potentially death if untreated.
In South and Southeast Asia, envenomation from a many-banded krait bite is a common and life-threatening medical condition when not promptly treated. Nevertheless, krait bites usually take place at night and do not show any local symptoms, so victims are not aware of the bite. This can delay medical care, which makes it the major cause of mortality associated with krait venom.
The first time that the many-banded krait was described was in 1861 by the scientist Edward Blyth. It was characterized by its distinctive black-and-white banded pattern along its body, with a maximum length of 1.85 m. This very venomous species is found in central and southern China and Southeast Asia. Their venom contains various neurotoxins, including α-BTX.
According to later research on its mechanism of action, α-bungarotoxin binds irreversibly to the postsynaptic nicotinic acetylcholine receptor (nAChR) at the neuromuscular junction. It inhibits the action of acetylcholine competitively, leading to respiratory failure, paralysis and even death.
α-bungarotoxin specifically targets the nervous system, interfering with the nerve impulse transmission. The primary target of the toxin is the neuromuscular junction of skeletal muscles, where the motor nerve terminal and the nicotinic acetylcholine receptor are the major target sites. Their neurotoxic effect is often referred to as resistant neurotoxicity. This is because of the damage caused to nerve terminals that leads to acetylcholine depletion at the neuromuscular junction. The regeneration of the synapses can take days, which prolongs the paralysis and recovery process for the victim. In addition, the severity of the paralysis ranges from mild to life-threatening depending on the degree of envenomation, its composition and the early therapeutic intervention.
Antivenom therapy is the current standard treatment for snake envenoming. In China, the Bungarus multicinctus monovalent antivenom (BMMAV) is produced and, in Taiwan the Neuro bivalent antivenom (NBAV). Both antivenoms are immunoreactive to the neurotoxins found in the venom, including the α-BTX, which neutralize the venom lethality. BMMAV is specifically designed to neutralize the venom of the Bungarus multicinctus, therefore being more efficacious compared to NBAV. On the other hand, NBAV targets the venom from multiple species of snakes that produce neurotoxic effects, including the Bungarus multicinctus. The use of BMMAV or NBAV might differ based on availability, regional protocols and the specific venomous snake that is present in the area.