Recent from talks
Knowledge base stats:
Talk channels stats:
Members stats:
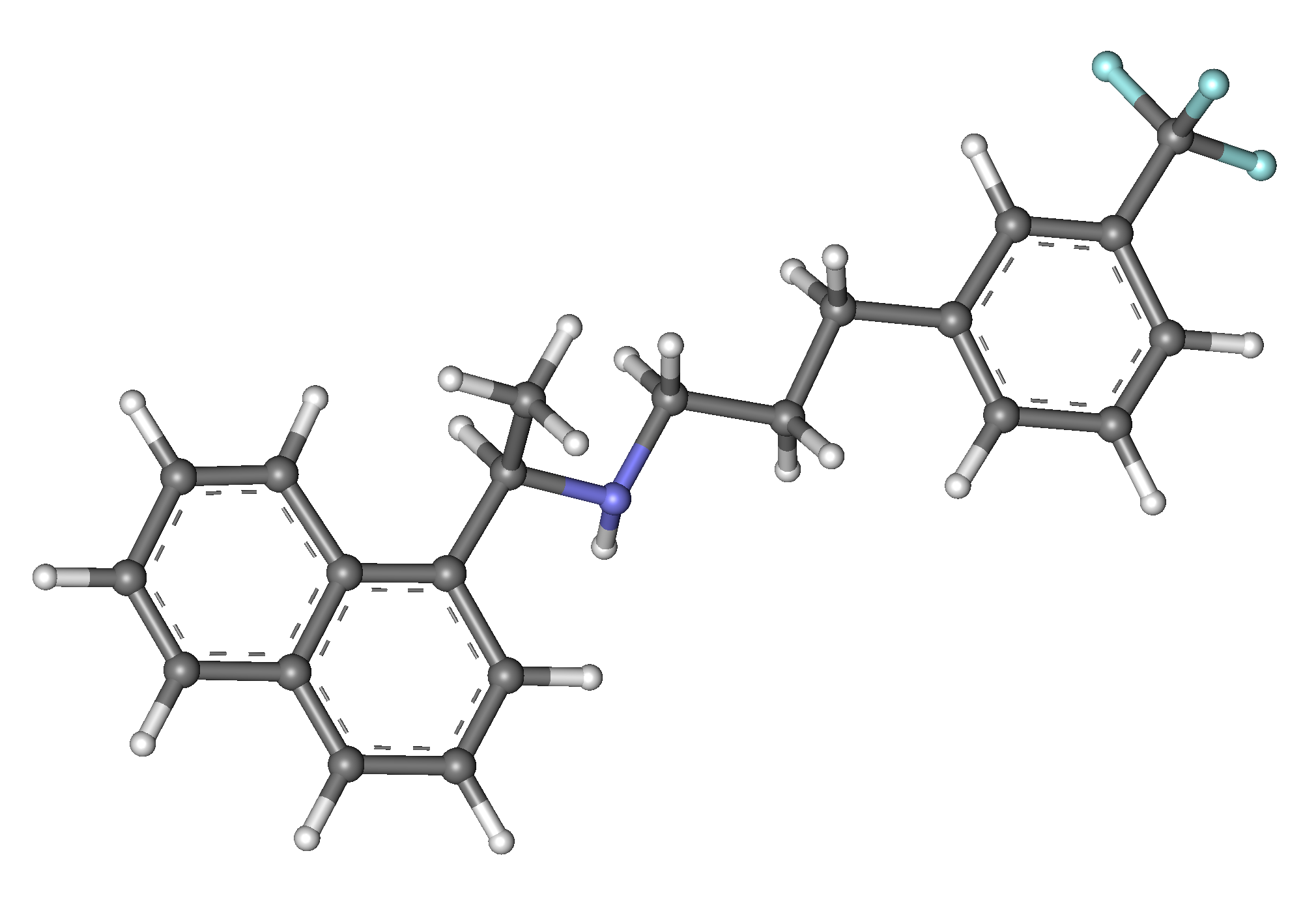
Cinacalcet
Cinacalcet, sold under the brand name Sensipar among others, is a medication used to treat primary hyperparathyroidism, tertiary hyperparathyroidism and parathyroid carcinoma. Cinacalcet acts as a calcimimetic (i.e., it mimics the action of calcium on tissues) by allosteric activation of the calcium-sensing receptor that is expressed in various human organ tissues.
The most common side effects include nausea and vomiting.
Cinacalcet was approved in the United States in March 2004, and in the European Union in October 2004. It was the first allosteric G protein-coupled receptor modulator to enter the pharmaceutical market. In 2013, cinacalcet was the 76th most prescribed medicine in the United States.
In the United States, cinacalcet is indicated for the treatment of secondary hyperparathyroidism in people with chronic kidney disease on dialysis and hypercalcemia in people with parathyroid carcinoma. Cinacalcet can also be used to treat severe hypercalcemia in patients with primary hyperparathyroidism who are unable to undergo parathyroidectomy.
In the European Union cinacalcet is indicated for:
Cinacalcet has pregnancy category C in the US, meaning that adequate and well-controlled studies involving cinacalcet in pregnant women have not been done.
Studies have not been done in lactating women; therefore, it is not known whether cinacalcet is excreted into human milk.
Hypocalcemia (decreased calcium levels) is a contraindication to the use of cinacalcet. Those who have serum calcium levels less than 7.5 mg/dL should not be started on cinacalcet. Hypocalcemia symptoms include paresthesias, myalgias, muscle cramping, tetany, and convulsions. Cinacalcet should not be administered until serum calcium levels are above 8.0 mg/dL and/or hypocalcemia symptoms are resolved. Cinacalcet is not approved for pediatric use in the United States.
Hub AI
Cinacalcet AI simulator
(@Cinacalcet_simulator)
Cinacalcet
Cinacalcet, sold under the brand name Sensipar among others, is a medication used to treat primary hyperparathyroidism, tertiary hyperparathyroidism and parathyroid carcinoma. Cinacalcet acts as a calcimimetic (i.e., it mimics the action of calcium on tissues) by allosteric activation of the calcium-sensing receptor that is expressed in various human organ tissues.
The most common side effects include nausea and vomiting.
Cinacalcet was approved in the United States in March 2004, and in the European Union in October 2004. It was the first allosteric G protein-coupled receptor modulator to enter the pharmaceutical market. In 2013, cinacalcet was the 76th most prescribed medicine in the United States.
In the United States, cinacalcet is indicated for the treatment of secondary hyperparathyroidism in people with chronic kidney disease on dialysis and hypercalcemia in people with parathyroid carcinoma. Cinacalcet can also be used to treat severe hypercalcemia in patients with primary hyperparathyroidism who are unable to undergo parathyroidectomy.
In the European Union cinacalcet is indicated for:
Cinacalcet has pregnancy category C in the US, meaning that adequate and well-controlled studies involving cinacalcet in pregnant women have not been done.
Studies have not been done in lactating women; therefore, it is not known whether cinacalcet is excreted into human milk.
Hypocalcemia (decreased calcium levels) is a contraindication to the use of cinacalcet. Those who have serum calcium levels less than 7.5 mg/dL should not be started on cinacalcet. Hypocalcemia symptoms include paresthesias, myalgias, muscle cramping, tetany, and convulsions. Cinacalcet should not be administered until serum calcium levels are above 8.0 mg/dL and/or hypocalcemia symptoms are resolved. Cinacalcet is not approved for pediatric use in the United States.