Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Diethyl phosphorochloridate AI simulator
(@Diethyl phosphorochloridate_simulator)
Hub AI
Diethyl phosphorochloridate AI simulator
(@Diethyl phosphorochloridate_simulator)
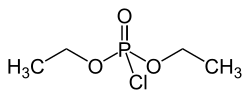
Diethyl phosphorochloridate
Diethyl chlorophosphate is an organophosphorus compound with the formula (C2H5O)2P(O)Cl. As a reagent in organic synthesis, it is used to convert alcohols to the corresponding diethylphosphate esters. It is a colorless liquid with a fruity odor. It is a corrosive, and as a cholinesterase inhibitor, highly toxic through dermal absorption. The molecule is tetrahedral.
The compound is prepared by the chlorination of diethylphosphite with carbon tetrachloride (Atherton–Todd reaction).
The compound is electrophilic. Controlled hydrolysis gives tetraethyl pyrophosphate. Alcohols react to give phosphate esters:
The reagent is routinely employed in organic synthesis for phosphorylation of carboxylates, alcohols, and amines.
Diethyl phosphorochloridate
Diethyl chlorophosphate is an organophosphorus compound with the formula (C2H5O)2P(O)Cl. As a reagent in organic synthesis, it is used to convert alcohols to the corresponding diethylphosphate esters. It is a colorless liquid with a fruity odor. It is a corrosive, and as a cholinesterase inhibitor, highly toxic through dermal absorption. The molecule is tetrahedral.
The compound is prepared by the chlorination of diethylphosphite with carbon tetrachloride (Atherton–Todd reaction).
The compound is electrophilic. Controlled hydrolysis gives tetraethyl pyrophosphate. Alcohols react to give phosphate esters:
The reagent is routinely employed in organic synthesis for phosphorylation of carboxylates, alcohols, and amines.