Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Lurasidone AI simulator
(@Lurasidone_simulator)
Hub AI
Lurasidone AI simulator
(@Lurasidone_simulator)
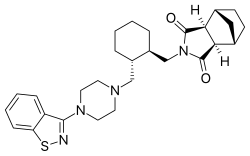
Lurasidone
Lurasidone, sold under the brand name Latuda among others, is an atypical antipsychotic medication used to treat schizophrenia and bipolar depression. It is taken by mouth.
Common side effects include sedation, indigestion, nausea, and insomnia. At higher dosages, there is an increased risk for restlessness and movement problems. Serious side effects are valid for all atypical antipsychotics and may include the potentially permanent movement disorder tardive dyskinesia, as well as neuroleptic malignant syndrome, angioedema, and high blood sugar levels. Although lurasidone is less likely to cause high blood sugar levels in most patients, hyperosmolar hyperglycemic syndrome may occur. In older people with psychosis as a result of dementia, it may increase the risk of dying. Use during pregnancy is of unclear safety.
Lurasidone was first approved for medical use in the United States in 2010, for treating schizophrenia. In 2013, it was approved in Canada and by the U.S. Food and Drug Administration (FDA) to treat bipolar depression, either as monotherapy or adjunctively with lithium or valproate. It is not FDA approved for treating manic symptoms associated with bipolar disorder despite its FDA approval for treating bipolar depression. Generic versions were approved in the United States in 2019, and became available in 2023. In 2021, it was the 193rd most commonly prescribed medication in the United States, with more than 2 million prescriptions.
Lurasidone is used to treat schizophrenia and bipolar disorder. In bipolar disorder, it has been studied both as a monotherapy and adjunctive treatment to lithium or valproate.
The European Medicines Agency approved lurasidone for the treatment of schizophrenia for people aged 13 years and older. Its use in Europe for bipolar disorder is considered off-label. In the United States, it is used to treat schizophrenia for people aged 13 years and older. In July 2013, lurasidone received approval for bipolar I depression. This includes depressive episodes of bipolar disorder age 10 and over as a monotherapy, and in conjunction with lithium or valproate in adults.
In June 2020, lurasidone was approved in Japan, eight years after its first approval in the United States. In Japan it is approved for bipolar depression and schizophrenia. Lurasidone is not approved by the Food and Drug Administration (FDA) for the treatment of behavior disorders in older adults with dementia.
Few available atypical antipsychotics are known to possess antidepressant efficacy in bipolar disorder (with the notable exceptions being cariprazine, quetiapine, olanzapine and possibly asenapine) as a monotherapy. The majority of atypical antipsychotics are known to possess significant antimanic activity, however lurasidone is unusual as it was never studied for acute mania.
In the early post approval period lurasidone-treated patients with bipolar disorder were retrospectively found to have more complex clinical profiles, comorbidities, and prior treatment history compared to patients initiated with other atypical antipsychotics. The study authors suggest this may be due to "the overall clinical profile of lurasidone, the role perceived for lurasidone in the therapeutic armamentarium by practitioners, and the recent introduction of lurasidone into clinical practice during the study period".
Lurasidone
Lurasidone, sold under the brand name Latuda among others, is an atypical antipsychotic medication used to treat schizophrenia and bipolar depression. It is taken by mouth.
Common side effects include sedation, indigestion, nausea, and insomnia. At higher dosages, there is an increased risk for restlessness and movement problems. Serious side effects are valid for all atypical antipsychotics and may include the potentially permanent movement disorder tardive dyskinesia, as well as neuroleptic malignant syndrome, angioedema, and high blood sugar levels. Although lurasidone is less likely to cause high blood sugar levels in most patients, hyperosmolar hyperglycemic syndrome may occur. In older people with psychosis as a result of dementia, it may increase the risk of dying. Use during pregnancy is of unclear safety.
Lurasidone was first approved for medical use in the United States in 2010, for treating schizophrenia. In 2013, it was approved in Canada and by the U.S. Food and Drug Administration (FDA) to treat bipolar depression, either as monotherapy or adjunctively with lithium or valproate. It is not FDA approved for treating manic symptoms associated with bipolar disorder despite its FDA approval for treating bipolar depression. Generic versions were approved in the United States in 2019, and became available in 2023. In 2021, it was the 193rd most commonly prescribed medication in the United States, with more than 2 million prescriptions.
Lurasidone is used to treat schizophrenia and bipolar disorder. In bipolar disorder, it has been studied both as a monotherapy and adjunctive treatment to lithium or valproate.
The European Medicines Agency approved lurasidone for the treatment of schizophrenia for people aged 13 years and older. Its use in Europe for bipolar disorder is considered off-label. In the United States, it is used to treat schizophrenia for people aged 13 years and older. In July 2013, lurasidone received approval for bipolar I depression. This includes depressive episodes of bipolar disorder age 10 and over as a monotherapy, and in conjunction with lithium or valproate in adults.
In June 2020, lurasidone was approved in Japan, eight years after its first approval in the United States. In Japan it is approved for bipolar depression and schizophrenia. Lurasidone is not approved by the Food and Drug Administration (FDA) for the treatment of behavior disorders in older adults with dementia.
Few available atypical antipsychotics are known to possess antidepressant efficacy in bipolar disorder (with the notable exceptions being cariprazine, quetiapine, olanzapine and possibly asenapine) as a monotherapy. The majority of atypical antipsychotics are known to possess significant antimanic activity, however lurasidone is unusual as it was never studied for acute mania.
In the early post approval period lurasidone-treated patients with bipolar disorder were retrospectively found to have more complex clinical profiles, comorbidities, and prior treatment history compared to patients initiated with other atypical antipsychotics. The study authors suggest this may be due to "the overall clinical profile of lurasidone, the role perceived for lurasidone in the therapeutic armamentarium by practitioners, and the recent introduction of lurasidone into clinical practice during the study period".