Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Leptophos AI simulator
(@Leptophos_simulator)
Hub AI
Leptophos AI simulator
(@Leptophos_simulator)
Leptophos
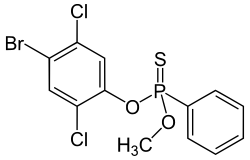
Leptophos (O-(4-bromo-2,5-dichlorophenyl) O-methyl phenylphosphonothioate) belongs to the organophosphates and at room temperature it is a stable white solid. It is also known as Phosvel, Abar and Vcs 506. Leptophos was primarily used as a pesticide and fungicide for rice, cotton, fruit, and vegetables until its use was discontinued in 1975 in the United States, but still sold in South-Eastern Asia in 1981.
Leptophos was first discovered to be toxic in 1974 when more than 1000 water buffaloes died after exposure to leptophos in Egypt. In response to this event, the effect of leptophos was investigated on chickens, mice, and sheep.
In Egypt, leptophos used on cotton in 1971 caused the death of more than 1000 water buffaloes and a number of farmers. The compound was never registered for domestic use by the Environmental Protection Agency (EPA) but was exported from the U.S. to at least 30 countries. Leptophos was discontinued for use in late 1975 due to its high toxicity. Between 1971 and 1976 the U.S. used $4 million in United States Agency for International Development funds to ship 13.9 million pounds of leptophos and other banned pesticides to 50 countries.[citation needed] In 1975 U.S. companies alone, exported over 3 million pounds of leptophos.
In 1976, workers in the Velsicol's chemical plant in Bayport, Texas, reported serious neurological symptoms, the Phosvel zombies, and filed a lawsuit against the company. When Colombia banned leptophos in 1977, the American company Velsicol stopped the production and shipped its Colombian stocks to El Salvador. No prohibitions exist in El Salvador. In other instances leptophos was imported to Costa Rica via Mexico and Panama, and until 1981 leptophos was being sold in Indonesia.
It is stable at normal temperatures; at 180 degrees Celsius 85 percent of the material is decomposed in 5 hours, and at 208 degrees Celsius it decomposes in 2 hours. The main product of thermal decomposition is the S-methyl isomer O-(4-bromo-2,5-chlorophenyl) S-methyl phenylphosphonothioate. Leptophos is hydrolyzed slowly under alkaline conditions. The material is stable toward acid.[citation needed]
In the laboratory, when irradiated with high intensity UV light in the presence of a strong UV sensitizer, leptophos is rapidly converted first to O-(2,5-dichlorophenyl) O-methylphenyl-phosphonothioate, referred to as the dichloro-photoproduct, and then to a material with the empirical formula C13H10ClO2PS (tentatively identified as 3-chloro-6-methoxydibenz [1,2]-oxaphosphorin-6-thione or O-methyl-O,P-(4-chlorobiphenyl-2,6-ylene) phosphonothioate and referred to as the monochloro-photoproduct. UV light increases the rate of hydrolysis under field conditions.
There are multiple ways to synthesize leptophos. One of the methods that is possible for doing that is to let O-methyl phenylthiophosponyl chloride react with 4-bromo-2,5-dichlorophenol:
C7H8ClOPS + C6H3BrCl2O → C13H10BrCl2O2PS + HCl.
Leptophos
Leptophos (O-(4-bromo-2,5-dichlorophenyl) O-methyl phenylphosphonothioate) belongs to the organophosphates and at room temperature it is a stable white solid. It is also known as Phosvel, Abar and Vcs 506. Leptophos was primarily used as a pesticide and fungicide for rice, cotton, fruit, and vegetables until its use was discontinued in 1975 in the United States, but still sold in South-Eastern Asia in 1981.
Leptophos was first discovered to be toxic in 1974 when more than 1000 water buffaloes died after exposure to leptophos in Egypt. In response to this event, the effect of leptophos was investigated on chickens, mice, and sheep.
In Egypt, leptophos used on cotton in 1971 caused the death of more than 1000 water buffaloes and a number of farmers. The compound was never registered for domestic use by the Environmental Protection Agency (EPA) but was exported from the U.S. to at least 30 countries. Leptophos was discontinued for use in late 1975 due to its high toxicity. Between 1971 and 1976 the U.S. used $4 million in United States Agency for International Development funds to ship 13.9 million pounds of leptophos and other banned pesticides to 50 countries.[citation needed] In 1975 U.S. companies alone, exported over 3 million pounds of leptophos.
In 1976, workers in the Velsicol's chemical plant in Bayport, Texas, reported serious neurological symptoms, the Phosvel zombies, and filed a lawsuit against the company. When Colombia banned leptophos in 1977, the American company Velsicol stopped the production and shipped its Colombian stocks to El Salvador. No prohibitions exist in El Salvador. In other instances leptophos was imported to Costa Rica via Mexico and Panama, and until 1981 leptophos was being sold in Indonesia.
It is stable at normal temperatures; at 180 degrees Celsius 85 percent of the material is decomposed in 5 hours, and at 208 degrees Celsius it decomposes in 2 hours. The main product of thermal decomposition is the S-methyl isomer O-(4-bromo-2,5-chlorophenyl) S-methyl phenylphosphonothioate. Leptophos is hydrolyzed slowly under alkaline conditions. The material is stable toward acid.[citation needed]
In the laboratory, when irradiated with high intensity UV light in the presence of a strong UV sensitizer, leptophos is rapidly converted first to O-(2,5-dichlorophenyl) O-methylphenyl-phosphonothioate, referred to as the dichloro-photoproduct, and then to a material with the empirical formula C13H10ClO2PS (tentatively identified as 3-chloro-6-methoxydibenz [1,2]-oxaphosphorin-6-thione or O-methyl-O,P-(4-chlorobiphenyl-2,6-ylene) phosphonothioate and referred to as the monochloro-photoproduct. UV light increases the rate of hydrolysis under field conditions.
There are multiple ways to synthesize leptophos. One of the methods that is possible for doing that is to let O-methyl phenylthiophosponyl chloride react with 4-bromo-2,5-dichlorophenol:
C7H8ClOPS + C6H3BrCl2O → C13H10BrCl2O2PS + HCl.