Recent from talks
Knowledge base stats:
Talk channels stats:
Members stats:
PTEN (gene)
PTEN (phosphatase and tensin homolog) is a gene found in humans which encodes for the protein PTEN, also known as phosphatidylinositol-3,4,5-trisphosphate 3-phosphatase. PTEN acts as a tumor suppressor gene through the action of its phosphatase protein product. Mutations of this gene are linked to many cancers, specifically glioblastoma, lung cancer, breast cancer, and prostate cancer. [citation needed] Genes corresponding to PTEN (orthologs) have been identified in most mammals for which complete genome data are available.
The PTEN protein contains both a tensin-like domain and a catalytic domain similar to that of the dual specificity phosphatases. Unlike most protein tyrosine phosphatases, the PTEN protein preferentially dephosphorylates phosphoinositide substrates. Specifically, it catalyzes the conversion of phosphatidylinositol-3,4,5-trisphosphate (PIP3) to phosphatidylinositol 4,5-bisphosphate (PIP2). Decreased PIP3 levels, in turn, lead to decreased activation of the Akt/PKB signaling pathway, an important pathway in cell growth, survival, and proliferation.[citation needed]
PTEN protein is primarily a lipid phosphatase, which dephosphorylates phosphatidylinositol (3,4,5)-trisphosphate (PtdIns (3,4,5)P3 or PIP3) into the biphosphate product phosphatidylinositol 4,5-bisphosphate (PtdIns(4,5)P2 or PIP2). PTEN specifically catalyses the dephosphorylation of the 3` phosphate of the inositol ring in PIP3. This dephosphorylation results in inhibition of the Akt signaling pathway, which plays an important role in regulating cellular behaviors such as cell growth, survival, and migration.[citation needed]
PTEN also has weak protein phosphatase activity, which is also crucial for its role as a tumor suppressor. PTEN's protein phosphatase activity may be involved in the regulation of the cell cycle, preventing cells from growing and dividing too rapidly. There have been numerous reported protein substrates for PTEN, including IRS1 and Dishevelled.
PTEN appears to play a critical role in the DNA damage response and the repair of DNA damage, particularly in double-strand break repair and nucleotide excision repair.
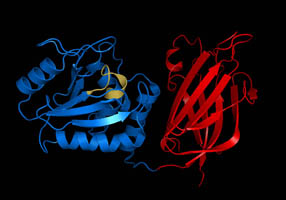
The structure of the core of PTEN (solved by X-ray crystallography, see figure) reveals that it consists primarily of a phosphatase domain and a C2 domain. The phosphatase domain contains the active site, which carries out the enzymatic function of the protein, while the C2 domain binds the phospholipid membrane. Thus PTEN binds the membrane through both its phosphatase and C2 domains, bringing the active site to the membrane-bound PIP3 to dephosphorylate it.
The two domains of PTEN, a protein tyrosine phosphatase domain and a C2 domain, are inherited together as a single unit and thus constitute a superdomain, not only in PTEN but also in various other proteins in fungi, plants and animals, for example, tensin proteins and auxilin.
The active site of PTEN consists of three loops, the TI Loop, the P Loop, and the WPD Loop, all named following the PTPB1 nomenclature. Together they form an unusually deep and wide pocket which allows PTEN to accommodate the bulky phosphatidylinositol 3,4,5-trisphosphate substrate. The dephosphorylation reaction mechanism of PTEN is thought to proceed through a phosphoenzyme intermediate, with the formation of a phosphodiester bond on the active site cysteine, C124.
Hub AI
PTEN (gene) AI simulator
(@PTEN (gene)_simulator)
PTEN (gene)
PTEN (phosphatase and tensin homolog) is a gene found in humans which encodes for the protein PTEN, also known as phosphatidylinositol-3,4,5-trisphosphate 3-phosphatase. PTEN acts as a tumor suppressor gene through the action of its phosphatase protein product. Mutations of this gene are linked to many cancers, specifically glioblastoma, lung cancer, breast cancer, and prostate cancer. [citation needed] Genes corresponding to PTEN (orthologs) have been identified in most mammals for which complete genome data are available.
The PTEN protein contains both a tensin-like domain and a catalytic domain similar to that of the dual specificity phosphatases. Unlike most protein tyrosine phosphatases, the PTEN protein preferentially dephosphorylates phosphoinositide substrates. Specifically, it catalyzes the conversion of phosphatidylinositol-3,4,5-trisphosphate (PIP3) to phosphatidylinositol 4,5-bisphosphate (PIP2). Decreased PIP3 levels, in turn, lead to decreased activation of the Akt/PKB signaling pathway, an important pathway in cell growth, survival, and proliferation.[citation needed]
PTEN protein is primarily a lipid phosphatase, which dephosphorylates phosphatidylinositol (3,4,5)-trisphosphate (PtdIns (3,4,5)P3 or PIP3) into the biphosphate product phosphatidylinositol 4,5-bisphosphate (PtdIns(4,5)P2 or PIP2). PTEN specifically catalyses the dephosphorylation of the 3` phosphate of the inositol ring in PIP3. This dephosphorylation results in inhibition of the Akt signaling pathway, which plays an important role in regulating cellular behaviors such as cell growth, survival, and migration.[citation needed]
PTEN also has weak protein phosphatase activity, which is also crucial for its role as a tumor suppressor. PTEN's protein phosphatase activity may be involved in the regulation of the cell cycle, preventing cells from growing and dividing too rapidly. There have been numerous reported protein substrates for PTEN, including IRS1 and Dishevelled.
PTEN appears to play a critical role in the DNA damage response and the repair of DNA damage, particularly in double-strand break repair and nucleotide excision repair.
The structure of the core of PTEN (solved by X-ray crystallography, see figure) reveals that it consists primarily of a phosphatase domain and a C2 domain. The phosphatase domain contains the active site, which carries out the enzymatic function of the protein, while the C2 domain binds the phospholipid membrane. Thus PTEN binds the membrane through both its phosphatase and C2 domains, bringing the active site to the membrane-bound PIP3 to dephosphorylate it.
The two domains of PTEN, a protein tyrosine phosphatase domain and a C2 domain, are inherited together as a single unit and thus constitute a superdomain, not only in PTEN but also in various other proteins in fungi, plants and animals, for example, tensin proteins and auxilin.
The active site of PTEN consists of three loops, the TI Loop, the P Loop, and the WPD Loop, all named following the PTPB1 nomenclature. Together they form an unusually deep and wide pocket which allows PTEN to accommodate the bulky phosphatidylinositol 3,4,5-trisphosphate substrate. The dephosphorylation reaction mechanism of PTEN is thought to proceed through a phosphoenzyme intermediate, with the formation of a phosphodiester bond on the active site cysteine, C124.