Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Peptide synthesis AI simulator
(@Peptide synthesis_simulator)
Hub AI
Peptide synthesis AI simulator
(@Peptide synthesis_simulator)
Peptide synthesis
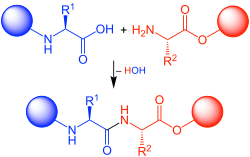
In organic chemistry, peptide synthesis is the production of peptides, compounds where multiple amino acids are linked via amide bonds, also known as peptide bonds. Peptides are chemically synthesized by the condensation reaction of the carboxyl group of one amino acid to the amino group of another. Protecting group strategies are usually necessary to prevent undesirable side reactions with the various amino acid side chains. Chemical peptide synthesis most commonly starts at the carboxyl end of the peptide (C-terminus), and proceeds toward the amino-terminus (N-terminus). Protein biosynthesis (long peptides) in living organisms occurs in the opposite direction.
The chemical synthesis of peptides can be carried out using classical solution-phase techniques, although these have been replaced in most research and development settings by solid phase methods (see below). Solution phase synthesis retains its usefulness in production of small peptides for industrial purposes.
Chemical peptide synthesis facilitates the production of peptides that are difficult to express in bacteria, the incorporation of unnatural amino acids, peptide/protein backbone modification, and the synthesis of peptides containing D-amino acids.
The established method for the production of synthetic peptides is known as solid phase peptide synthesis (SPPS). Pioneered by Robert Bruce Merrifield, SPPS allows the rapid assembly of a peptide chain through stepwise addition of amino acids on a macroscopically insoluble solvent-swollen beaded resin support.
The solid support consists of small (~50 micron diameter), polymeric resin beads functionalized with reactive groups (such as amine or hydroxyl groups) that link the nascent peptide chain to the resin polymer. Since the peptide remains covalently attached to the support throughout the synthesis, excess reagents and soluble side products can be removed by washing and filtration. This approach circumvents the time-consuming isolation of the product peptide after each reaction step that is required when using conventional solution phase synthesis.
Stepwise SPPS proceeds from the C-terminal amino acid residue of the target peptide attached to the resin support. Each amino acid to be coupled to the N-terminus of the resin bound nascent peptide chain must be protected on its alpha amino group, and on any reactive side chain functionalities using appropriate protecting groups such as Boc (acid-labile) or Fmoc (base-labile), depending on the protection strategy used (see below).
The general SPPS procedure is one of repeated cycles of alternate N-terminal deprotection and coupling reactions. The resin can be washed between each steps. Reactions in SPPS are conducted as follows:
SPPS is limited by reaction yields due to the exponential accumulation of by-products, and typically peptides and proteins in the range of 40 or 50 amino acid residues are pushing the limits of synthetic accessibility of SPPS products as homogeneous molecules of defined chemical structure. Synthetic difficulty also is sequence dependent; typically aggregation-prone sequences such as amyloids are difficult to make. Longer peptides can be accessed by using approaches such as native chemical ligation, where two unprotected synthetic peptides can be covalently condensed in aqueous solution.
Peptide synthesis
In organic chemistry, peptide synthesis is the production of peptides, compounds where multiple amino acids are linked via amide bonds, also known as peptide bonds. Peptides are chemically synthesized by the condensation reaction of the carboxyl group of one amino acid to the amino group of another. Protecting group strategies are usually necessary to prevent undesirable side reactions with the various amino acid side chains. Chemical peptide synthesis most commonly starts at the carboxyl end of the peptide (C-terminus), and proceeds toward the amino-terminus (N-terminus). Protein biosynthesis (long peptides) in living organisms occurs in the opposite direction.
The chemical synthesis of peptides can be carried out using classical solution-phase techniques, although these have been replaced in most research and development settings by solid phase methods (see below). Solution phase synthesis retains its usefulness in production of small peptides for industrial purposes.
Chemical peptide synthesis facilitates the production of peptides that are difficult to express in bacteria, the incorporation of unnatural amino acids, peptide/protein backbone modification, and the synthesis of peptides containing D-amino acids.
The established method for the production of synthetic peptides is known as solid phase peptide synthesis (SPPS). Pioneered by Robert Bruce Merrifield, SPPS allows the rapid assembly of a peptide chain through stepwise addition of amino acids on a macroscopically insoluble solvent-swollen beaded resin support.
The solid support consists of small (~50 micron diameter), polymeric resin beads functionalized with reactive groups (such as amine or hydroxyl groups) that link the nascent peptide chain to the resin polymer. Since the peptide remains covalently attached to the support throughout the synthesis, excess reagents and soluble side products can be removed by washing and filtration. This approach circumvents the time-consuming isolation of the product peptide after each reaction step that is required when using conventional solution phase synthesis.
Stepwise SPPS proceeds from the C-terminal amino acid residue of the target peptide attached to the resin support. Each amino acid to be coupled to the N-terminus of the resin bound nascent peptide chain must be protected on its alpha amino group, and on any reactive side chain functionalities using appropriate protecting groups such as Boc (acid-labile) or Fmoc (base-labile), depending on the protection strategy used (see below).
The general SPPS procedure is one of repeated cycles of alternate N-terminal deprotection and coupling reactions. The resin can be washed between each steps. Reactions in SPPS are conducted as follows:
SPPS is limited by reaction yields due to the exponential accumulation of by-products, and typically peptides and proteins in the range of 40 or 50 amino acid residues are pushing the limits of synthetic accessibility of SPPS products as homogeneous molecules of defined chemical structure. Synthetic difficulty also is sequence dependent; typically aggregation-prone sequences such as amyloids are difficult to make. Longer peptides can be accessed by using approaches such as native chemical ligation, where two unprotected synthetic peptides can be covalently condensed in aqueous solution.