Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Phosphoryl chloride AI simulator
(@Phosphoryl chloride_simulator)
Hub AI
Phosphoryl chloride AI simulator
(@Phosphoryl chloride_simulator)
Phosphoryl chloride
Phosphoryl chloride (commonly called phosphorus oxychloride) is a colourless liquid with the formula POCl3. It hydrolyses in moist air releasing phosphoric acid and fumes of hydrogen chloride. It is manufactured industrially on a large scale from phosphorus trichloride and oxygen or phosphorus pentoxide. It is mainly used to make phosphate esters.
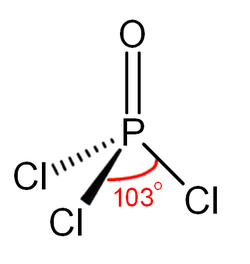
Like phosphate, POCl3 is tetrahedral in shape. It features three P−Cl bonds and one strong P–O bond, with an estimated bond dissociation energy of 533.5 kJ/mol. Unlike in the case of POF3, the Schomaker-Stevenson rule predicts appropriate bond length for the P–O bond only if the P–O bond is treated as a double bond, P=O.[citation needed] More modern treatments explain the tight P–O bond as a combination of lone pair transfer from the phosphorus to the oxygen atom and a dative π back-bond that produces an effective [P+]-[O−] configuration.
Phosphoryl chloride exists as neutral POCl3 molecules in the solid, liquid and gas states. This is unlike phosphorus pentachloride which exists as neutral PCl5 molecules in the gas and liquid states but adopts the ionic form [PCl4]+[PCl6]− (tetrachlorophosphonium hexachlorophosphate(V)) in the solid state. The average bond lengths in the crystal structure of POCl3 are 1.98 Å for P–Cl and 1.46 Å for P=O.
It has a critical pressure of 3.4 atm. With a freezing point of 1 °C and boiling point of 106 °C, the liquid range of POCl3 is rather similar to water. Also like water, POCl3 autoionizes, owing to the reversible formation of [POCl2]+ cations (dichlorooxophosphonium cations) and Cl− anions.
POCl3 reacts with water to give hydrogen chloride and phosphoric acid:
Intermediates in the conversion have been isolated, including pyrophosphoryl chloride, O(−P(=O)Cl2)2.
Upon treatment with excess alcohols and phenols, POCl3 gives phosphate esters:
Such reactions are often performed in the presence of an HCl acceptor such as pyridine or an amine.
Phosphoryl chloride
Phosphoryl chloride (commonly called phosphorus oxychloride) is a colourless liquid with the formula POCl3. It hydrolyses in moist air releasing phosphoric acid and fumes of hydrogen chloride. It is manufactured industrially on a large scale from phosphorus trichloride and oxygen or phosphorus pentoxide. It is mainly used to make phosphate esters.
Like phosphate, POCl3 is tetrahedral in shape. It features three P−Cl bonds and one strong P–O bond, with an estimated bond dissociation energy of 533.5 kJ/mol. Unlike in the case of POF3, the Schomaker-Stevenson rule predicts appropriate bond length for the P–O bond only if the P–O bond is treated as a double bond, P=O.[citation needed] More modern treatments explain the tight P–O bond as a combination of lone pair transfer from the phosphorus to the oxygen atom and a dative π back-bond that produces an effective [P+]-[O−] configuration.
Phosphoryl chloride exists as neutral POCl3 molecules in the solid, liquid and gas states. This is unlike phosphorus pentachloride which exists as neutral PCl5 molecules in the gas and liquid states but adopts the ionic form [PCl4]+[PCl6]− (tetrachlorophosphonium hexachlorophosphate(V)) in the solid state. The average bond lengths in the crystal structure of POCl3 are 1.98 Å for P–Cl and 1.46 Å for P=O.
It has a critical pressure of 3.4 atm. With a freezing point of 1 °C and boiling point of 106 °C, the liquid range of POCl3 is rather similar to water. Also like water, POCl3 autoionizes, owing to the reversible formation of [POCl2]+ cations (dichlorooxophosphonium cations) and Cl− anions.
POCl3 reacts with water to give hydrogen chloride and phosphoric acid:
Intermediates in the conversion have been isolated, including pyrophosphoryl chloride, O(−P(=O)Cl2)2.
Upon treatment with excess alcohols and phenols, POCl3 gives phosphate esters:
Such reactions are often performed in the presence of an HCl acceptor such as pyridine or an amine.