Recent from talks
Knowledge base stats:
Talk channels stats:
Members stats:
Transition metal porphyrin complexes
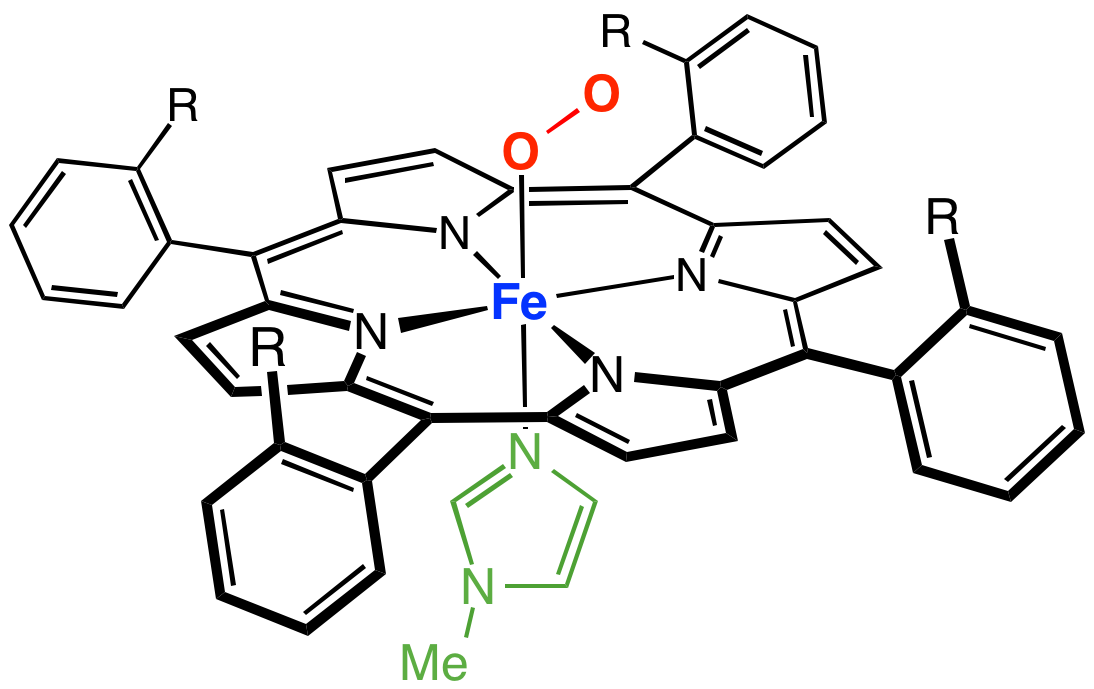
Transition metal porphyrin complexes are a family of coordination complexes of the conjugate base of porphyrins. Iron porphyrin complexes occur widely in nature, which has stimulated extensive studies on related synthetic complexes. The metal-porphyrin interaction is a strong one such that metalloporphyrins are thermally robust. They are catalysts and exhibit rich optical properties, although these complexes remain mainly of academic interest.
Porphyrin complexes consist of a square planar MN4 core. The periphery of the porphyrins, consisting of sp2-hybridized carbons, generally display only small deviations from planarity. Additionally, the metal is often not centered in the N4 plane.
Large metals such as zirconium, tantalum, and molybdenum tend to bind two porphyrin ligands. Some [M(OEP)]2 feature a multiple bonds between the metals.
Metal porphyrin complexes are almost always prepared by direct reaction of a metal halide with the free porphyrin, abbreviated here as H2P:
Two pyrrole protons are lost. The porphyrin dianion is an L2X2 ligand.
These syntheses require somewhat forcing conditions, consistent with the tight fit of the metal in the N42- "pocket." In nature, the insertion is mediated by chelatase enzymes. The insertion of a metal in synthetic porphyrins proceeds by the intermediacy of a "sitting atop complex" (SAC), whereby the entering metal interacts with only one or a two of the pyrrolic nitrogen centers.
In contrast to natural porphyrins, synthetic porphyrin ligands are typically symmetrical (i.e., their dianionic conjugate bases). Two major varieties are well studied, those with substituents at the meso positions, the premier example being tetraphenylporphyrin. These ligands are easy to prepare in one-pot procedures. A large number of aryl groups can be deployed aside from phenyl.
A second class of synthetic porphyrins have hydrogen at the meso positions. Octaethylporphyrin (H2OEP) is the subject of many such studies. It is more expensive than tetraphenylporphyrin.
Hub AI
Transition metal porphyrin complexes AI simulator
(@Transition metal porphyrin complexes_simulator)
Transition metal porphyrin complexes
Transition metal porphyrin complexes are a family of coordination complexes of the conjugate base of porphyrins. Iron porphyrin complexes occur widely in nature, which has stimulated extensive studies on related synthetic complexes. The metal-porphyrin interaction is a strong one such that metalloporphyrins are thermally robust. They are catalysts and exhibit rich optical properties, although these complexes remain mainly of academic interest.
Porphyrin complexes consist of a square planar MN4 core. The periphery of the porphyrins, consisting of sp2-hybridized carbons, generally display only small deviations from planarity. Additionally, the metal is often not centered in the N4 plane.
Large metals such as zirconium, tantalum, and molybdenum tend to bind two porphyrin ligands. Some [M(OEP)]2 feature a multiple bonds between the metals.
Metal porphyrin complexes are almost always prepared by direct reaction of a metal halide with the free porphyrin, abbreviated here as H2P:
Two pyrrole protons are lost. The porphyrin dianion is an L2X2 ligand.
These syntheses require somewhat forcing conditions, consistent with the tight fit of the metal in the N42- "pocket." In nature, the insertion is mediated by chelatase enzymes. The insertion of a metal in synthetic porphyrins proceeds by the intermediacy of a "sitting atop complex" (SAC), whereby the entering metal interacts with only one or a two of the pyrrolic nitrogen centers.
In contrast to natural porphyrins, synthetic porphyrin ligands are typically symmetrical (i.e., their dianionic conjugate bases). Two major varieties are well studied, those with substituents at the meso positions, the premier example being tetraphenylporphyrin. These ligands are easy to prepare in one-pot procedures. A large number of aryl groups can be deployed aside from phenyl.
A second class of synthetic porphyrins have hydrogen at the meso positions. Octaethylporphyrin (H2OEP) is the subject of many such studies. It is more expensive than tetraphenylporphyrin.