Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Primordial nuclide AI simulator
(@Primordial nuclide_simulator)
Hub AI
Primordial nuclide AI simulator
(@Primordial nuclide_simulator)
Primordial nuclide
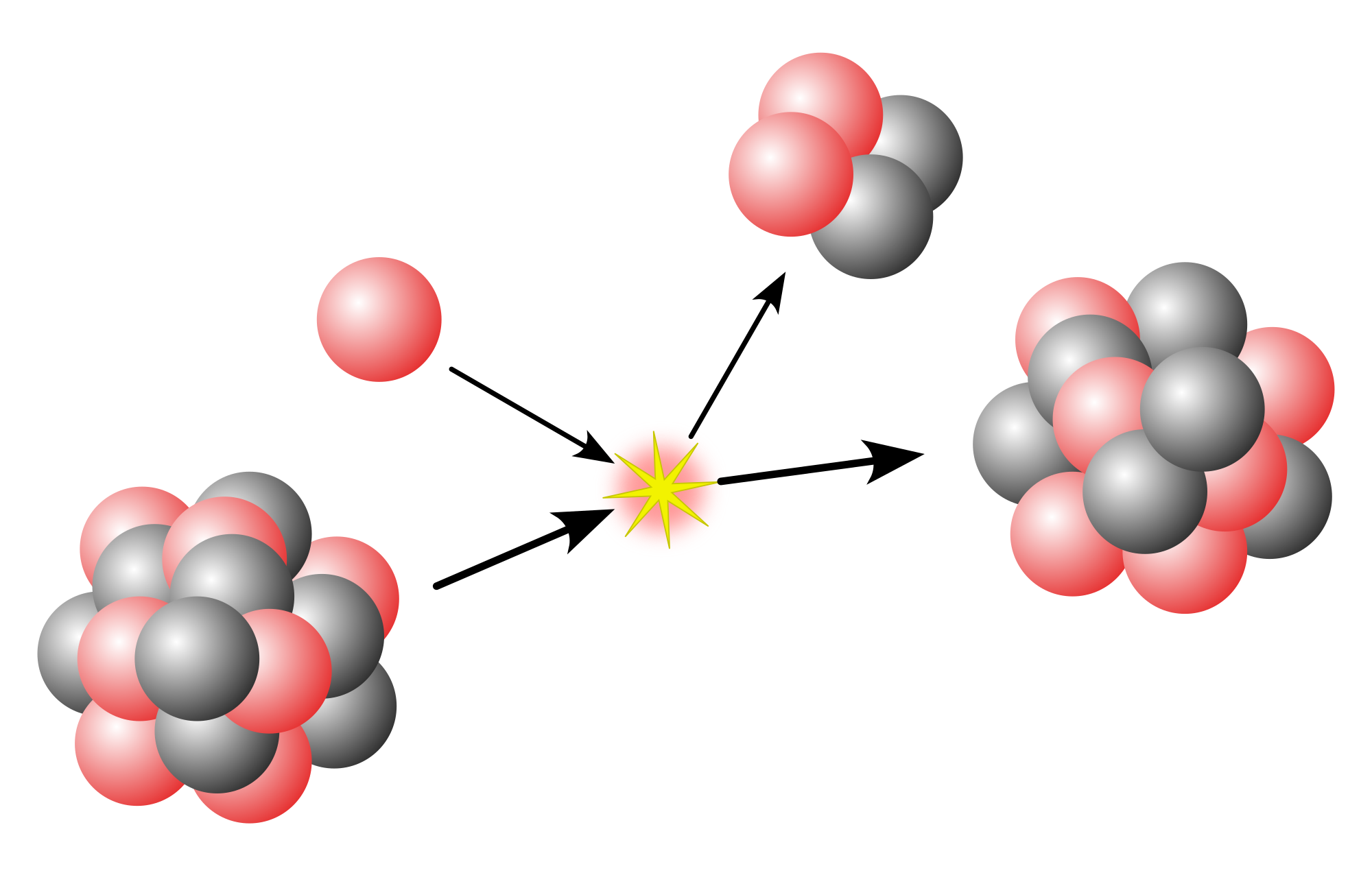
In geochemistry, geophysics and nuclear physics, primordial nuclides, or primordial isotopes, are nuclides found on Earth that have existed in their current form since before Earth was formed. Primordial nuclides were present in the interstellar medium from which the Solar System was formed, and were formed in the Big Bang, by nucleosynthesis in stars and supernovae followed by mass ejection, by cosmic ray spallation, or from other processes throughout the history of the universe. They are the stable nuclides plus the fraction of the long-lived radionuclides surviving from the primordial solar nebula through planet accretion until the present; 286 such nuclides are known.
All of the known 251 stable nuclides, plus another 35 nuclides that have half-lives long enough to have survived from the formation of the Earth, occur as primordial nuclides. These 35 primordial radionuclides represent isotopes of 28 separate elements.
Cadmium, tellurium, xenon, neodymium, samarium, osmium, and uranium each have two primordial radioisotopes (113
Cd, 116
Cd; 128
Te, 130
Te; 124
Xe, 136
Xe; 144
Nd, 150
Nd; 147
Sm, 148
Sm; 184
Os, 186
Os; and 235
U, 238
U).
Because the age of the Earth is 4.58×109 years (4.58 billion years), the half-life of the given nuclides must be greater than about 108 years (100 million years) for practical detectability. For example, for a nuclide with half-life 6×107 years (60 million years), this means 77 half-lives have elapsed, meaning that for each mole (6.02×1023 atoms) of that nuclide being present at the formation of Earth, only 4 atoms remain today.
The seven shortest-lived primordial nuclides (i.e., the nuclides with the shortest half-lives) to be detected as primordial are 87
Rb (4.92×1010 years), 187
Re (4.12×1010 years), 176
Lu (3.70×1010 years), 232
Th (1.40×1010 years), 238
U (4.46×109 years), 40
K (1.25×109 years), and 235
U (7.04×108 years).
These are the seven nuclides with half-lives comparable to, or somewhat less than, the estimated age of the universe. (87Rb, 187Re, 176Lu, and 232Th have half-lives somewhat longer than the age of the universe.) For a complete list of the 35 known primordial radionuclides, including the next 28 with half-lives much longer than the age of the universe, see the complete list below. For practical purposes, nuclides with half-lives much longer than the age of the universe may be treated as if they were stable. 87Rb, 187Re, 176Lu, 232Th, and 238U have half-lives long enough that their decay is limited over geological time scales; 40K and 235U have shorter half-lives and are hence severely depleted, but are still long-lived enough to remain present in significant amount on Earth.
The longest-lived isotope not proven to be primordial is 146
Sm, which has a half-life of 9.20×107 years, followed by 244
Pu (8.13×107 years) and 92
Nb (3.47×107 years). 244Pu was reported to exist in nature as a primordial nuclide in 1971, but this detection could not be confirmed by further studies in 2012 and 2022.
Taking into account that all these nuclides must exist for at least 4.58×109 years, 146Sm must survive 50 half-lives (and hence be reduced by 250 ≈ 1×1015), 244Pu must survive 57 (and be reduced by a factor of 257 ≈ 1×1017), and 92Nb must survive 130 (and be reduced by 2130 ≈ 1×1039). Mathematically, considering the likely initial abundances of these nuclides, primordial 146Sm and 244Pu should persist somewhere within the Earth today, even if they are not identifiable in the relatively minor portion of the Earth's crust available to human assays, while 92Nb and all shorter-lived nuclides should not. Nuclides such as 92Nb that were present in the primordial solar nebula but have long since decayed away completely are termed extinct radionuclides if they have no other means of being regenerated. As for 244Pu, calculations suggest that as of 2022, sensitivity limits were about one order of magnitude away from detecting it as a primordial nuclide.
Because primordial chemical elements often consist of more than one primordial isotope, there are only 83 distinct primordial chemical elements. Of these, 80 have at least one observationally stable isotope and three additional primordial elements have only radioactive isotopes (bismuth, thorium, and uranium).
Primordial nuclide
In geochemistry, geophysics and nuclear physics, primordial nuclides, or primordial isotopes, are nuclides found on Earth that have existed in their current form since before Earth was formed. Primordial nuclides were present in the interstellar medium from which the Solar System was formed, and were formed in the Big Bang, by nucleosynthesis in stars and supernovae followed by mass ejection, by cosmic ray spallation, or from other processes throughout the history of the universe. They are the stable nuclides plus the fraction of the long-lived radionuclides surviving from the primordial solar nebula through planet accretion until the present; 286 such nuclides are known.
All of the known 251 stable nuclides, plus another 35 nuclides that have half-lives long enough to have survived from the formation of the Earth, occur as primordial nuclides. These 35 primordial radionuclides represent isotopes of 28 separate elements.
Cadmium, tellurium, xenon, neodymium, samarium, osmium, and uranium each have two primordial radioisotopes (113
Cd, 116
Cd; 128
Te, 130
Te; 124
Xe, 136
Xe; 144
Nd, 150
Nd; 147
Sm, 148
Sm; 184
Os, 186
Os; and 235
U, 238
U).
Because the age of the Earth is 4.58×109 years (4.58 billion years), the half-life of the given nuclides must be greater than about 108 years (100 million years) for practical detectability. For example, for a nuclide with half-life 6×107 years (60 million years), this means 77 half-lives have elapsed, meaning that for each mole (6.02×1023 atoms) of that nuclide being present at the formation of Earth, only 4 atoms remain today.
The seven shortest-lived primordial nuclides (i.e., the nuclides with the shortest half-lives) to be detected as primordial are 87
Rb (4.92×1010 years), 187
Re (4.12×1010 years), 176
Lu (3.70×1010 years), 232
Th (1.40×1010 years), 238
U (4.46×109 years), 40
K (1.25×109 years), and 235
U (7.04×108 years).
These are the seven nuclides with half-lives comparable to, or somewhat less than, the estimated age of the universe. (87Rb, 187Re, 176Lu, and 232Th have half-lives somewhat longer than the age of the universe.) For a complete list of the 35 known primordial radionuclides, including the next 28 with half-lives much longer than the age of the universe, see the complete list below. For practical purposes, nuclides with half-lives much longer than the age of the universe may be treated as if they were stable. 87Rb, 187Re, 176Lu, 232Th, and 238U have half-lives long enough that their decay is limited over geological time scales; 40K and 235U have shorter half-lives and are hence severely depleted, but are still long-lived enough to remain present in significant amount on Earth.
The longest-lived isotope not proven to be primordial is 146
Sm, which has a half-life of 9.20×107 years, followed by 244
Pu (8.13×107 years) and 92
Nb (3.47×107 years). 244Pu was reported to exist in nature as a primordial nuclide in 1971, but this detection could not be confirmed by further studies in 2012 and 2022.
Taking into account that all these nuclides must exist for at least 4.58×109 years, 146Sm must survive 50 half-lives (and hence be reduced by 250 ≈ 1×1015), 244Pu must survive 57 (and be reduced by a factor of 257 ≈ 1×1017), and 92Nb must survive 130 (and be reduced by 2130 ≈ 1×1039). Mathematically, considering the likely initial abundances of these nuclides, primordial 146Sm and 244Pu should persist somewhere within the Earth today, even if they are not identifiable in the relatively minor portion of the Earth's crust available to human assays, while 92Nb and all shorter-lived nuclides should not. Nuclides such as 92Nb that were present in the primordial solar nebula but have long since decayed away completely are termed extinct radionuclides if they have no other means of being regenerated. As for 244Pu, calculations suggest that as of 2022, sensitivity limits were about one order of magnitude away from detecting it as a primordial nuclide.
Because primordial chemical elements often consist of more than one primordial isotope, there are only 83 distinct primordial chemical elements. Of these, 80 have at least one observationally stable isotope and three additional primordial elements have only radioactive isotopes (bismuth, thorium, and uranium).