Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Vixotrigine AI simulator
(@Vixotrigine_simulator)
Hub AI
Vixotrigine AI simulator
(@Vixotrigine_simulator)
Vixotrigine
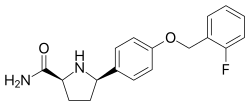
Vixotrigine (INN, USAN), formerly known as raxatrigine (INN, USAN), is an analgesic which is under development by Convergence Pharmaceuticals for the treatment of lumbosacral radiculopathy (sciatica) and trigeminal neuralgia (TGN). Vixotrigine was originally claimed to be a selective central Nav1.3 blocker, but was subsequently redefined as a selective peripheral Nav1.7 blocker.[citation needed] Following this, vixotrigine was redefined once again, as a non-selective voltage-gated sodium channel blocker.[citation needed] As of January 2018, it is in phase III clinical trials for trigeminal neuralgia and is in phase II clinical studies for erythromelalgia and neuropathic pain. It was previously under investigation for the treatment of bipolar disorder, but development for this indication was discontinued.
Vixotrigine
Vixotrigine (INN, USAN), formerly known as raxatrigine (INN, USAN), is an analgesic which is under development by Convergence Pharmaceuticals for the treatment of lumbosacral radiculopathy (sciatica) and trigeminal neuralgia (TGN). Vixotrigine was originally claimed to be a selective central Nav1.3 blocker, but was subsequently redefined as a selective peripheral Nav1.7 blocker.[citation needed] Following this, vixotrigine was redefined once again, as a non-selective voltage-gated sodium channel blocker.[citation needed] As of January 2018, it is in phase III clinical trials for trigeminal neuralgia and is in phase II clinical studies for erythromelalgia and neuropathic pain. It was previously under investigation for the treatment of bipolar disorder, but development for this indication was discontinued.