Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Pyrimidine dimer AI simulator
(@Pyrimidine dimer_simulator)
Hub AI
Pyrimidine dimer AI simulator
(@Pyrimidine dimer_simulator)
Pyrimidine dimer
Main article: Pyrimidine
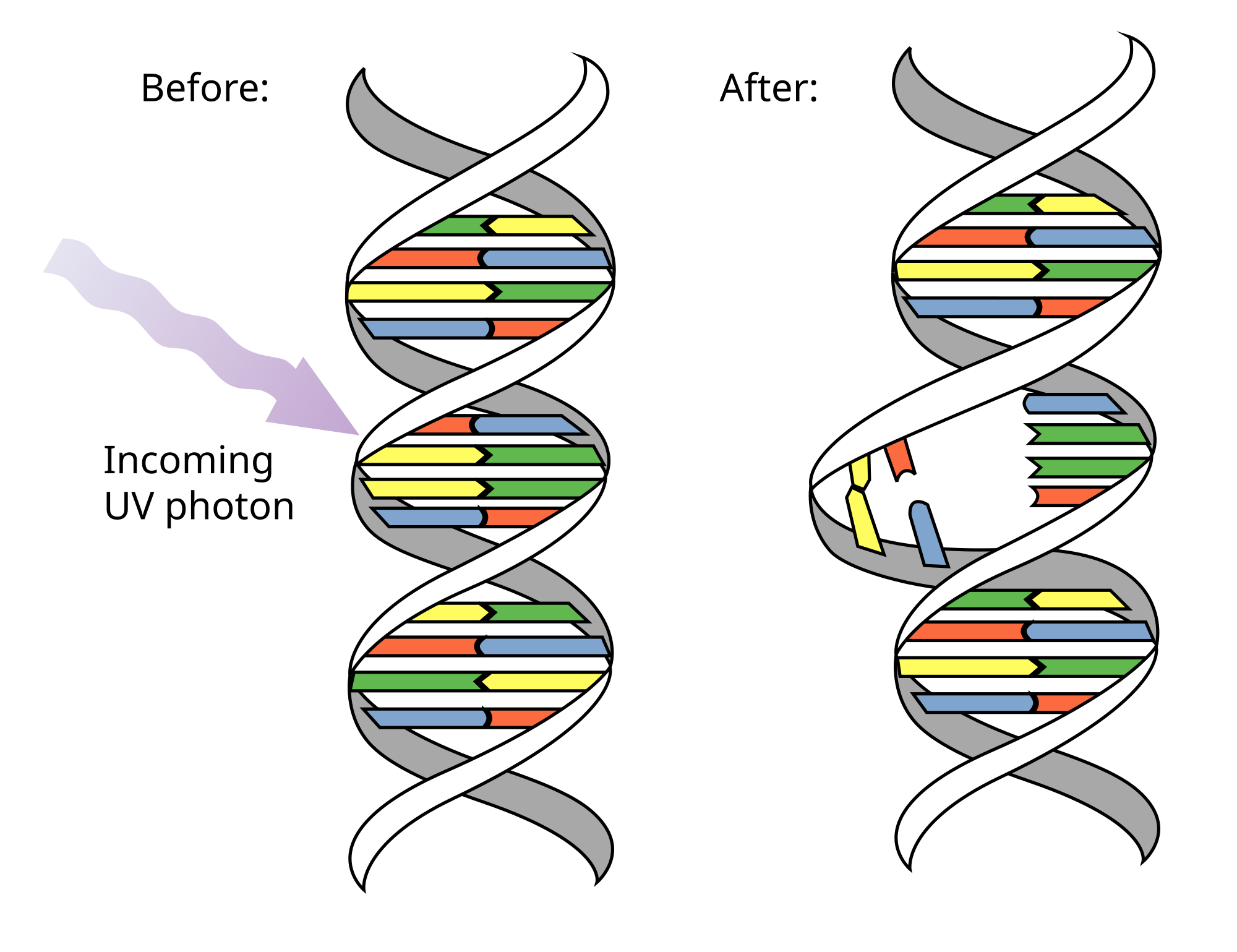
A pyrimidine dimer is a type of molecular lesion that arises when adjacent thymine or cytosine bases are bonded together in an atypical way, often as a result of a photochemical reaction. Ultraviolet light (UV), particularly UVC, often causes this direct DNA damage, causing the formation of covalent bonds near the nucleotides' carbon–carbon double bonds. The resulting photo-coupled dimers are fluorescent, and are commonly classified as cyclobutane pyrimidine dimers (CPDs) and 6–4 photoproducts. These pre-mutagenic lesions modify the DNA helix structure by distorting it.
Up to 100 dimerization reactions per second can occur in a skin cell exposed to sunlight, resulting in DNA damage. However, these lesions are typically rectified by DNA repair mechanisms before they can cause lasting damage. These mechanisms include photolyase reactivation and nucleotide excision repair, the latter of which is prevalent in humans. A failure to quickly repair these lesions may lead to erroneous (non-canonical) nucleotide incorporation by polymerase machinery. Extreme DNA damage can precipitate mutations within an organism's genome, potentially culminating in cancer cell formation. These lesions may also interfere with polymerase function, induce transcription or replication errors, or halt replication.
Pyrimidine dimers contribute to sunburn and melanin production, and are a primary factor in melanoma development in humans.
The different types of pyrimidine dimers each have distinct structures and implications for DNA integrity.
A cyclobutane dimer (CPD) features a four-membered ring formed by the fusion of two double-bonded carbons from adjacent pyrimidines. CPDs disrupt the formation of the base pair during DNA replication, which can potentially lead to mutations.
The 6–4 photoproduct (6–4 pyrimidine–pyrimidone, or 6–4 pyrimidine–pyrimidinone) is an alternate dimer configuration which covalently links the carbon at the 6 (C6) position of one pyrimidine ring and the carbon at the 4 (C4) position of the adjoining base's ring. This type of conversion occurs at one third of the frequency of CPDs but has a higher mutagenic risk.
A Dewar pyrimidinone results from the reversible isomerization of a 6–4 photoproduct under further light exposure.
Pyrimidine dimer
Main article: Pyrimidine
A pyrimidine dimer is a type of molecular lesion that arises when adjacent thymine or cytosine bases are bonded together in an atypical way, often as a result of a photochemical reaction. Ultraviolet light (UV), particularly UVC, often causes this direct DNA damage, causing the formation of covalent bonds near the nucleotides' carbon–carbon double bonds. The resulting photo-coupled dimers are fluorescent, and are commonly classified as cyclobutane pyrimidine dimers (CPDs) and 6–4 photoproducts. These pre-mutagenic lesions modify the DNA helix structure by distorting it.
Up to 100 dimerization reactions per second can occur in a skin cell exposed to sunlight, resulting in DNA damage. However, these lesions are typically rectified by DNA repair mechanisms before they can cause lasting damage. These mechanisms include photolyase reactivation and nucleotide excision repair, the latter of which is prevalent in humans. A failure to quickly repair these lesions may lead to erroneous (non-canonical) nucleotide incorporation by polymerase machinery. Extreme DNA damage can precipitate mutations within an organism's genome, potentially culminating in cancer cell formation. These lesions may also interfere with polymerase function, induce transcription or replication errors, or halt replication.
Pyrimidine dimers contribute to sunburn and melanin production, and are a primary factor in melanoma development in humans.
The different types of pyrimidine dimers each have distinct structures and implications for DNA integrity.
A cyclobutane dimer (CPD) features a four-membered ring formed by the fusion of two double-bonded carbons from adjacent pyrimidines. CPDs disrupt the formation of the base pair during DNA replication, which can potentially lead to mutations.
The 6–4 photoproduct (6–4 pyrimidine–pyrimidone, or 6–4 pyrimidine–pyrimidinone) is an alternate dimer configuration which covalently links the carbon at the 6 (C6) position of one pyrimidine ring and the carbon at the 4 (C4) position of the adjoining base's ring. This type of conversion occurs at one third of the frequency of CPDs but has a higher mutagenic risk.
A Dewar pyrimidinone results from the reversible isomerization of a 6–4 photoproduct under further light exposure.