Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Abiraterone acetate AI simulator
(@Abiraterone acetate_simulator)
Hub AI
Abiraterone acetate AI simulator
(@Abiraterone acetate_simulator)
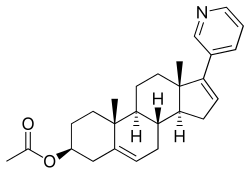
Abiraterone acetate
Abiraterone acetate, sold under the brand name Zytiga among others, is a medication used to treat prostate cancer. Specifically it is used together with a corticosteroid for metastatic castration-resistant prostate cancer (mCRPC) and metastatic high-risk castration-sensitive prostate cancer (mCSPC). It should either be used following removal of the testicles or along with a gonadotropin-releasing hormone (GnRH) analog. It is taken by mouth.
Common side effects include tiredness, vomiting, headache, joint pain, high blood pressure, swelling, low blood potassium, high blood sugar, hot flashes, diarrhea, and cough. Other severe side effects may include liver failure and adrenocortical insufficiency. In males whose partners can become pregnant, birth control is recommended. Supplied as abiraterone acetate it is converted in the body to abiraterone. Abiraterone acetate works by suppressing the production of androgens – specifically it inhibits CYP17A1 – and thereby decreases the production of testosterone. In doing so, it prevents the effects of these hormones in prostate cancer.
Abiraterone acetate was described in 1995, and approved for medical use in the United States and the European Union in 2011. It is on the World Health Organization's List of Essential Medicines. It is available as a generic medication.
Abiraterone acetate is used in combination with prednisone, a corticosteroid, as a treatment for mCRPC (previously called hormone-resistant or hormone-refractory prostate cancer). This is a form of prostate cancer that is not responding to first-line androgen deprivation therapy or treatment with androgen receptor antagonists. Abiraterone acetate has received Food and Drug Administration (FDA) (28 April 2011), European Medicines Agency (EMA) (23 September 2011), Medicines and Healthcare products Regulatory Agency (MHRA) (5 September 2011) and Therapeutic Goods Administration (TGA) (1 March 2012) approval for this indication. In Australia it is covered by the Pharmaceutical Benefits Scheme when being used to treat castration-resistant prostate cancer and given in combination with prednisone/prednisolone (subject to the conditions that the patient is not currently receiving chemotherapy, is either resistant or intolerant of docetaxel, has a WHO performance status of <2, and his disease has not since become progressive since treatment with PBS-subsidised abiraterone acetate has commenced).
Abiraterone acetate/methylprednisolone, sold under the brand name Yonsa Mpred, is a composite package that contains both abiraterone acetate (Yonsa) and methylprednisolone. It was approved for medical use in Australia in March 2022.
Contraindications include hypersensitivity to abiraterone acetate. Although documents state that it should not be taken by women who are or who may become pregnant, there is no medical reason that any woman should take it. Women who are pregnant should not even touch the pills unless they are wearing gloves. Other cautions include severe baseline hepatic impairment, mineralocorticoid excess, cardiovascular disease including heart failure and hypertension, uncorrected hypokalemia, and adrenocorticoid insufficiency.
Side effects by frequency:
Very common (>10% frequency):
Abiraterone acetate
Abiraterone acetate, sold under the brand name Zytiga among others, is a medication used to treat prostate cancer. Specifically it is used together with a corticosteroid for metastatic castration-resistant prostate cancer (mCRPC) and metastatic high-risk castration-sensitive prostate cancer (mCSPC). It should either be used following removal of the testicles or along with a gonadotropin-releasing hormone (GnRH) analog. It is taken by mouth.
Common side effects include tiredness, vomiting, headache, joint pain, high blood pressure, swelling, low blood potassium, high blood sugar, hot flashes, diarrhea, and cough. Other severe side effects may include liver failure and adrenocortical insufficiency. In males whose partners can become pregnant, birth control is recommended. Supplied as abiraterone acetate it is converted in the body to abiraterone. Abiraterone acetate works by suppressing the production of androgens – specifically it inhibits CYP17A1 – and thereby decreases the production of testosterone. In doing so, it prevents the effects of these hormones in prostate cancer.
Abiraterone acetate was described in 1995, and approved for medical use in the United States and the European Union in 2011. It is on the World Health Organization's List of Essential Medicines. It is available as a generic medication.
Abiraterone acetate is used in combination with prednisone, a corticosteroid, as a treatment for mCRPC (previously called hormone-resistant or hormone-refractory prostate cancer). This is a form of prostate cancer that is not responding to first-line androgen deprivation therapy or treatment with androgen receptor antagonists. Abiraterone acetate has received Food and Drug Administration (FDA) (28 April 2011), European Medicines Agency (EMA) (23 September 2011), Medicines and Healthcare products Regulatory Agency (MHRA) (5 September 2011) and Therapeutic Goods Administration (TGA) (1 March 2012) approval for this indication. In Australia it is covered by the Pharmaceutical Benefits Scheme when being used to treat castration-resistant prostate cancer and given in combination with prednisone/prednisolone (subject to the conditions that the patient is not currently receiving chemotherapy, is either resistant or intolerant of docetaxel, has a WHO performance status of <2, and his disease has not since become progressive since treatment with PBS-subsidised abiraterone acetate has commenced).
Abiraterone acetate/methylprednisolone, sold under the brand name Yonsa Mpred, is a composite package that contains both abiraterone acetate (Yonsa) and methylprednisolone. It was approved for medical use in Australia in March 2022.
Contraindications include hypersensitivity to abiraterone acetate. Although documents state that it should not be taken by women who are or who may become pregnant, there is no medical reason that any woman should take it. Women who are pregnant should not even touch the pills unless they are wearing gloves. Other cautions include severe baseline hepatic impairment, mineralocorticoid excess, cardiovascular disease including heart failure and hypertension, uncorrected hypokalemia, and adrenocorticoid insufficiency.
Side effects by frequency:
Very common (>10% frequency):