Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
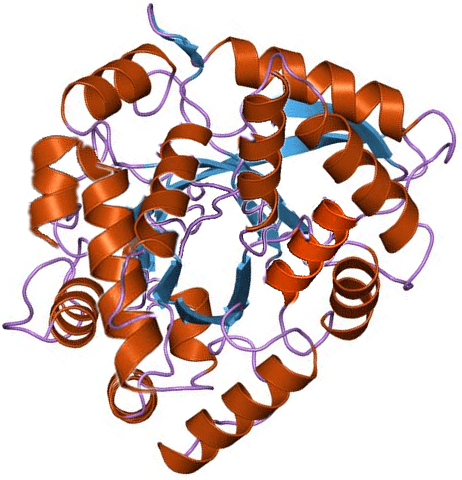
Gliadin
Gliadin (a type of prolamin) is a class of proteins present in wheat and several other cereals within the grass genera Triticum and Hordeum. Gliadins, which are a component of gluten, are essential for giving bread the ability to rise properly during baking. Gliadins and glutenins are the two main components of the gluten fraction of the wheat seed. This gluten is found in products such as wheat flour. Gluten is split about evenly between the gliadins and glutenins, although there are variations found in different sources.
Neither gliadins nor glutenins are water-soluble, but gliadins are soluble in 70% aqueous ethanol. There are three main types of gliadin (α, γ, and ω), to which the body is intolerant in coeliac (or celiac) disease. Diagnosis of this disease has recently been improving.
Gliadin can cross the intestinal epithelium. Breast milk of healthy human mothers who eat gluten-containing foods presents high levels of non-degraded gliadin.
The α, γ, and ω gliadin types are separated and distinguished based on their amino acid sequences in the N-terminal cysteine domain.
The gliadins are intrinsically disordered proteins meaning that they have continuously altering shapes making it difficult to study them. The performed image analysis and computer simulations of the proteins show that the average shape of the gliadins follows an elliptical shape. More specifically the protein likely has a tadpole-like structure with a hydrophobic core and a loose disordered tail. Compared to the other gluten proteins like the glutenins, which form extended networks of polymers due to disulphide bonds, gliadins are monomeric molecules in the cell, even if they in many ways are very similar. Especially the low molecular weight glutenins are similar in the way that they have cysteines located in matching locations as many of the gliadins. However, the gliadins are unable to form polymers in the cell since its cysteines form intra-chain disulphide bonds at synthesis due to hydrophobic interactions.
Gliadins are capable to aggregate into larger oligomers and interact with other gluten proteins, due to large hydrophobic sections, poly-Q and repetitive sequences. These sections are likely to aggregate hydrophobically, liquid-liquid phase separate, potentially form β-sheets aggregates or simply entangles by its structural properties.
Gliadins are prolamins and are separated on the basis of electrophoretic mobility and isoelectric focusing. Gliadin peptides cross the intestinal barrier by active transport. [citation needed]
Gliadins are known for their role, along with glutenin, in the formation of gluten. They are slightly soluble in ethanol and contain only intramolecular disulfide links. They also cause some of the best examples of food-derived pathogenesis. People with celiac disease (also known as gluten-sensitive enteropathy) are sensitive to α, β, and γ gliadins. Those with wheat-dependent urticaria and baker's asthma are sensitive to ω-gliadins.[citation needed]
Gliadin
Gliadin (a type of prolamin) is a class of proteins present in wheat and several other cereals within the grass genera Triticum and Hordeum. Gliadins, which are a component of gluten, are essential for giving bread the ability to rise properly during baking. Gliadins and glutenins are the two main components of the gluten fraction of the wheat seed. This gluten is found in products such as wheat flour. Gluten is split about evenly between the gliadins and glutenins, although there are variations found in different sources.
Neither gliadins nor glutenins are water-soluble, but gliadins are soluble in 70% aqueous ethanol. There are three main types of gliadin (α, γ, and ω), to which the body is intolerant in coeliac (or celiac) disease. Diagnosis of this disease has recently been improving.
Gliadin can cross the intestinal epithelium. Breast milk of healthy human mothers who eat gluten-containing foods presents high levels of non-degraded gliadin.
The α, γ, and ω gliadin types are separated and distinguished based on their amino acid sequences in the N-terminal cysteine domain.
The gliadins are intrinsically disordered proteins meaning that they have continuously altering shapes making it difficult to study them. The performed image analysis and computer simulations of the proteins show that the average shape of the gliadins follows an elliptical shape. More specifically the protein likely has a tadpole-like structure with a hydrophobic core and a loose disordered tail. Compared to the other gluten proteins like the glutenins, which form extended networks of polymers due to disulphide bonds, gliadins are monomeric molecules in the cell, even if they in many ways are very similar. Especially the low molecular weight glutenins are similar in the way that they have cysteines located in matching locations as many of the gliadins. However, the gliadins are unable to form polymers in the cell since its cysteines form intra-chain disulphide bonds at synthesis due to hydrophobic interactions.
Gliadins are capable to aggregate into larger oligomers and interact with other gluten proteins, due to large hydrophobic sections, poly-Q and repetitive sequences. These sections are likely to aggregate hydrophobically, liquid-liquid phase separate, potentially form β-sheets aggregates or simply entangles by its structural properties.
Gliadins are prolamins and are separated on the basis of electrophoretic mobility and isoelectric focusing. Gliadin peptides cross the intestinal barrier by active transport. [citation needed]
Gliadins are known for their role, along with glutenin, in the formation of gluten. They are slightly soluble in ethanol and contain only intramolecular disulfide links. They also cause some of the best examples of food-derived pathogenesis. People with celiac disease (also known as gluten-sensitive enteropathy) are sensitive to α, β, and γ gliadins. Those with wheat-dependent urticaria and baker's asthma are sensitive to ω-gliadins.[citation needed]