Recent from talks
Knowledge base stats:
Talk channels stats:
Members stats:
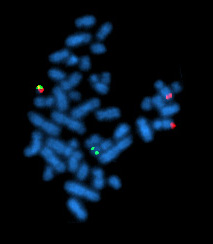
Philadelphia chromosome
The Philadelphia chromosome or Philadelphia translocation (Ph) is an abnormal version of chromosome 22 where a part of the Abelson murine leukemia 1 (ABL1) gene on chromosome 9 breaks off and attaches to the breakpoint cluster region (BCR) gene in chromosome 22. The balanced reciprocal translocation between the long arms of 9 and 22 chromosomes [t (9; 22) (q34; q11)] results in the fusion gene BCR::ABL1. The oncogenic protein with persistently enhanced tyrosine kinase (TK) activity transcribed by the BCR::ABL1 fusion gene can lead to rapid, uncontrolled growth of immature white blood cells that accumulates in the blood and bone marrow.
The Philadelphia chromosome is present in the bone marrow cells of a vast majority chronic myelogenous leukemia (CML) patients. The expression patterns of different BCR-ABL1 transcripts vary during the progression of CML. Each variant is present in a distinct leukemia phenotype and can be used to predict response to therapy and clinical outcomes. The Ph is also observed in patients with acute lymphocytic leukemia (ALL), acute myelogenous leukemia (AML), and mixed-phenotype acute leukemia.
The chromosomal abnormality in the Philadelphia chromosome from the reciprocal translocation t(9;22)(q34;q11), is the result of fragments from chromosomes 9 and 22 swapping places. The ABL proto-oncogene 1 on chromosome 9, from region q34, is juxtaposed with a portion of the BCR gene on chromosome 22, region q11.2. The derivative chromosome 22 produced by this translocation is known as the Philadelphia chromosome. This translocation creates a fusion gene, BCR::ABL1, which codes for a constitutively active ("always on") tyrosine kinase signaling protein, driving uncontrolled cell division.
The formation of the Philadelphia chromosome is due to the fusion of the BCR and ABL1 genes. ABL1 is derived from Abelson murine leukemia, a retrovirus that causes leukemia and lymphoma in mice. It is named after Herbert T. Abelson, who discovered it in 1970. BCR stands for breakpoint cluster region because of the relatively small genomic region where the DNA breaks occurs. The fusion can happen at different points in the BCR gene, where the gene will fuse with exon 2 of ABL (breakpoints in exon 3 of ABL1 have also been observed, but are less frequent). The BCR::ABL1 oncogene exists in three primary isoforms depending on the breakpoint site of the BCR gene and are named after the fuse region, and the molecular weight of the transcribed BCR-ABL1 fusion protein, and all encode for a tyrosine kinase protein. The e1a2 transcript is a fusion between exon 1 of BCR, also called the minor breakpoint region (m-BCR), and exon 2 of ABL1 and encodes an oncoprotein of 185-190 kDa, referred to as P190. BCR::ABL1 is associated with around 20-30% of all Philadelphia chromosome positive B-cell ALL (ph+ B-ALL) and is the most genetic subgroup of B-ALL. The incident rate for ALL is age related, as the incident rate increases to 50% for ALL in patients aged 50 years and older. where P190 is associated with 60-80% of these. The e13a2 and e14a2 transcripts found in the major breakpoint region (M-BCR), which consists of exons 12 through 16. These transcripts encode for a oncoprotein of size 210kDa, and is referred to as P210. P210 is associated with over 95% of CML cases, with a 50/50 split between the e13a2 and e14a2 variants. Additionally, e13a2 and e14a2 has been found to be co-expressed in an estimated 5-10% of CML patients. P210 is also found to be present in 40% of adult and 10% of child B-ALL cases. CML has an incidence of 50 cases per million per year Lastly, the e19a2 transcript, located in the μ-BCR region, produces an oncoprotein of 230kDa which is referred to as P230. This variant is uncommon in comparison, and has been linked to the rare disease chronic neutrophilic leukemia (CNL), which falls under mixed-phenotype acute leukemias.
Detection of these variants is carried out using methods such as sanger sequencing, reverse transcription polymerase chain reaction (RT-PCR), qPCR, Flourescense In Situ Hybridization (FISH), and southern blotting. However, many laboratories are working on incorporating Next Generation Sequencing (NGS) into routine diagnostic analysis as NGS technology is rapidly improving, and will in the near future enable full clinical sequencing of the entire gene.
The normal BCR gene is ubiquitously expressed cytoplasmic protein with many known functionalities. ABL1 gene expresses a membrane-associated protein, a nonreceptor protein-tyrosine kinase. ABL1 is linked to multiple processes related to cell growth and survival, such as cytoskeleton and actin remodelling, and inhibition of cell cycle progression. ABL1 can also be found translocated in the nucleus and has DNA binding capabilities, as it is involved in DNA damage control and repair, and apoptosis. The BCR-ABL1 transcript is also translated into a tyrosine kinase containing domains from both the BCR and ABL1 genes. The activity of tyrosine kinases is typically regulated in an auto-inhibitory fashion, but the BCR-ABL1 fusion gene codes for a protein that is constitutively activated, leading to impaired DNA binding and unregulated cell division (i.e. cancer).
The formation of the BCR::ABL1 oncogene leads to a constitutively active Tyrosine kinase, which is important for transformation of hematopoietic cells. Kinases are enzymes that add phosphate groups to their substrates. In cell biology and cell signalling, phosphorylated substrates are mainly used as a "on" signal, usually setting in motion a cascade of downstream signalling pathways. The high activity of TK leads to a chronic activation of signalling pathways associated with all stages of cell transformation. Resulting in uncontrolled cell proliferation, blocked cell differentiation, and inhibited apoptosis. Meaning cells with the BCR::ABL1 fusion multiply uncontrollably, without differentiating into mature white blood cells that live longer due to a lack of apoptosis promoting signals. This leads to a buildup of these immature white blood cells in the bloodstream There are several signalling pathways associated with the BCR::ABL1 pathogenesis, such as: the Mitogen-activated protein kinase (MAPK/RAS) pathway, PI3K-AKT-mTOR (PAM) pathway, Janus kinase (JAK) - Signal transducers and activators of transcription (STAT) pathway and the Protein Phosphatase 2A (PP2A) tumour suppressor gene –β-catenin pathway.
The MAPK pathway includes several key signalling components, and phosphorylation events known to play a crucial part in carcinogenesis. MAPK is composed of multiple signalling cascades, of which the RAS-RAF-MEK-ERK signalling pathway can be found. This pathway is known to play a vital role in cell development, proliferation and survival. Mutations in, and abnormal activation of this pathway will induce tumours, being present in 30% of all cancers. BCR::ABL1 fusions will create constitutively active tyrosine kinases. BCR::ABL1 contains a kinase domain containing Tyr 177, which is a binding site for growth factor receptor binding protein 2 (GRB2). GRB2 binds to a protein called son of sevenless (SOS), a guanine nucleotide exchange factor (GEF). SOS facilitates the conversion of inactive RAS-GDP to active RAS-GTP, which turns on the cascade of enzymes which the RAS-RAF-MEK-ERK pathway is composed of. All steps in this pathway are phosphorylation of enzyme downstream of the signalling pathway, which ends with ERK which in turn phosphorylates hundreds of substrates in the cytoplasm and nucleus which regulate many cellular processes including proliferation, survival, and growth. The RAS/RAF/MEK/ERK pathway is also implicated in overexpression of osteopontin (OPN), which is important for maintenance of the hematopoietic stem cell niche, which indirectly influences unchecked proliferation characteristic of leukemic cells. The RAS-MAPK pathway is associated with many types of cancers, including CML and ph+ ALL, being also linked to imatinib resistance in some cases.
Hub AI
Philadelphia chromosome AI simulator
(@Philadelphia chromosome_simulator)
Philadelphia chromosome
The Philadelphia chromosome or Philadelphia translocation (Ph) is an abnormal version of chromosome 22 where a part of the Abelson murine leukemia 1 (ABL1) gene on chromosome 9 breaks off and attaches to the breakpoint cluster region (BCR) gene in chromosome 22. The balanced reciprocal translocation between the long arms of 9 and 22 chromosomes [t (9; 22) (q34; q11)] results in the fusion gene BCR::ABL1. The oncogenic protein with persistently enhanced tyrosine kinase (TK) activity transcribed by the BCR::ABL1 fusion gene can lead to rapid, uncontrolled growth of immature white blood cells that accumulates in the blood and bone marrow.
The Philadelphia chromosome is present in the bone marrow cells of a vast majority chronic myelogenous leukemia (CML) patients. The expression patterns of different BCR-ABL1 transcripts vary during the progression of CML. Each variant is present in a distinct leukemia phenotype and can be used to predict response to therapy and clinical outcomes. The Ph is also observed in patients with acute lymphocytic leukemia (ALL), acute myelogenous leukemia (AML), and mixed-phenotype acute leukemia.
The chromosomal abnormality in the Philadelphia chromosome from the reciprocal translocation t(9;22)(q34;q11), is the result of fragments from chromosomes 9 and 22 swapping places. The ABL proto-oncogene 1 on chromosome 9, from region q34, is juxtaposed with a portion of the BCR gene on chromosome 22, region q11.2. The derivative chromosome 22 produced by this translocation is known as the Philadelphia chromosome. This translocation creates a fusion gene, BCR::ABL1, which codes for a constitutively active ("always on") tyrosine kinase signaling protein, driving uncontrolled cell division.
The formation of the Philadelphia chromosome is due to the fusion of the BCR and ABL1 genes. ABL1 is derived from Abelson murine leukemia, a retrovirus that causes leukemia and lymphoma in mice. It is named after Herbert T. Abelson, who discovered it in 1970. BCR stands for breakpoint cluster region because of the relatively small genomic region where the DNA breaks occurs. The fusion can happen at different points in the BCR gene, where the gene will fuse with exon 2 of ABL (breakpoints in exon 3 of ABL1 have also been observed, but are less frequent). The BCR::ABL1 oncogene exists in three primary isoforms depending on the breakpoint site of the BCR gene and are named after the fuse region, and the molecular weight of the transcribed BCR-ABL1 fusion protein, and all encode for a tyrosine kinase protein. The e1a2 transcript is a fusion between exon 1 of BCR, also called the minor breakpoint region (m-BCR), and exon 2 of ABL1 and encodes an oncoprotein of 185-190 kDa, referred to as P190. BCR::ABL1 is associated with around 20-30% of all Philadelphia chromosome positive B-cell ALL (ph+ B-ALL) and is the most genetic subgroup of B-ALL. The incident rate for ALL is age related, as the incident rate increases to 50% for ALL in patients aged 50 years and older. where P190 is associated with 60-80% of these. The e13a2 and e14a2 transcripts found in the major breakpoint region (M-BCR), which consists of exons 12 through 16. These transcripts encode for a oncoprotein of size 210kDa, and is referred to as P210. P210 is associated with over 95% of CML cases, with a 50/50 split between the e13a2 and e14a2 variants. Additionally, e13a2 and e14a2 has been found to be co-expressed in an estimated 5-10% of CML patients. P210 is also found to be present in 40% of adult and 10% of child B-ALL cases. CML has an incidence of 50 cases per million per year Lastly, the e19a2 transcript, located in the μ-BCR region, produces an oncoprotein of 230kDa which is referred to as P230. This variant is uncommon in comparison, and has been linked to the rare disease chronic neutrophilic leukemia (CNL), which falls under mixed-phenotype acute leukemias.
Detection of these variants is carried out using methods such as sanger sequencing, reverse transcription polymerase chain reaction (RT-PCR), qPCR, Flourescense In Situ Hybridization (FISH), and southern blotting. However, many laboratories are working on incorporating Next Generation Sequencing (NGS) into routine diagnostic analysis as NGS technology is rapidly improving, and will in the near future enable full clinical sequencing of the entire gene.
The normal BCR gene is ubiquitously expressed cytoplasmic protein with many known functionalities. ABL1 gene expresses a membrane-associated protein, a nonreceptor protein-tyrosine kinase. ABL1 is linked to multiple processes related to cell growth and survival, such as cytoskeleton and actin remodelling, and inhibition of cell cycle progression. ABL1 can also be found translocated in the nucleus and has DNA binding capabilities, as it is involved in DNA damage control and repair, and apoptosis. The BCR-ABL1 transcript is also translated into a tyrosine kinase containing domains from both the BCR and ABL1 genes. The activity of tyrosine kinases is typically regulated in an auto-inhibitory fashion, but the BCR-ABL1 fusion gene codes for a protein that is constitutively activated, leading to impaired DNA binding and unregulated cell division (i.e. cancer).
The formation of the BCR::ABL1 oncogene leads to a constitutively active Tyrosine kinase, which is important for transformation of hematopoietic cells. Kinases are enzymes that add phosphate groups to their substrates. In cell biology and cell signalling, phosphorylated substrates are mainly used as a "on" signal, usually setting in motion a cascade of downstream signalling pathways. The high activity of TK leads to a chronic activation of signalling pathways associated with all stages of cell transformation. Resulting in uncontrolled cell proliferation, blocked cell differentiation, and inhibited apoptosis. Meaning cells with the BCR::ABL1 fusion multiply uncontrollably, without differentiating into mature white blood cells that live longer due to a lack of apoptosis promoting signals. This leads to a buildup of these immature white blood cells in the bloodstream There are several signalling pathways associated with the BCR::ABL1 pathogenesis, such as: the Mitogen-activated protein kinase (MAPK/RAS) pathway, PI3K-AKT-mTOR (PAM) pathway, Janus kinase (JAK) - Signal transducers and activators of transcription (STAT) pathway and the Protein Phosphatase 2A (PP2A) tumour suppressor gene –β-catenin pathway.
The MAPK pathway includes several key signalling components, and phosphorylation events known to play a crucial part in carcinogenesis. MAPK is composed of multiple signalling cascades, of which the RAS-RAF-MEK-ERK signalling pathway can be found. This pathway is known to play a vital role in cell development, proliferation and survival. Mutations in, and abnormal activation of this pathway will induce tumours, being present in 30% of all cancers. BCR::ABL1 fusions will create constitutively active tyrosine kinases. BCR::ABL1 contains a kinase domain containing Tyr 177, which is a binding site for growth factor receptor binding protein 2 (GRB2). GRB2 binds to a protein called son of sevenless (SOS), a guanine nucleotide exchange factor (GEF). SOS facilitates the conversion of inactive RAS-GDP to active RAS-GTP, which turns on the cascade of enzymes which the RAS-RAF-MEK-ERK pathway is composed of. All steps in this pathway are phosphorylation of enzyme downstream of the signalling pathway, which ends with ERK which in turn phosphorylates hundreds of substrates in the cytoplasm and nucleus which regulate many cellular processes including proliferation, survival, and growth. The RAS/RAF/MEK/ERK pathway is also implicated in overexpression of osteopontin (OPN), which is important for maintenance of the hematopoietic stem cell niche, which indirectly influences unchecked proliferation characteristic of leukemic cells. The RAS-MAPK pathway is associated with many types of cancers, including CML and ph+ ALL, being also linked to imatinib resistance in some cases.