Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
COVIran Barekat AI simulator
(@COVIran Barekat_simulator)
Hub AI
COVIran Barekat AI simulator
(@COVIran Barekat_simulator)
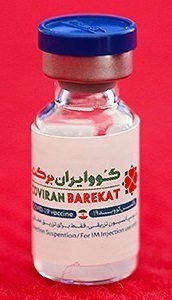
COVIran Barekat
COVIran Barekat (Persian: کووایران برکت) is a COVID-19 vaccine developed in Iran by Shifa Pharmed Industrial Group, a subsidiary of the Barkat Pharmaceutical Group. It is an inactivated virus-based vaccine. Iranian authorities have authorized its emergency use. This makes it the first locally developed COVID-19 vaccine to be approved for emergency use in the Middle East.
Officials in charge say they are in the process to publish the results of the clinical trials in a peer-reviewed journal. The interim results of the phases 1 and 2 trials showed 93.5% (95% CI, 88.4–99.6%) of the receivers of the vaccine have produced neutralizing antibodies against SARS-CoV-2. Those results have not been peer-reviewed and describe the immunogenicity of the vaccine and not its efficacy. On 3 March 2022, peer-reviewed results have been published in the Clinical Microbiology and Infection. [further explanation needed]
As of February 27, 2022, approximately 60 million doses have been produced according to Shifa Pharmed's CEO. and the annual production capacity of this vaccine has reached 350 million doses. Multiple Iranian personalities have received the vaccine, including the Supreme Leader, Ali Khamenei and the President, Ebrahim Raisi.
A version of the vaccine based on the Omicron variant and named CovIran Barkat Plus is in development and studied to be given as a third dose.
It is given by intramuscular injection and requires two doses given 28 days apart.
COVIran Barekat is an inactivated virus-based vaccine. In other words, "it is made of a coronavirus that has been weakened or killed by chemicals, similar to how polio immunizations are made".
The Execution of Imam Khomeini's Order (EIKO) and Barkat Pharmaceutical Group is parent company of Shifa Pharmed Industrial Group. It's reported to be "state-affiliated". Shifa Pharmed products include seven drugs and three biologics besides their COVID-19 vaccine which is their first vaccine to be produced. Around 650 people worked in three shifts, around the clock, to develop the vaccine.
Dr. Minoo Mohraz has been selected as the lead of the "Corona vaccine project in Iran". Mohraz is an Iranian physician, scientist, and AIDS specialist. She is a full professor (emeritus) of infectious diseases at Tehran University of Medical Sciences and head of the Iranian Centre for HIV/AIDS. Mohraz has also served, within the World Health Organization, as an expert on HIV/AIDS in Iran and the Eastern Mediterranean.
COVIran Barekat
COVIran Barekat (Persian: کووایران برکت) is a COVID-19 vaccine developed in Iran by Shifa Pharmed Industrial Group, a subsidiary of the Barkat Pharmaceutical Group. It is an inactivated virus-based vaccine. Iranian authorities have authorized its emergency use. This makes it the first locally developed COVID-19 vaccine to be approved for emergency use in the Middle East.
Officials in charge say they are in the process to publish the results of the clinical trials in a peer-reviewed journal. The interim results of the phases 1 and 2 trials showed 93.5% (95% CI, 88.4–99.6%) of the receivers of the vaccine have produced neutralizing antibodies against SARS-CoV-2. Those results have not been peer-reviewed and describe the immunogenicity of the vaccine and not its efficacy. On 3 March 2022, peer-reviewed results have been published in the Clinical Microbiology and Infection. [further explanation needed]
As of February 27, 2022, approximately 60 million doses have been produced according to Shifa Pharmed's CEO. and the annual production capacity of this vaccine has reached 350 million doses. Multiple Iranian personalities have received the vaccine, including the Supreme Leader, Ali Khamenei and the President, Ebrahim Raisi.
A version of the vaccine based on the Omicron variant and named CovIran Barkat Plus is in development and studied to be given as a third dose.
It is given by intramuscular injection and requires two doses given 28 days apart.
COVIran Barekat is an inactivated virus-based vaccine. In other words, "it is made of a coronavirus that has been weakened or killed by chemicals, similar to how polio immunizations are made".
The Execution of Imam Khomeini's Order (EIKO) and Barkat Pharmaceutical Group is parent company of Shifa Pharmed Industrial Group. It's reported to be "state-affiliated". Shifa Pharmed products include seven drugs and three biologics besides their COVID-19 vaccine which is their first vaccine to be produced. Around 650 people worked in three shifts, around the clock, to develop the vaccine.
Dr. Minoo Mohraz has been selected as the lead of the "Corona vaccine project in Iran". Mohraz is an Iranian physician, scientist, and AIDS specialist. She is a full professor (emeritus) of infectious diseases at Tehran University of Medical Sciences and head of the Iranian Centre for HIV/AIDS. Mohraz has also served, within the World Health Organization, as an expert on HIV/AIDS in Iran and the Eastern Mediterranean.