Recent from talks
CREB-binding protein
Knowledge base stats:
Talk channels stats:
Members stats:
CREB-binding protein
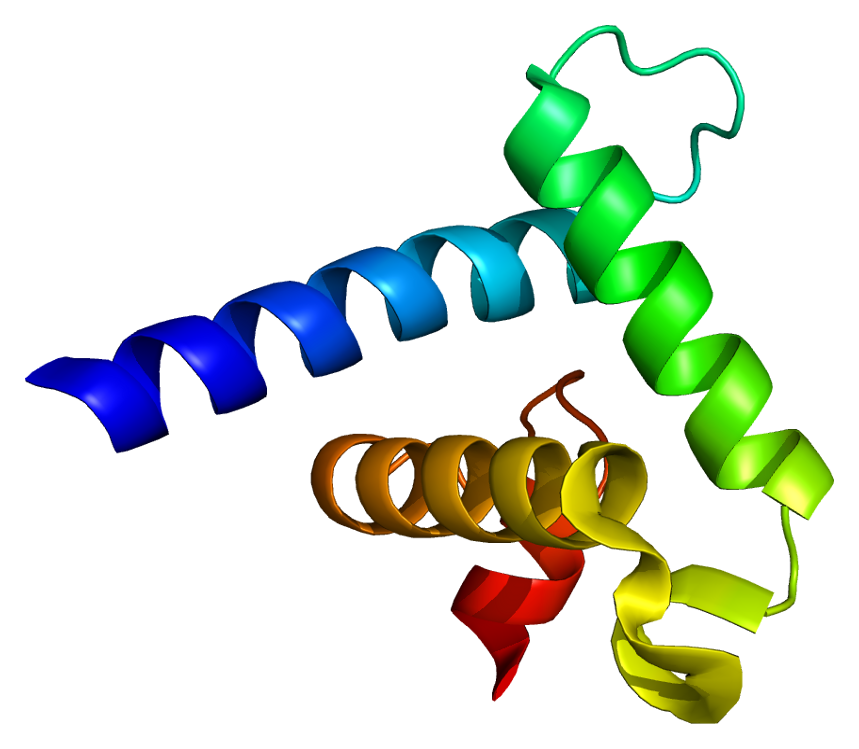
CREB-binding protein, also known as CREBBP or CBP or KAT3A, (where CREB is cAMP response element-binding protein) is a coactivator encoded by the CREBBP gene in humans, located on chromosome 16p13.3. CBP has intrinsic acetyltransferase functions; it is able to add acetyl groups to both transcription factors as well as histone lysines, the latter of which has been shown to alter chromatin structure making genes more accessible for transcription. This relatively unique acetyltransferase activity is also seen in another transcription enzyme, EP300 (p300). Together, they are known as the p300-CBP coactivator family and are known to associate with more than 16,000 genes in humans; however, while these proteins share many structural features, emerging evidence suggests that these two co-activators may promote transcription of genes with different biological functions.
For example, CBP alone has been implicated in a wide variety of pathophysiologies including colorectal cancer as well as head and neck squamous cell carcinoma. In these diseases, association of CBP with β-catenin has been shown to promote cancer cell proliferation and disease aggressiveness, whereas p300/ β-catenin leads to cell differentiation and/ or apoptosis. CBP has also been shown to help modulate liver function via maintenance of energy homeostasis in response to changes in cell nutrition conditions by regulating the activity of transcription factors and genes responsible for lipogenesis and gluconeogenesis. CBP is also implicated in the etiologies of several other diseases including hematologic malignancies and other solid tumors, diabetes, schizophrenia, Alzheimer's disease, depression, and many other neurological conditions.
The functional CBP is approximately 7362 nucleotides long and encodes 2,441 amino acids. CBP does not directly interact with promoter elements; it is brought to the site via protein-protein interactions that are the result of its different structural domains complexing with other transcriptional co-activators.
CBP has two transitional adapter zinc finger (TAZ) domains that each consist of four alpha helices stabilized by zinc ions. Both TAZ1 and TAZ2 domains favor the hydrophobic residues within amphipathic amino acid sequences, and their binding affinity is enhanced by the interaction of acidic residues with positive side chains. Factors that bind to TAZ2 are also potentially regulated via acetylation due to its proximity to the acetyltransferase domain (KAT).
Although there are three cysteine-histidine rich domains identified within CBP, only domains 1 and 3 (CH1, CH3) have had their structural functions resolved. A number of factors that associate with CBP bind to either the CH1 or CH3 domain, or to both, despite their locations at opposing ends of the protein. To date, little is known about the interaction between these two domains. The primary structure of CH1 and CH3 has been shown to break down into consensus sequences that are capable of chelating zinc ions (Zn2+). Experiments performed also showed that the residues in these sequences are obligatory for transcriptional coactivation to occur by either CH1 or CH3. The CH2 region, located in the middle of the protein, in its acetyltransferase domain, does not contain this consensus sequence, and has not been conclusively shown to bind zinc ions.
Aka KIX domain, CREB binding domain, MYB interaction domain
The kinase-inducible domain (KID) interacting domain (KIX) also is the protein domain on CBP and p300 where heterodimers form from the interaction of CBP (or p300) with other transcription factors and coactivators. It consists of three alpha and two 310 helices that have high affinity for amphipathic protein sequences. Interestingly, these helices can fold into a number of different conformers which enables the domain to maintain both a level of promiscuity while also exerting regulatory control. The KIX domain controls the rate of transcription and has been shown to be critical for hematopoietic differentiation. Some of the proteins that bind to this domain have been shown to bind competitively—such as CREB and Myb—whereas others bind via allosteric cooperation as in the case of MLL and Myb.
Bromodomains (BRD) consist of approximately 110 amino acids, that function to recognize acetylated lysine molecules. They consist of four left-handed alpha helices connected by loops which form hydrophobic binding pockets. The bromodomain of CBP binds to regions of the genome are rich with acetylated lysine residues, meaning they have lost their positive charge, decreasing histone affinity for DNA, which makes the region more open and accessible for transcription. Acetylated p53 and STAT3 have been shown to bind to CBP's bromodomain.
Hub AI
CREB-binding protein AI simulator
(@CREB-binding protein_simulator)
CREB-binding protein
CREB-binding protein, also known as CREBBP or CBP or KAT3A, (where CREB is cAMP response element-binding protein) is a coactivator encoded by the CREBBP gene in humans, located on chromosome 16p13.3. CBP has intrinsic acetyltransferase functions; it is able to add acetyl groups to both transcription factors as well as histone lysines, the latter of which has been shown to alter chromatin structure making genes more accessible for transcription. This relatively unique acetyltransferase activity is also seen in another transcription enzyme, EP300 (p300). Together, they are known as the p300-CBP coactivator family and are known to associate with more than 16,000 genes in humans; however, while these proteins share many structural features, emerging evidence suggests that these two co-activators may promote transcription of genes with different biological functions.
For example, CBP alone has been implicated in a wide variety of pathophysiologies including colorectal cancer as well as head and neck squamous cell carcinoma. In these diseases, association of CBP with β-catenin has been shown to promote cancer cell proliferation and disease aggressiveness, whereas p300/ β-catenin leads to cell differentiation and/ or apoptosis. CBP has also been shown to help modulate liver function via maintenance of energy homeostasis in response to changes in cell nutrition conditions by regulating the activity of transcription factors and genes responsible for lipogenesis and gluconeogenesis. CBP is also implicated in the etiologies of several other diseases including hematologic malignancies and other solid tumors, diabetes, schizophrenia, Alzheimer's disease, depression, and many other neurological conditions.
The functional CBP is approximately 7362 nucleotides long and encodes 2,441 amino acids. CBP does not directly interact with promoter elements; it is brought to the site via protein-protein interactions that are the result of its different structural domains complexing with other transcriptional co-activators.
CBP has two transitional adapter zinc finger (TAZ) domains that each consist of four alpha helices stabilized by zinc ions. Both TAZ1 and TAZ2 domains favor the hydrophobic residues within amphipathic amino acid sequences, and their binding affinity is enhanced by the interaction of acidic residues with positive side chains. Factors that bind to TAZ2 are also potentially regulated via acetylation due to its proximity to the acetyltransferase domain (KAT).
Although there are three cysteine-histidine rich domains identified within CBP, only domains 1 and 3 (CH1, CH3) have had their structural functions resolved. A number of factors that associate with CBP bind to either the CH1 or CH3 domain, or to both, despite their locations at opposing ends of the protein. To date, little is known about the interaction between these two domains. The primary structure of CH1 and CH3 has been shown to break down into consensus sequences that are capable of chelating zinc ions (Zn2+). Experiments performed also showed that the residues in these sequences are obligatory for transcriptional coactivation to occur by either CH1 or CH3. The CH2 region, located in the middle of the protein, in its acetyltransferase domain, does not contain this consensus sequence, and has not been conclusively shown to bind zinc ions.
Aka KIX domain, CREB binding domain, MYB interaction domain
The kinase-inducible domain (KID) interacting domain (KIX) also is the protein domain on CBP and p300 where heterodimers form from the interaction of CBP (or p300) with other transcription factors and coactivators. It consists of three alpha and two 310 helices that have high affinity for amphipathic protein sequences. Interestingly, these helices can fold into a number of different conformers which enables the domain to maintain both a level of promiscuity while also exerting regulatory control. The KIX domain controls the rate of transcription and has been shown to be critical for hematopoietic differentiation. Some of the proteins that bind to this domain have been shown to bind competitively—such as CREB and Myb—whereas others bind via allosteric cooperation as in the case of MLL and Myb.
Bromodomains (BRD) consist of approximately 110 amino acids, that function to recognize acetylated lysine molecules. They consist of four left-handed alpha helices connected by loops which form hydrophobic binding pockets. The bromodomain of CBP binds to regions of the genome are rich with acetylated lysine residues, meaning they have lost their positive charge, decreasing histone affinity for DNA, which makes the region more open and accessible for transcription. Acetylated p53 and STAT3 have been shown to bind to CBP's bromodomain.