Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Golimumab AI simulator
(@Golimumab_simulator)
Hub AI
Golimumab AI simulator
(@Golimumab_simulator)
Golimumab
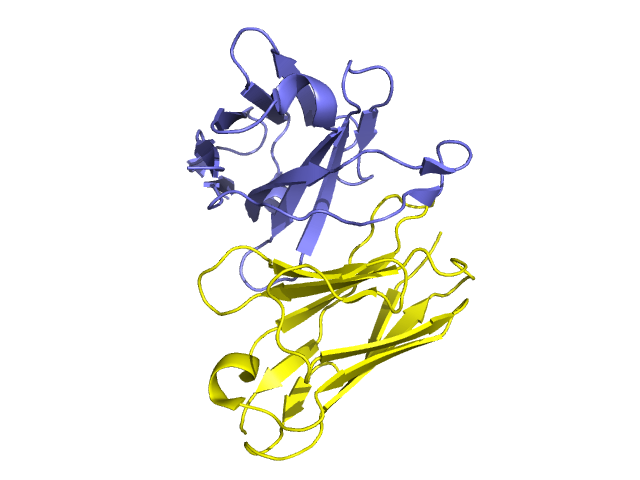
Golimumab, sold under the brand name Simponi, is a human monoclonal antibody which is used as an immunosuppressive medication. Golimumab targets tumor necrosis factor alpha (TNF-alpha), a pro-inflammatory molecule and hence is a TNF inhibitor. Reduction in C-reactive protein (CRP) levels, interleukin (IL)-6, intercellular adhesion molecules (ICAM)-1, matrix metalloproteinase (MMP)-3, and vascular endothelial growth factor (VEGF) demonstrates golimumab as an effective modulator of inflammatory markers and bone metabolism. Golimumab is given via subcutaneous injection.
Golimumab is a tumor necrosis factor alpha (TNF-a) inhibitor. Golimumab is a human monoclonal antibody that forms high affinity, stable complexes with both the soluble and transmembrane bioactive forms of human TNF-a, which prevents the binding of TNF-a to its receptors. By blocking TNF-a, golimumab reduces the inflammation and other symptoms of the diseases it is used for.
Golimumab is a therapeutic alternative on the World Health Organization's List of Essential Medicines.
The European Medicines Agency (EMA) approved the use of golimumab as a treatment for rheumatoid arthritis, psoriatic arthritis and ankylosing spondylitis. Golimumab was approved for the treatment by the US Food and Drug Administration (FDA) as well as the European Medicines Agency (EMA) in 2013 for the treatment of ulcerative colitis.
Golimumab is approved in Canada and the United States as a once monthly subcutaneous treatment for adults with moderately to severely active rheumatoid arthritis, psoriatic arthritis, juvenile idiopathic arthritis, and ankylosing spondylitis.
The most common adverse reactions (incidence >5%) are upper respiratory tract infection, nasopharyngitis, and injection site reactions.
Golimumab binds to both soluble and transmembrane forms of TNFα. The antibody was isolated from a hybridoma clone produced by transgenic mice immunized with human TNFα. The golimumab-secreting clone was selected after being assayed for human light and heavy chains and TNFα-binding. The commercial product is produced in a recombinant cell line cultured by continuous perfusion.
Golimumab was developed by Janssen Biotech, Inc. (formerly Centocor Biotech, Inc.) which also markets the product in the United States. Janssen markets golimumab in Canada, Central and South America, the Middle East, Africa and Asia Pacific. In the European Union, Russia, and Turkey, golimumab distribution rights are held by MSD (Ireland), a subsidiary of Merck & Co., Inc. In Japan, Indonesia, and Taiwan, distribution rights are held by Mitsubishi Tanabe Pharma Corporation.
Golimumab
Golimumab, sold under the brand name Simponi, is a human monoclonal antibody which is used as an immunosuppressive medication. Golimumab targets tumor necrosis factor alpha (TNF-alpha), a pro-inflammatory molecule and hence is a TNF inhibitor. Reduction in C-reactive protein (CRP) levels, interleukin (IL)-6, intercellular adhesion molecules (ICAM)-1, matrix metalloproteinase (MMP)-3, and vascular endothelial growth factor (VEGF) demonstrates golimumab as an effective modulator of inflammatory markers and bone metabolism. Golimumab is given via subcutaneous injection.
Golimumab is a tumor necrosis factor alpha (TNF-a) inhibitor. Golimumab is a human monoclonal antibody that forms high affinity, stable complexes with both the soluble and transmembrane bioactive forms of human TNF-a, which prevents the binding of TNF-a to its receptors. By blocking TNF-a, golimumab reduces the inflammation and other symptoms of the diseases it is used for.
Golimumab is a therapeutic alternative on the World Health Organization's List of Essential Medicines.
The European Medicines Agency (EMA) approved the use of golimumab as a treatment for rheumatoid arthritis, psoriatic arthritis and ankylosing spondylitis. Golimumab was approved for the treatment by the US Food and Drug Administration (FDA) as well as the European Medicines Agency (EMA) in 2013 for the treatment of ulcerative colitis.
Golimumab is approved in Canada and the United States as a once monthly subcutaneous treatment for adults with moderately to severely active rheumatoid arthritis, psoriatic arthritis, juvenile idiopathic arthritis, and ankylosing spondylitis.
The most common adverse reactions (incidence >5%) are upper respiratory tract infection, nasopharyngitis, and injection site reactions.
Golimumab binds to both soluble and transmembrane forms of TNFα. The antibody was isolated from a hybridoma clone produced by transgenic mice immunized with human TNFα. The golimumab-secreting clone was selected after being assayed for human light and heavy chains and TNFα-binding. The commercial product is produced in a recombinant cell line cultured by continuous perfusion.
Golimumab was developed by Janssen Biotech, Inc. (formerly Centocor Biotech, Inc.) which also markets the product in the United States. Janssen markets golimumab in Canada, Central and South America, the Middle East, Africa and Asia Pacific. In the European Union, Russia, and Turkey, golimumab distribution rights are held by MSD (Ireland), a subsidiary of Merck & Co., Inc. In Japan, Indonesia, and Taiwan, distribution rights are held by Mitsubishi Tanabe Pharma Corporation.