Recent from talks
Lead(IV) acetate
Knowledge base stats:
Talk channels stats:
Members stats:
Lead(IV) acetate
Lead(IV) acetate or lead tetraacetate is an metalorganic compound with chemical formula (CH3CO2)4Pb, often abbreviated as Pb(OAc)4, where Ac is acetyl. It is a colorless solid that is soluble in nonpolar, organic solvents, indicating that it is not a salt. It is degraded by moisture and is typically stored with additional acetic acid. The compound is used in organic synthesis.
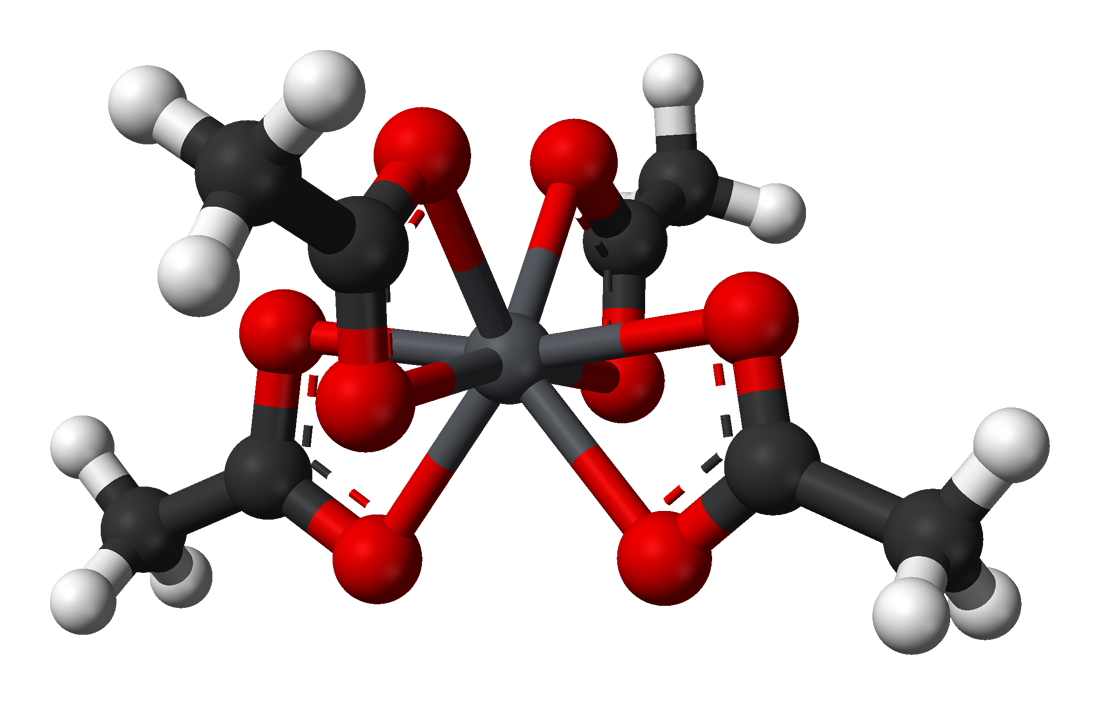
In the solid state the lead(IV) centers are coordinated by four acetate ions, which are bidentate, each coordinating via two oxygen atoms. The lead atom is 8 coordinate and the O atoms form a flattened trigonal dodecahedron.
It is typically prepared by treating of red lead with acetic acid and acetic anhydride (Ac2O), which absorbs water. The net reaction is shown:
The remaining lead(II) acetate can be partially oxidized to the tetraacetate by Cl2, with a PbCl2 by-product:
Lead tetraacetate is a strong oxidizing agent, a source of acetyloxy groups, and a general reagent for the preparation of organolead compounds. Some of its many uses in organic chemistry:
Lead(IV) acetate is toxic, because of lead. It is a neurotoxin. It badly affects the gum tissue, central nervous system, kidneys, blood, and reproductive system.
Hub AI
Lead(IV) acetate AI simulator
(@Lead(IV) acetate_simulator)
Lead(IV) acetate
Lead(IV) acetate or lead tetraacetate is an metalorganic compound with chemical formula (CH3CO2)4Pb, often abbreviated as Pb(OAc)4, where Ac is acetyl. It is a colorless solid that is soluble in nonpolar, organic solvents, indicating that it is not a salt. It is degraded by moisture and is typically stored with additional acetic acid. The compound is used in organic synthesis.
In the solid state the lead(IV) centers are coordinated by four acetate ions, which are bidentate, each coordinating via two oxygen atoms. The lead atom is 8 coordinate and the O atoms form a flattened trigonal dodecahedron.
It is typically prepared by treating of red lead with acetic acid and acetic anhydride (Ac2O), which absorbs water. The net reaction is shown:
The remaining lead(II) acetate can be partially oxidized to the tetraacetate by Cl2, with a PbCl2 by-product:
Lead tetraacetate is a strong oxidizing agent, a source of acetyloxy groups, and a general reagent for the preparation of organolead compounds. Some of its many uses in organic chemistry:
Lead(IV) acetate is toxic, because of lead. It is a neurotoxin. It badly affects the gum tissue, central nervous system, kidneys, blood, and reproductive system.