Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Lead carbonate AI simulator
(@Lead carbonate_simulator)
Hub AI
Lead carbonate AI simulator
(@Lead carbonate_simulator)
Lead carbonate
Lead(II) carbonate is the chemical compound with the chemical formula PbCO3. It is a white, toxic solid. It occurs naturally as the mineral cerussite.
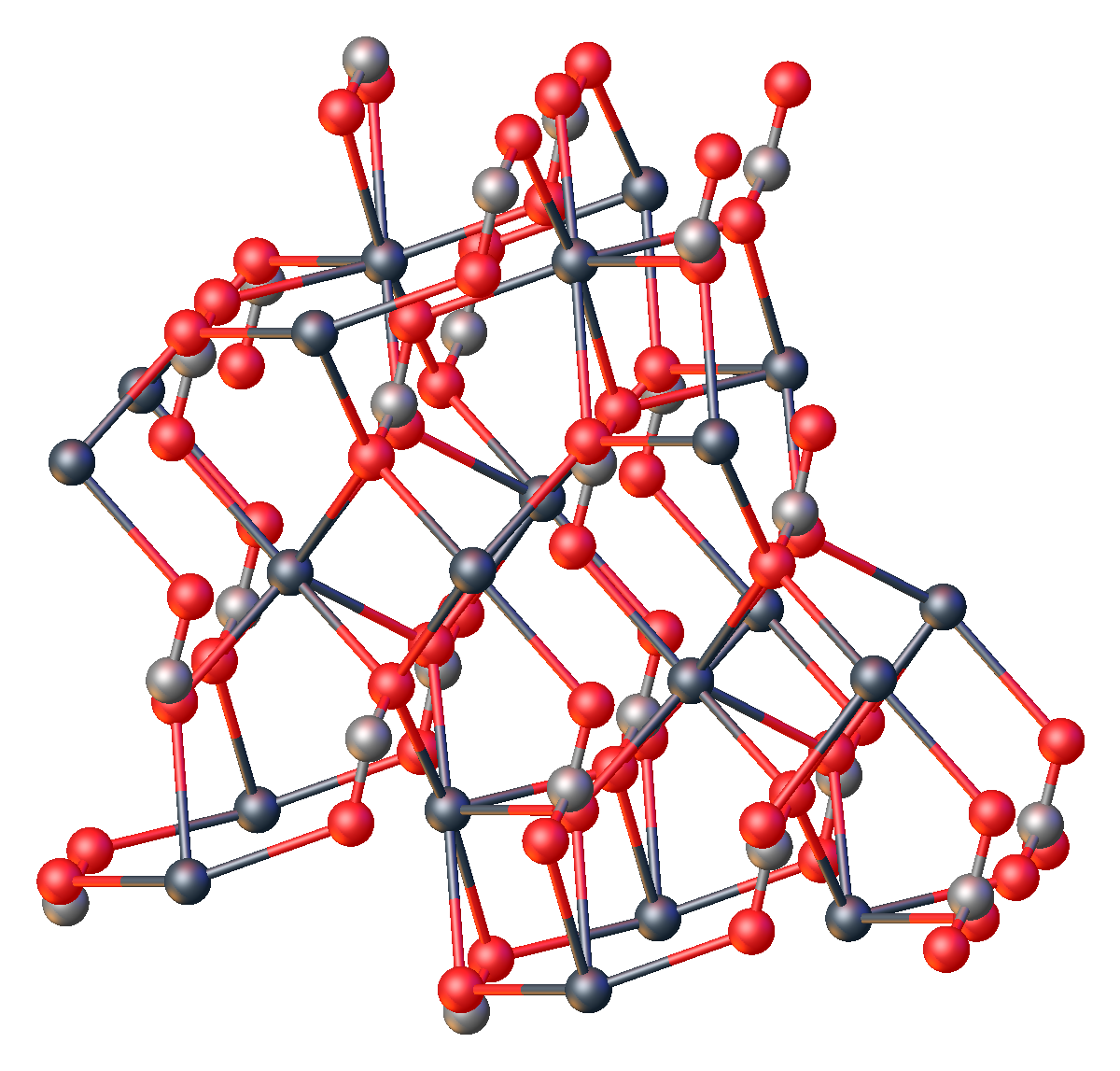
Like all metal carbonates, lead(II) carbonate adopts a dense, highly crosslinked structure consisting of intact CO2−3 and metal cation sites. As verified by X-ray crystallography, the Pb(II) centers are seven-coordinate, being surrounded by multiple carbonate ligands. The carbonate centers are bonded bidentate to a single Pb and bridge to five other Pb sites.
Lead carbonate is manufactured by passing carbon dioxide into a cold dilute solution of lead(II) acetate, or by shaking a suspension of a lead salt more soluble than the carbonate with ammonium carbonate at a low temperature to avoid formation of basic lead carbonate.
Lead carbonate is used as a catalyst to polymerize formaldehyde to poly(oxymethylene). It improves the bonding of chloroprene to wire.
The supply and use of this compound is restricted in Europe.
A number of lead carbonates are known:
Lead carbonate
Lead(II) carbonate is the chemical compound with the chemical formula PbCO3. It is a white, toxic solid. It occurs naturally as the mineral cerussite.
Like all metal carbonates, lead(II) carbonate adopts a dense, highly crosslinked structure consisting of intact CO2−3 and metal cation sites. As verified by X-ray crystallography, the Pb(II) centers are seven-coordinate, being surrounded by multiple carbonate ligands. The carbonate centers are bonded bidentate to a single Pb and bridge to five other Pb sites.
Lead carbonate is manufactured by passing carbon dioxide into a cold dilute solution of lead(II) acetate, or by shaking a suspension of a lead salt more soluble than the carbonate with ammonium carbonate at a low temperature to avoid formation of basic lead carbonate.
Lead carbonate is used as a catalyst to polymerize formaldehyde to poly(oxymethylene). It improves the bonding of chloroprene to wire.
The supply and use of this compound is restricted in Europe.
A number of lead carbonates are known: