Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Nuclear isomer AI simulator
(@Nuclear isomer_simulator)
Hub AI
Nuclear isomer AI simulator
(@Nuclear isomer_simulator)
Nuclear isomer
A nuclear isomer is a metastable state of an atomic nucleus, in which one or more nucleons (protons or neutrons) occupy excited state levels (higher energy levels). "Metastable" describes nuclei whose excited states have half-lives of 10−9 seconds or longer, 100 to 1000 times longer than the half-lives of the excited nuclear states that decay with a "prompt" half-life (ordinarily on the order of 10−12 seconds). Some references recommend 5×10−9 seconds to distinguish the metastable half-life from the normal "prompt" gamma-emission half-life.
The half-lives of a number of isomers are far longer than this and may be minutes, hours, or years. For example, the 180m
73Ta nuclear isomer survives so long (at least 2.9×1017 years) that it has never been observed to decay spontaneously, and occurs naturally as a primordial nuclide, though uncommon at only 1/8000 of all tantalum. The second most stable isomer is 210m
83Bi, which does not occur naturally; its half-life is 3.04×106 years to alpha decay. The half-life of a nuclear isomer can exceed that of the ground state of the same nuclide, as with the two above, as well as, for example, 186m
75Re, 192m2
77Ir, 212m
84Po, 242m
95Am and multiple holmium isomers.
The gamma decay from a metastable state is referred to as isomeric transition (IT), or internal transition, though it resembles shorter-lived "prompt" gamma decays in all external aspects with the exception of the longer life. This is generally associated with a high nuclear spin change, or "forbiddenness", which would be required in gamma emission to reach the ground state; this is even more true of beta decays. A low transition energy both slows the transition rate and makes it more likely that only highly forbidden decays are available, so most long-lived isomers have a relatively low excitation energy above the ground state (in the extreme case of thorium-229m, low excitation alone causes the measurably long life). In 210m
83Bi, the forbiddenness of available beta and gamma decays is so high that alpha decay is observed exclusively, though even that is more slow than for the ground state. For most lighter isomers including 180m
73Ta, alpha decay is not practically available, but others are not quite so forbidden as those two.
The first nuclear isomer and decay-daughter system (uranium X2/uranium Z, now known as 234m
91Pa/234
91Pa) was discovered by Otto Hahn in 1921.
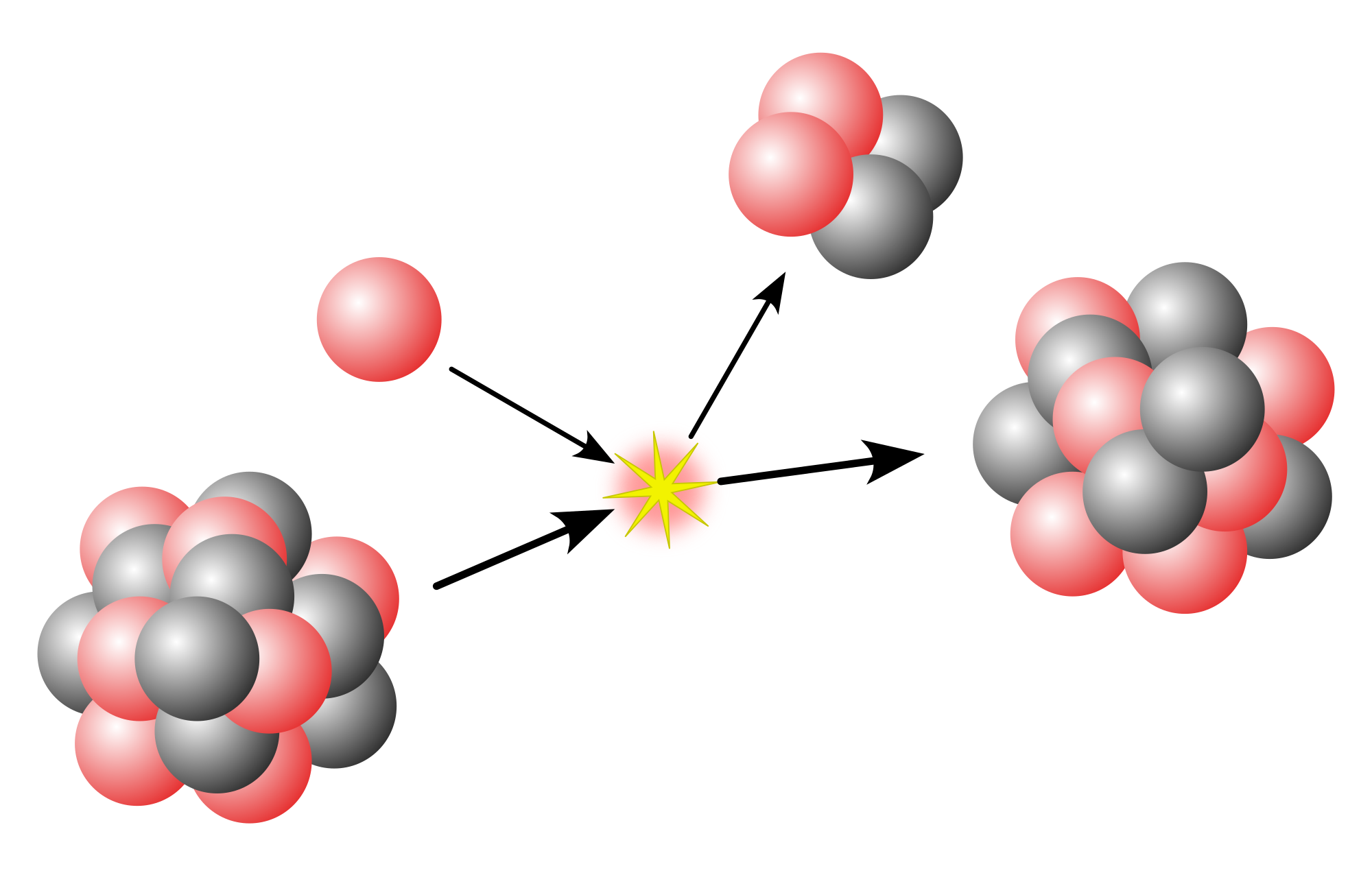
Metastable isomers can be produced through any nuclear reaction, including radioactive decay, neutron capture, nuclear fission, and bombardment by accelerated charged particles. A nucleus produced this way generally starts its existence in an excited state that loses its excess energy through the emission of one or more gamma rays or conversion electrons. This is normally a "prompt" process, but sometimes does not rapidly reach to the nuclear ground state, in which case a metastable isomer has formed. This usually occurs as a spin isomer when the formation of an intermediate excited state has a spin far different from that of the ground state. Gamma-ray emission is hindered if the spin of the post-emission state differs greatly from that of the emitting state, and if the excitation energy is low; such excited states will generally have long lives and be considered metastable.
After fission, several of the fission fragments may be produced in a metastable isomeric state, after their prompt de-excitation. At the end of this process, the nuclei can populate both the ground and the isomeric states. If the half-life of an isomer is long enough, it is possible to measure its production rate, and comparing it to that of the ground state gives the so-called isomeric yield ratio.
A particular kind of metastable isomer is the fission isomer or shape isomer. Most actinide nuclei in their ground states are not spherical, but rather prolate spheroidal, with an axis of symmetry longer than the other axes, similar to an American football or rugby ball. This geometry can result in quantum-mechanical states where the distribution of protons and neutrons is so much further from spherical geometry that de-excitation to the nuclear ground state is strongly hindered. In general, these states either de-excite to the ground state far more slowly than a "usual" excited state, or they undergo spontaneous fission with half-lives of the order of nanoseconds or microseconds—a very short time, but many orders of magnitude longer than the half-life of a more usual nuclear excited state. Fission isomers may be denoted with a postscript or superscript "f" rather than "m", so that a fission isomer, e.g. of plutonium-240, can be denoted as plutonium-240f or 240f
94Pu.
Metastable isomers of a particular isotope are usually designated with an "m". This designation is placed after the mass number of the atom; for example, cobalt-58m1 is abbreviated 58m1
27Co, where 27 is the atomic number of cobalt. For isotopes with more than one metastable isomer, "indices" are placed after the designation, and the labeling becomes m1, m2, m3, and so on. Increasing indices, m1, m2, etc., correlate with increasing levels of excitation energy stored in each of the isomeric states (e.g., hafnium-178m2, or 178m2
72Hf). The index may be omitted if only one isomer is relevant.
Nuclear isomer
A nuclear isomer is a metastable state of an atomic nucleus, in which one or more nucleons (protons or neutrons) occupy excited state levels (higher energy levels). "Metastable" describes nuclei whose excited states have half-lives of 10−9 seconds or longer, 100 to 1000 times longer than the half-lives of the excited nuclear states that decay with a "prompt" half-life (ordinarily on the order of 10−12 seconds). Some references recommend 5×10−9 seconds to distinguish the metastable half-life from the normal "prompt" gamma-emission half-life.
The half-lives of a number of isomers are far longer than this and may be minutes, hours, or years. For example, the 180m
73Ta nuclear isomer survives so long (at least 2.9×1017 years) that it has never been observed to decay spontaneously, and occurs naturally as a primordial nuclide, though uncommon at only 1/8000 of all tantalum. The second most stable isomer is 210m
83Bi, which does not occur naturally; its half-life is 3.04×106 years to alpha decay. The half-life of a nuclear isomer can exceed that of the ground state of the same nuclide, as with the two above, as well as, for example, 186m
75Re, 192m2
77Ir, 212m
84Po, 242m
95Am and multiple holmium isomers.
The gamma decay from a metastable state is referred to as isomeric transition (IT), or internal transition, though it resembles shorter-lived "prompt" gamma decays in all external aspects with the exception of the longer life. This is generally associated with a high nuclear spin change, or "forbiddenness", which would be required in gamma emission to reach the ground state; this is even more true of beta decays. A low transition energy both slows the transition rate and makes it more likely that only highly forbidden decays are available, so most long-lived isomers have a relatively low excitation energy above the ground state (in the extreme case of thorium-229m, low excitation alone causes the measurably long life). In 210m
83Bi, the forbiddenness of available beta and gamma decays is so high that alpha decay is observed exclusively, though even that is more slow than for the ground state. For most lighter isomers including 180m
73Ta, alpha decay is not practically available, but others are not quite so forbidden as those two.
The first nuclear isomer and decay-daughter system (uranium X2/uranium Z, now known as 234m
91Pa/234
91Pa) was discovered by Otto Hahn in 1921.
Metastable isomers can be produced through any nuclear reaction, including radioactive decay, neutron capture, nuclear fission, and bombardment by accelerated charged particles. A nucleus produced this way generally starts its existence in an excited state that loses its excess energy through the emission of one or more gamma rays or conversion electrons. This is normally a "prompt" process, but sometimes does not rapidly reach to the nuclear ground state, in which case a metastable isomer has formed. This usually occurs as a spin isomer when the formation of an intermediate excited state has a spin far different from that of the ground state. Gamma-ray emission is hindered if the spin of the post-emission state differs greatly from that of the emitting state, and if the excitation energy is low; such excited states will generally have long lives and be considered metastable.
After fission, several of the fission fragments may be produced in a metastable isomeric state, after their prompt de-excitation. At the end of this process, the nuclei can populate both the ground and the isomeric states. If the half-life of an isomer is long enough, it is possible to measure its production rate, and comparing it to that of the ground state gives the so-called isomeric yield ratio.
A particular kind of metastable isomer is the fission isomer or shape isomer. Most actinide nuclei in their ground states are not spherical, but rather prolate spheroidal, with an axis of symmetry longer than the other axes, similar to an American football or rugby ball. This geometry can result in quantum-mechanical states where the distribution of protons and neutrons is so much further from spherical geometry that de-excitation to the nuclear ground state is strongly hindered. In general, these states either de-excite to the ground state far more slowly than a "usual" excited state, or they undergo spontaneous fission with half-lives of the order of nanoseconds or microseconds—a very short time, but many orders of magnitude longer than the half-life of a more usual nuclear excited state. Fission isomers may be denoted with a postscript or superscript "f" rather than "m", so that a fission isomer, e.g. of plutonium-240, can be denoted as plutonium-240f or 240f
94Pu.
Metastable isomers of a particular isotope are usually designated with an "m". This designation is placed after the mass number of the atom; for example, cobalt-58m1 is abbreviated 58m1
27Co, where 27 is the atomic number of cobalt. For isotopes with more than one metastable isomer, "indices" are placed after the designation, and the labeling becomes m1, m2, m3, and so on. Increasing indices, m1, m2, etc., correlate with increasing levels of excitation energy stored in each of the isomeric states (e.g., hafnium-178m2, or 178m2
72Hf). The index may be omitted if only one isomer is relevant.