Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Spontaneous fission AI simulator
(@Spontaneous fission_simulator)
Hub AI
Spontaneous fission AI simulator
(@Spontaneous fission_simulator)
Spontaneous fission
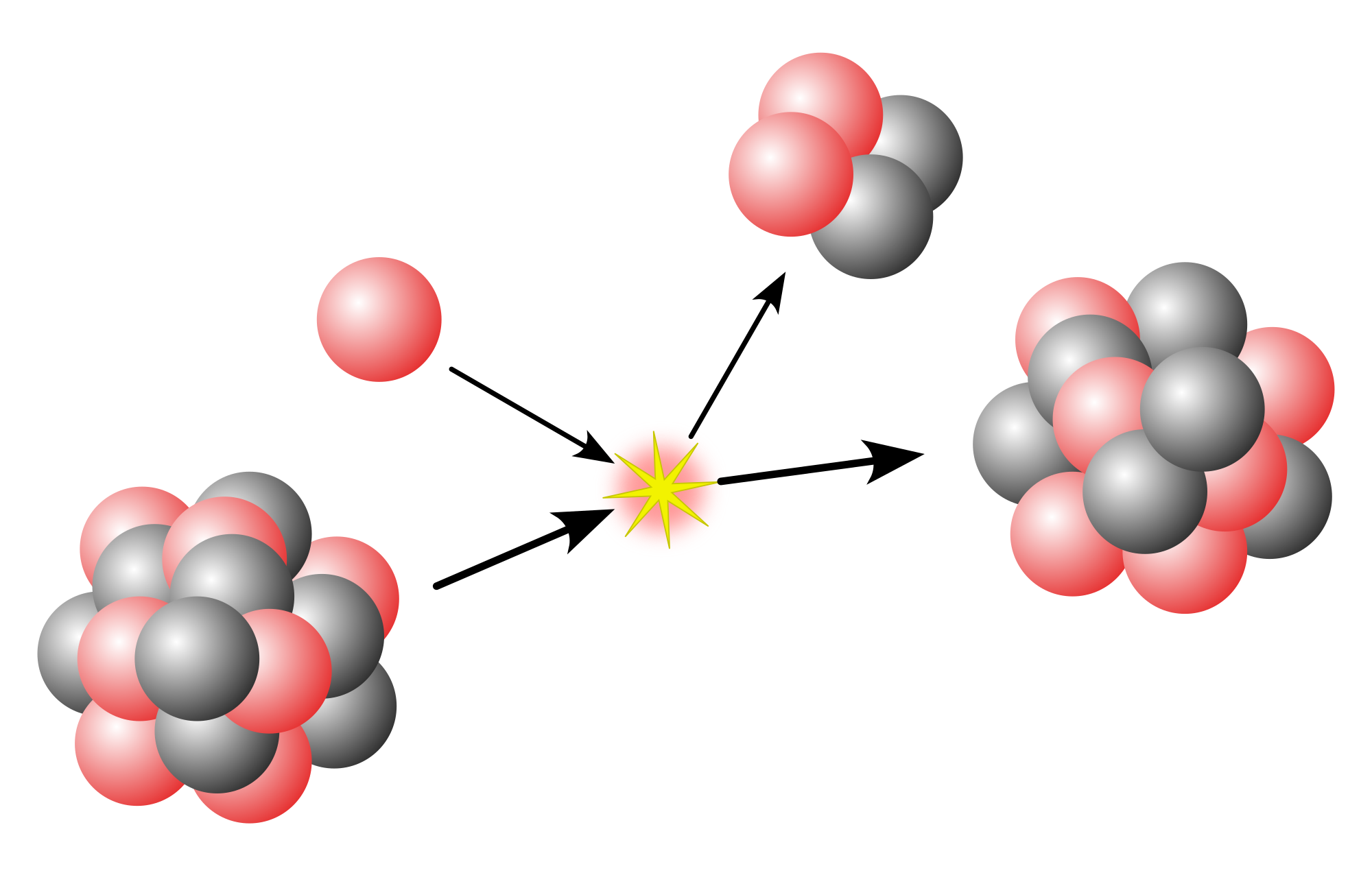
Spontaneous fission (SF) is a form of radioactive decay in which a heavy atomic nucleus splits into two or more lighter nuclei. In contrast to induced fission, there is no inciting particle to trigger the decay; it is a purely probabilistic process.
Spontaneous fission is a dominant decay mode for superheavy elements, with nuclear stability generally falling as nuclear mass increases. It thus forms a practical limit to heavy element nucleon number. Heavier nuclides may be created instantaneously by physical processes, both natural (via the r-process) and artificial, though rapidly decay to more stable nuclides. As such, apart from minor decay branches in primordial radionuclides, spontaneous fission is not observed in nature.
Observed fission half-lives range from 60 nanoseconds (252
104Rf) to greater than the current age of the universe (232
90Th).
Following the discovery of induced fission by Otto Hahn and Fritz Strassmann in 1938, Soviet physicists Georgy Flyorov and Konstantin Petrzhak began conducting experiments to explore the effects of incident neutron energy on uranium nuclei. Their equipment recorded fission fragments even when no neutrons were present to induce the decay, and the effect persisted even after the equipment was moved 60 meters underground into the tunnels of the Moscow Metro's Dinamo station in an effort to insulate it from the effects of cosmic rays. The discovery of induced fission itself had come as a surprise, and no other mechanism was known that could account for the observed decays. Such an effect could only be explained by spontaneous fission of the uranium nuclei without external influence.
Spontaneous fission arises as a result of competition between the attractive properties of the strong nuclear force and the mutual coulombic repulsion of the constituent protons. Nuclear binding energy increases in proportion to atomic mass number (A), while coulombic repulsion increases with the square of the proton number (Z). Thus, at high mass and proton numbers, coulombic repulsion overpowers the nuclear binding forces, and the nucleus is energetically more stable as two separate fragments than as a single bound system.
Spontaneous fission is usually a slow process, as the nucleus cannot simply jump to the lower energy (divided) state. Instead it must tunnel through a potential barrier, with a probability determined by the height of the barrier. Such a barrier is energetically possible for all A ≥ 93, though its height generally decreases with increasing Z, and fission is only practically observed for A ≥ 232.
The stability of a nuclide against fission is expressed as the ratio of the Coulomb energy to the surface energy, which can be empirically estimated as the fissility parameter, x: with and . For light nuclei, x is small and a sizeable fission barrier exists. As nuclear mass increases, so too does the fissility parameter, eventually approaching and exceeding unity, where stability against fission is lost altogether.
Shell effects and nucleon pairing effects may further affect observed half-lives. Decays of odd-A nuclides are hindered by 3–5 orders of magnitude compared to even–even nuclides. The barrier to fission is expected to be zero around A = 300, though an island of stability may exist centred around Z = 114, N = 184.
Spontaneous fission
Spontaneous fission (SF) is a form of radioactive decay in which a heavy atomic nucleus splits into two or more lighter nuclei. In contrast to induced fission, there is no inciting particle to trigger the decay; it is a purely probabilistic process.
Spontaneous fission is a dominant decay mode for superheavy elements, with nuclear stability generally falling as nuclear mass increases. It thus forms a practical limit to heavy element nucleon number. Heavier nuclides may be created instantaneously by physical processes, both natural (via the r-process) and artificial, though rapidly decay to more stable nuclides. As such, apart from minor decay branches in primordial radionuclides, spontaneous fission is not observed in nature.
Observed fission half-lives range from 60 nanoseconds (252
104Rf) to greater than the current age of the universe (232
90Th).
Following the discovery of induced fission by Otto Hahn and Fritz Strassmann in 1938, Soviet physicists Georgy Flyorov and Konstantin Petrzhak began conducting experiments to explore the effects of incident neutron energy on uranium nuclei. Their equipment recorded fission fragments even when no neutrons were present to induce the decay, and the effect persisted even after the equipment was moved 60 meters underground into the tunnels of the Moscow Metro's Dinamo station in an effort to insulate it from the effects of cosmic rays. The discovery of induced fission itself had come as a surprise, and no other mechanism was known that could account for the observed decays. Such an effect could only be explained by spontaneous fission of the uranium nuclei without external influence.
Spontaneous fission arises as a result of competition between the attractive properties of the strong nuclear force and the mutual coulombic repulsion of the constituent protons. Nuclear binding energy increases in proportion to atomic mass number (A), while coulombic repulsion increases with the square of the proton number (Z). Thus, at high mass and proton numbers, coulombic repulsion overpowers the nuclear binding forces, and the nucleus is energetically more stable as two separate fragments than as a single bound system.
Spontaneous fission is usually a slow process, as the nucleus cannot simply jump to the lower energy (divided) state. Instead it must tunnel through a potential barrier, with a probability determined by the height of the barrier. Such a barrier is energetically possible for all A ≥ 93, though its height generally decreases with increasing Z, and fission is only practically observed for A ≥ 232.
The stability of a nuclide against fission is expressed as the ratio of the Coulomb energy to the surface energy, which can be empirically estimated as the fissility parameter, x: with and . For light nuclei, x is small and a sizeable fission barrier exists. As nuclear mass increases, so too does the fissility parameter, eventually approaching and exceeding unity, where stability against fission is lost altogether.
Shell effects and nucleon pairing effects may further affect observed half-lives. Decays of odd-A nuclides are hindered by 3–5 orders of magnitude compared to even–even nuclides. The barrier to fission is expected to be zero around A = 300, though an island of stability may exist centred around Z = 114, N = 184.