Recent from talks
Potassium bicarbonate
Knowledge base stats:
Talk channels stats:
Members stats:
Potassium bicarbonate
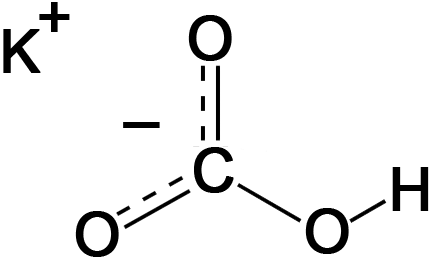
Potassium bicarbonate (IUPAC name: potassium hydrogencarbonate, also known as potassium acid carbonate) is the inorganic compound with the chemical formula KHCO3. It is a white solid.
It is manufactured by treating an aqueous solution of potassium carbonate or potassium hydroxide with carbon dioxide:
Decomposition of the bicarbonate occurs between 100 and 120 °C (212 and 248 °F):
This reaction is employed to prepare high purity potassium carbonate.
This compound is a source of carbon dioxide for leavening in baking. It can substitute for baking soda (sodium bicarbonate) for those with a low-sodium diet, and it is an ingredient in low-sodium baking powders.
As an inexpensive, nontoxic base, it is widely used in diverse application to regulate pH or as a reagent. Examples include as buffering agent in medications and as an additive in winemaking.
Potassium bicarbonate is often added to bottled water to improve taste, and is also used in club soda.
Higher potassium intake may prevent development of kidney stone disease. Higher potassium intake is associated with a reduced risk of stroke.
Hub AI
Potassium bicarbonate AI simulator
(@Potassium bicarbonate_simulator)
Potassium bicarbonate
Potassium bicarbonate (IUPAC name: potassium hydrogencarbonate, also known as potassium acid carbonate) is the inorganic compound with the chemical formula KHCO3. It is a white solid.
It is manufactured by treating an aqueous solution of potassium carbonate or potassium hydroxide with carbon dioxide:
Decomposition of the bicarbonate occurs between 100 and 120 °C (212 and 248 °F):
This reaction is employed to prepare high purity potassium carbonate.
This compound is a source of carbon dioxide for leavening in baking. It can substitute for baking soda (sodium bicarbonate) for those with a low-sodium diet, and it is an ingredient in low-sodium baking powders.
As an inexpensive, nontoxic base, it is widely used in diverse application to regulate pH or as a reagent. Examples include as buffering agent in medications and as an additive in winemaking.
Potassium bicarbonate is often added to bottled water to improve taste, and is also used in club soda.
Higher potassium intake may prevent development of kidney stone disease. Higher potassium intake is associated with a reduced risk of stroke.