Recent from talks
Thermosiphon
Knowledge base stats:
Talk channels stats:
Members stats:
Thermosiphon
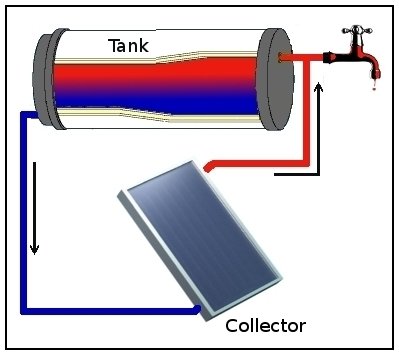
A thermosiphon (or thermosyphon) is a device that employs a method of passive heat exchange based on natural convection, which circulates a fluid without the necessity of a mechanical pump. Thermosiphoning is used for circulation of liquids and volatile gases in heating and cooling applications such as heat pumps, water heaters, boilers and furnaces. Thermosiphoning also occurs across air temperature gradients such as those occurring in a wood-fire chimney or solar chimney.
This circulation can either be open-loop, as when the substance in a holding tank is passed in one direction via a heated transfer tube mounted at the bottom of the tank to a distribution point — even one mounted above the originating tank — or it can be a vertical closed-loop circuit with return to the original container. Its purpose is to simplify the transfer of liquid or gas while avoiding the cost and complexity of a conventional pump.
Natural convection of the liquid starts when heat transfer to the liquid gives rise to a temperature difference from one side of the loop to the other. The phenomenon of thermal expansion means that a temperature difference will have a corresponding difference in density across the loop. The warmer fluid on one side of the loop is less dense and thus more buoyant than the cooler fluid on the other side. The warmer fluid will "float" above the cooler fluid, and the cooler fluid will "sink" below the warmer fluid. This phenomenon of natural convection is known by the saying "heat rises". Convection moves the heated liquid upwards in the system as it is simultaneously replaced by cooler liquid returning by gravity. A good thermosiphon has very little hydraulic resistance so that liquid can flow easily under the relatively low pressure produced by natural convection.
In some situations the flow of liquid may be reduced further, or stopped, perhaps because the loop is not entirely full of liquid. In this case, the system no longer convects, so it is not a usual "thermosiphon".
Heat can still be transferred in this system by the evaporation and condensation of vapor; however, the system is properly classified as a heat pipe thermosyphon. If the system also contains other fluids, such as air, then the heat flux density will be less than in a real heat pipe, which contains only a single substance.
The thermosiphon has been sometimes incorrectly described as a 'gravity return heat pipe'. Heat pipes usually have a wick to return the condensate to the evaporator via capillary action. A wick is not needed in a thermosiphon because gravity moves the liquid. The wick allows heat pipes to transfer heat when there is no gravity, which is useful in space. A thermosiphon is "simpler" than a heat pipe.
(Single-phase) thermosiphons can only transfer heat "upward", or away from the acceleration vector. Thus, orientation is much more important for thermosiphons than for heatpipes. Also, thermosiphons can fail because of a bubble in the loop, and require a circulating loop of pipes.
If the piping of a thermosiphon resists flow, or excessive heat is applied, the liquid may boil. Since the gas is more buoyant than the liquid, the convective pressure is greater. This is a well known invention called a reboiler. A group of reboilers attached to a pair of plena is called a calandria. In some circumstances, for example the cooling system for an older (pre 1950s) car, the boiling of the fluid will cause the system to stop working, as the volume of steam created displaces too much of the water and circulation stops.
Hub AI
Thermosiphon AI simulator
(@Thermosiphon_simulator)
Thermosiphon
A thermosiphon (or thermosyphon) is a device that employs a method of passive heat exchange based on natural convection, which circulates a fluid without the necessity of a mechanical pump. Thermosiphoning is used for circulation of liquids and volatile gases in heating and cooling applications such as heat pumps, water heaters, boilers and furnaces. Thermosiphoning also occurs across air temperature gradients such as those occurring in a wood-fire chimney or solar chimney.
This circulation can either be open-loop, as when the substance in a holding tank is passed in one direction via a heated transfer tube mounted at the bottom of the tank to a distribution point — even one mounted above the originating tank — or it can be a vertical closed-loop circuit with return to the original container. Its purpose is to simplify the transfer of liquid or gas while avoiding the cost and complexity of a conventional pump.
Natural convection of the liquid starts when heat transfer to the liquid gives rise to a temperature difference from one side of the loop to the other. The phenomenon of thermal expansion means that a temperature difference will have a corresponding difference in density across the loop. The warmer fluid on one side of the loop is less dense and thus more buoyant than the cooler fluid on the other side. The warmer fluid will "float" above the cooler fluid, and the cooler fluid will "sink" below the warmer fluid. This phenomenon of natural convection is known by the saying "heat rises". Convection moves the heated liquid upwards in the system as it is simultaneously replaced by cooler liquid returning by gravity. A good thermosiphon has very little hydraulic resistance so that liquid can flow easily under the relatively low pressure produced by natural convection.
In some situations the flow of liquid may be reduced further, or stopped, perhaps because the loop is not entirely full of liquid. In this case, the system no longer convects, so it is not a usual "thermosiphon".
Heat can still be transferred in this system by the evaporation and condensation of vapor; however, the system is properly classified as a heat pipe thermosyphon. If the system also contains other fluids, such as air, then the heat flux density will be less than in a real heat pipe, which contains only a single substance.
The thermosiphon has been sometimes incorrectly described as a 'gravity return heat pipe'. Heat pipes usually have a wick to return the condensate to the evaporator via capillary action. A wick is not needed in a thermosiphon because gravity moves the liquid. The wick allows heat pipes to transfer heat when there is no gravity, which is useful in space. A thermosiphon is "simpler" than a heat pipe.
(Single-phase) thermosiphons can only transfer heat "upward", or away from the acceleration vector. Thus, orientation is much more important for thermosiphons than for heatpipes. Also, thermosiphons can fail because of a bubble in the loop, and require a circulating loop of pipes.
If the piping of a thermosiphon resists flow, or excessive heat is applied, the liquid may boil. Since the gas is more buoyant than the liquid, the convective pressure is greater. This is a well known invention called a reboiler. A group of reboilers attached to a pair of plena is called a calandria. In some circumstances, for example the cooling system for an older (pre 1950s) car, the boiling of the fluid will cause the system to stop working, as the volume of steam created displaces too much of the water and circulation stops.