Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Thioureas AI simulator
(@Thioureas_simulator)
Hub AI
Thioureas AI simulator
(@Thioureas_simulator)
Thioureas
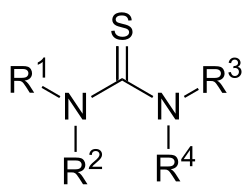
In organic chemistry, thioureas are members of a family of organosulfur compounds with the formula S=C(NR2)2 and structure R2N−C(=S)−NR2. The parent member of this class of compounds is thiourea (S=C(NH2)2). Substituted thioureas are found in several commercial chemicals.
Thioureas have a trigonal planar molecular geometry of the N2C=S core. The C=S bond distance is near 1.71 Å, which is 0.1 Å longer than in normal ketones (R2C=O). The C–N bond distances are short. Thioureas occurs in two tautomeric forms.
On the other hand, some compounds depicted as isothioureas and in fact thioureas, one example being mercaptobenzimidazole.
N,N′-unsubstituted thioureas can be prepared by treating the corresponding cyanamide with hydrogen sulfide or similar sulfide sources. Organic ammonium salts react with potassium thiocyanate as the source of the thiocarbonyl (C=S).
Alternatively, N,N′-disubstituted thioureas can be prepared by coupling two amines with thiophosgene:
Amines also condense with organic thiocyanates to give thioureas:
Cyclic thioureas are prepared by transamidation of thiourea with diamines. Ethylene thiourea is synthesized by treating ethylenediamine with carbon disulfide. In some cases, thioureas can be prepared by thiation of ureas using phosphorus pentasulfide.
Thioureas are susceptible to tautomerization. For the parent thiourea, the thione tautomer predominates in aqueous solutions. The thiol form, known as an isothiourea, can be encountered in substituted compounds such as isothiouronium salts.
Thioureas
In organic chemistry, thioureas are members of a family of organosulfur compounds with the formula S=C(NR2)2 and structure R2N−C(=S)−NR2. The parent member of this class of compounds is thiourea (S=C(NH2)2). Substituted thioureas are found in several commercial chemicals.
Thioureas have a trigonal planar molecular geometry of the N2C=S core. The C=S bond distance is near 1.71 Å, which is 0.1 Å longer than in normal ketones (R2C=O). The C–N bond distances are short. Thioureas occurs in two tautomeric forms.
On the other hand, some compounds depicted as isothioureas and in fact thioureas, one example being mercaptobenzimidazole.
N,N′-unsubstituted thioureas can be prepared by treating the corresponding cyanamide with hydrogen sulfide or similar sulfide sources. Organic ammonium salts react with potassium thiocyanate as the source of the thiocarbonyl (C=S).
Alternatively, N,N′-disubstituted thioureas can be prepared by coupling two amines with thiophosgene:
Amines also condense with organic thiocyanates to give thioureas:
Cyclic thioureas are prepared by transamidation of thiourea with diamines. Ethylene thiourea is synthesized by treating ethylenediamine with carbon disulfide. In some cases, thioureas can be prepared by thiation of ureas using phosphorus pentasulfide.
Thioureas are susceptible to tautomerization. For the parent thiourea, the thione tautomer predominates in aqueous solutions. The thiol form, known as an isothiourea, can be encountered in substituted compounds such as isothiouronium salts.