Recent from talks
Knowledge base stats:
Talk channels stats:
Members stats:
Tin(IV) oxide
Tin(IV) oxide, also known as stannic oxide, is the inorganic compound with the formula SnO2. The mineral form of SnO2 is called cassiterite, and this is the main ore of tin. With many other names, this oxide of tin is an important material in tin chemistry. It is a colourless, diamagnetic, amphoteric solid.
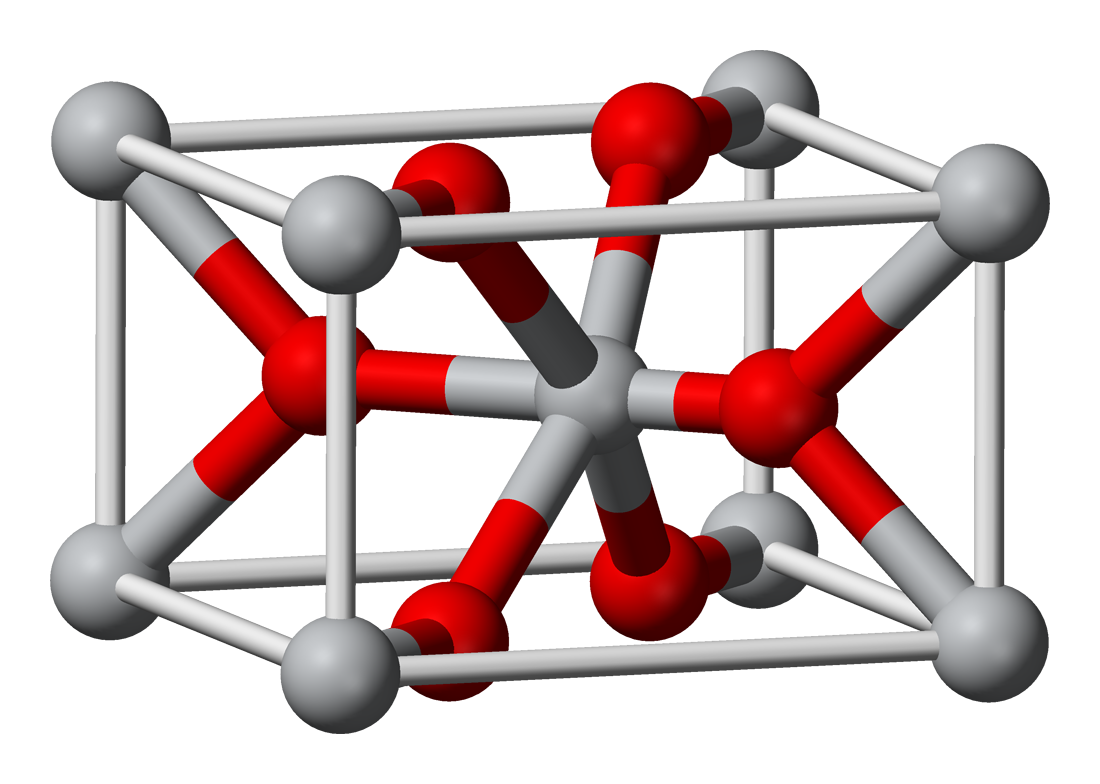
Tin(IV) oxide crystallises with the rutile structure. As such the tin atoms are six coordinate and the oxygen atoms three coordinate. SnO2 is usually regarded as an oxygen-deficient n-type semiconductor.
Hydrous forms of SnO2 have been described as stannic acid. Such materials appear to be hydrated particles of SnO2 where the composition reflects the particle size.
Tin(IV) oxide occurs naturally. Synthetic tin(IV) oxide is produced by burning tin metal in air. Annual production is in the range of 10 kilotons. SnO2 is reduced industrially to the metal with carbon in a reverberatory furnace at 1200–1300 °C.
The reaction from tin(IV) oxide with hot carbon monoxide is practiced on a large scale as this carbothermal reduction is used to obtain tin metal from its ores:
Some other reactions relevant to purifying tin from its ores are:
SnO2 converts to the monoxide at 1500 °C:
SnO2 is insoluble in water. It dissolves in sulfuric acid and in molten sodium hydroxide. It is not amphoteric. Like rutile, it is not attacked by solutions of acid or base.
Hub AI
Tin(IV) oxide AI simulator
(@Tin(IV) oxide_simulator)
Tin(IV) oxide
Tin(IV) oxide, also known as stannic oxide, is the inorganic compound with the formula SnO2. The mineral form of SnO2 is called cassiterite, and this is the main ore of tin. With many other names, this oxide of tin is an important material in tin chemistry. It is a colourless, diamagnetic, amphoteric solid.
Tin(IV) oxide crystallises with the rutile structure. As such the tin atoms are six coordinate and the oxygen atoms three coordinate. SnO2 is usually regarded as an oxygen-deficient n-type semiconductor.
Hydrous forms of SnO2 have been described as stannic acid. Such materials appear to be hydrated particles of SnO2 where the composition reflects the particle size.
Tin(IV) oxide occurs naturally. Synthetic tin(IV) oxide is produced by burning tin metal in air. Annual production is in the range of 10 kilotons. SnO2 is reduced industrially to the metal with carbon in a reverberatory furnace at 1200–1300 °C.
The reaction from tin(IV) oxide with hot carbon monoxide is practiced on a large scale as this carbothermal reduction is used to obtain tin metal from its ores:
Some other reactions relevant to purifying tin from its ores are:
SnO2 converts to the monoxide at 1500 °C:
SnO2 is insoluble in water. It dissolves in sulfuric acid and in molten sodium hydroxide. It is not amphoteric. Like rutile, it is not attacked by solutions of acid or base.