Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
ABO blood group system
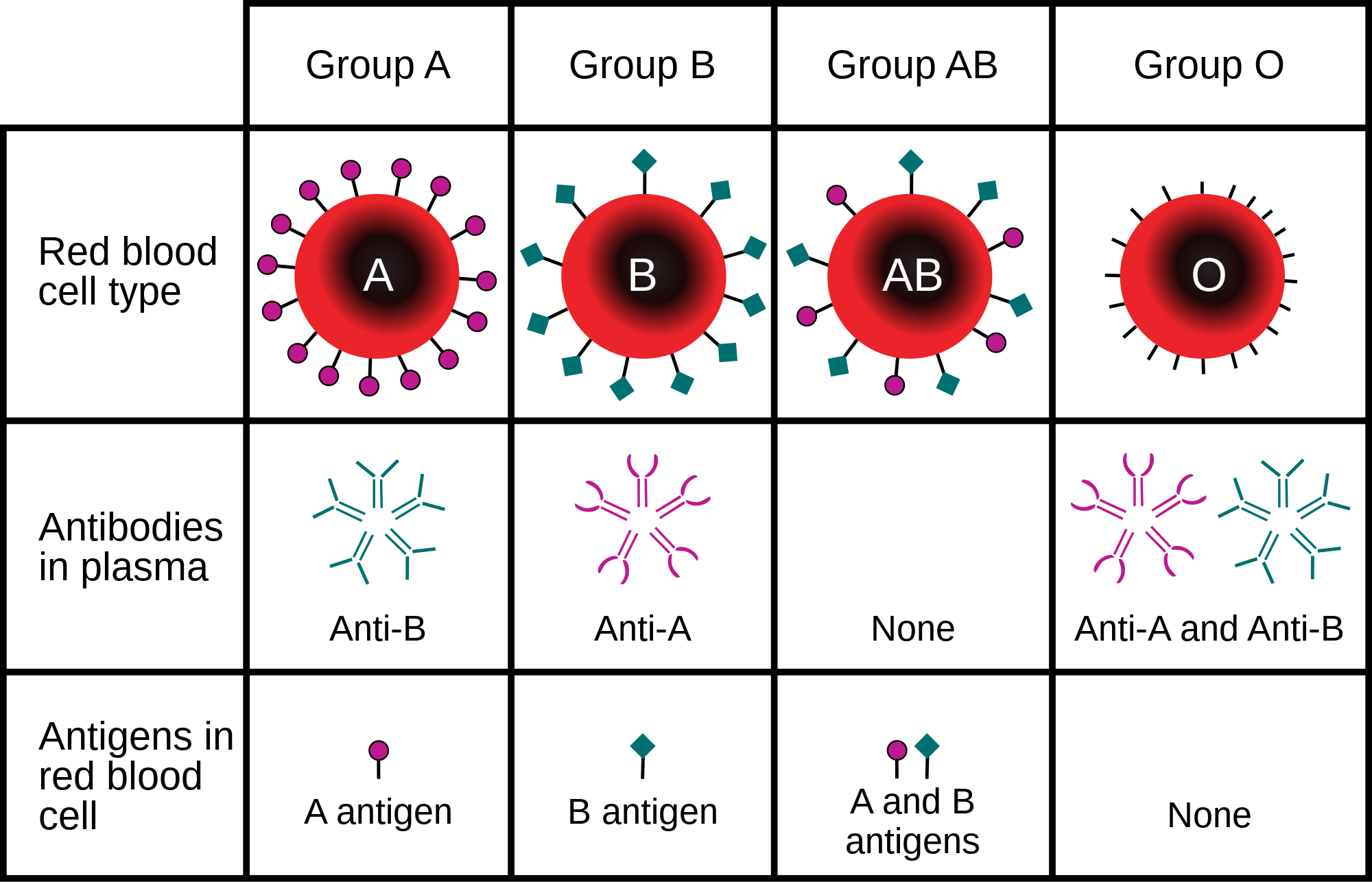
The ABO blood group system is used to denote the presence of one, both, or neither of the A and B antigens on erythrocytes (red blood cells). For human blood transfusions, it is the most important of the 48 different blood type (or group) classification systems currently recognized by the International Society of Blood Transfusions (ISBT) as of June 2025. A mismatch in this serotype (or in various others) can cause a potentially fatal adverse reaction after a transfusion, or an unwanted immune response to an organ transplant. Such mismatches are rare in modern medicine. The associated anti-A and anti-B antibodies are usually IgM antibodies, produced in the first years of life by sensitization to environmental substances such as food, bacteria, and viruses.
The ABO blood types were discovered by Karl Landsteiner in 1901; he received the Nobel Prize in Physiology or Medicine in 1930 for this discovery. ABO blood types are also present in other primates such as apes, monkeys and Old World monkeys.
The ABO blood types were first discovered by an Austrian physician, Karl Landsteiner, working at the Pathological-Anatomical Institute of the University of Vienna (now Medical University of Vienna). In 1900, he found that red blood cells would clump together (agglutinate) when mixed in test tubes with sera from different persons, and that some human blood also agglutinated with animal blood. He wrote a two-sentence footnote:
The serum of healthy human beings not only agglutinates animal red cells, but also often those of human origin, from other individuals. It remains to be seen whether this appearance is related to inborn differences between individuals or it is the result of some damage of bacterial kind.
This was the first evidence that blood variations exist in humans — it was believed that all humans have similar blood. The next year, in 1901, he made a definitive observation that blood serum of an individual would agglutinate with only those of certain individuals. Based on this he classified human blood into three groups, namely group A, group B, and group C. He defined that group A blood agglutinates with group B, but never with its own type. Similarly, group B blood agglutinates with group A. Group C blood is different in that it agglutinates with both A and B.
This was the discovery of blood groups for which Landsteiner was awarded the Nobel Prize in Physiology or Medicine in 1930. In his paper, he referred to the specific blood group interactions as isoagglutination, and also introduced the concept of agglutinins (antibodies), which is the actual basis of antigen-antibody reaction in the ABO system. He asserted:
[It] may be said that there exist at least two different types of agglutinins, one in A, another one in B, and both together in C. The red blood cells are inert to the agglutinins which are present in the same serum.
Thus, he discovered two antigens (agglutinogens A and B) and two antibodies (agglutinins — anti-A and anti-B). His third group (C) indicated absence of both A and B antigens, but contains anti-A and anti-B. The following year, his students Adriano Sturli and Alfred von Decastello discovered the fourth type (but not naming it, and simply referred to it as "no particular type").
ABO blood group system
The ABO blood group system is used to denote the presence of one, both, or neither of the A and B antigens on erythrocytes (red blood cells). For human blood transfusions, it is the most important of the 48 different blood type (or group) classification systems currently recognized by the International Society of Blood Transfusions (ISBT) as of June 2025. A mismatch in this serotype (or in various others) can cause a potentially fatal adverse reaction after a transfusion, or an unwanted immune response to an organ transplant. Such mismatches are rare in modern medicine. The associated anti-A and anti-B antibodies are usually IgM antibodies, produced in the first years of life by sensitization to environmental substances such as food, bacteria, and viruses.
The ABO blood types were discovered by Karl Landsteiner in 1901; he received the Nobel Prize in Physiology or Medicine in 1930 for this discovery. ABO blood types are also present in other primates such as apes, monkeys and Old World monkeys.
The ABO blood types were first discovered by an Austrian physician, Karl Landsteiner, working at the Pathological-Anatomical Institute of the University of Vienna (now Medical University of Vienna). In 1900, he found that red blood cells would clump together (agglutinate) when mixed in test tubes with sera from different persons, and that some human blood also agglutinated with animal blood. He wrote a two-sentence footnote:
The serum of healthy human beings not only agglutinates animal red cells, but also often those of human origin, from other individuals. It remains to be seen whether this appearance is related to inborn differences between individuals or it is the result of some damage of bacterial kind.
This was the first evidence that blood variations exist in humans — it was believed that all humans have similar blood. The next year, in 1901, he made a definitive observation that blood serum of an individual would agglutinate with only those of certain individuals. Based on this he classified human blood into three groups, namely group A, group B, and group C. He defined that group A blood agglutinates with group B, but never with its own type. Similarly, group B blood agglutinates with group A. Group C blood is different in that it agglutinates with both A and B.
This was the discovery of blood groups for which Landsteiner was awarded the Nobel Prize in Physiology or Medicine in 1930. In his paper, he referred to the specific blood group interactions as isoagglutination, and also introduced the concept of agglutinins (antibodies), which is the actual basis of antigen-antibody reaction in the ABO system. He asserted:
[It] may be said that there exist at least two different types of agglutinins, one in A, another one in B, and both together in C. The red blood cells are inert to the agglutinins which are present in the same serum.
Thus, he discovered two antigens (agglutinogens A and B) and two antibodies (agglutinins — anti-A and anti-B). His third group (C) indicated absence of both A and B antigens, but contains anti-A and anti-B. The following year, his students Adriano Sturli and Alfred von Decastello discovered the fourth type (but not naming it, and simply referred to it as "no particular type").