Recent from talks
Knowledge base stats:
Talk channels stats:
Members stats:
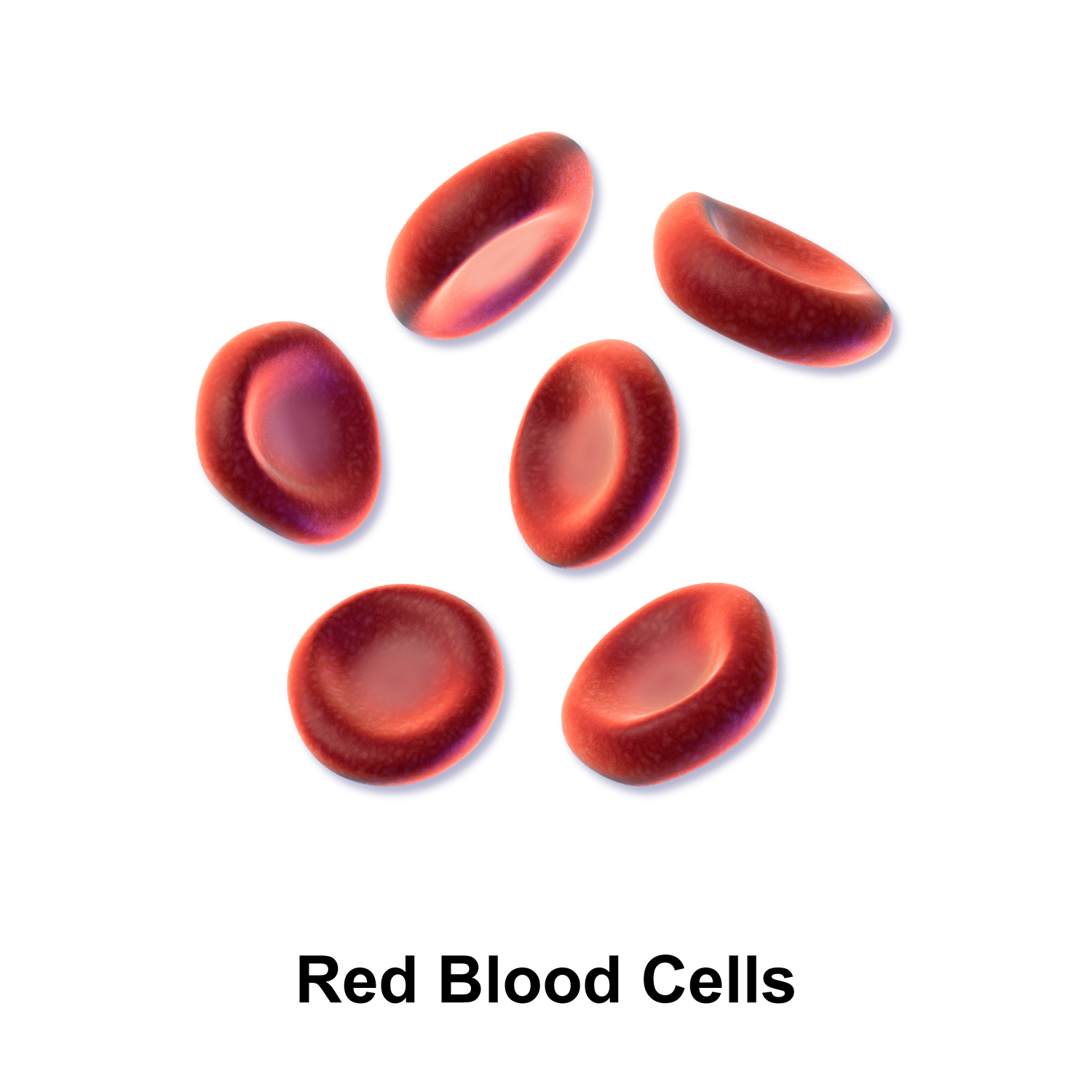
Red blood cell
Red blood cells (RBCs), referred to as erythrocytes (from Ancient Greek erythros 'red' and kytos 'hollow vessel', with -cyte translated as 'cell' in modern usage) in academia and medical publishing, also known as red cells, erythroid cells, and rarely haematids, are the most common type of blood cell and the vertebrate's principal means of delivering oxygen (O2) to the body tissues—via blood flow through the circulatory system. Erythrocytes take up oxygen in the lungs, or in fish the gills, and release it into tissues while squeezing through the body's capillaries.
The cytoplasm of a red blood cell is rich in hemoglobin (Hb), an iron-containing biomolecule that can bind oxygen and is responsible for the red color of the cells and the blood. Each human red blood cell contains approximately 270 million hemoglobin molecules. The cell membrane is composed of proteins and lipids, and this structure provides properties essential for physiological cell function such as deformability and stability of the blood cell while traversing the circulatory system and specifically the capillary network.
In humans, mature red blood cells are flexible biconcave disks. They lack a cell nucleus (which is expelled during development) and organelles, to accommodate maximum space for hemoglobin; they can be viewed as sacks of hemoglobin, with a plasma membrane as the sack. Approximately 2.4 million new erythrocytes are produced per second in human adults. The cells develop in the bone marrow and circulate for about 100–120 days in the body before their components are recycled by macrophages. Each circulation takes about 60 seconds (one minute). Approximately 84% of the cells in the human body are the 20–30 trillion red blood cells. Nearly half of the blood's volume (40% to 45%) is red blood cells.
Packed red blood cells are red blood cells that have been donated, processed, and stored in a blood bank for blood transfusion.
The vast majority of vertebrates, including mammals and humans, have red blood cells. These erythrocytes are cells present in blood to transport oxygen. The only known vertebrates without red blood cells are the crocodile icefish (family Channichthyidae); they live in very oxygen-rich cold water and transport oxygen freely dissolved in their blood. While they no longer use hemoglobin, remnants of hemoglobin genes can be found in their genome.
Vertebrate red blood cells consist mainly of hemoglobin, a complex metalloprotein containing heme groups whose iron atoms temporarily bind to oxygen molecules (O2) in the lungs or gills and release them throughout the body. Oxygen can easily diffuse through the erythrocyte's cell membrane. Hemoglobin in the red blood cells also carries some of the waste product carbon dioxide back from the tissues; most waste carbon dioxide, however, is transported back to the pulmonary capillaries of the lungs as bicarbonate (HCO3−) dissolved in the blood plasma. Myoglobin, a compound related to hemoglobin, acts to store oxygen in muscle cells.
The color of red blood cells is due to the heme group of hemoglobin. The blood plasma alone is straw-colored, but the red blood cells change color depending on the state of the hemoglobin: when combined with oxygen the resulting oxyhemoglobin is scarlet, and when oxygen has been released the resulting deoxyhemoglobin is of a dark red burgundy color. However, blood can appear bluish when seen through the vessel wall and skin. Pulse oximetry takes advantage of the hemoglobin color change to directly measure the arterial blood oxygen saturation using colorimetric techniques. Hemoglobin also has a very high affinity for carbon monoxide, forming carboxyhemoglobin which is a very bright red in color. Flushed, confused patients with a saturation reading of 100% on pulse oximetry are sometimes found to be suffering from carbon monoxide poisoning.[citation needed]
Having oxygen-carrying proteins inside specialized cells (as opposed to oxygen carriers being dissolved in body fluid) was an important step in the evolution of vertebrates as it allows for less viscous blood, higher concentrations of oxygen, and better diffusion of oxygen from the blood to the tissues. The size of red blood cells varies widely among vertebrate species; red blood cell width is on average about 25% larger than capillary diameter, and it has been hypothesized that this improves the oxygen transfer from red blood cells to tissues.
Hub AI
Red blood cell AI simulator
(@Red blood cell_simulator)
Red blood cell
Red blood cells (RBCs), referred to as erythrocytes (from Ancient Greek erythros 'red' and kytos 'hollow vessel', with -cyte translated as 'cell' in modern usage) in academia and medical publishing, also known as red cells, erythroid cells, and rarely haematids, are the most common type of blood cell and the vertebrate's principal means of delivering oxygen (O2) to the body tissues—via blood flow through the circulatory system. Erythrocytes take up oxygen in the lungs, or in fish the gills, and release it into tissues while squeezing through the body's capillaries.
The cytoplasm of a red blood cell is rich in hemoglobin (Hb), an iron-containing biomolecule that can bind oxygen and is responsible for the red color of the cells and the blood. Each human red blood cell contains approximately 270 million hemoglobin molecules. The cell membrane is composed of proteins and lipids, and this structure provides properties essential for physiological cell function such as deformability and stability of the blood cell while traversing the circulatory system and specifically the capillary network.
In humans, mature red blood cells are flexible biconcave disks. They lack a cell nucleus (which is expelled during development) and organelles, to accommodate maximum space for hemoglobin; they can be viewed as sacks of hemoglobin, with a plasma membrane as the sack. Approximately 2.4 million new erythrocytes are produced per second in human adults. The cells develop in the bone marrow and circulate for about 100–120 days in the body before their components are recycled by macrophages. Each circulation takes about 60 seconds (one minute). Approximately 84% of the cells in the human body are the 20–30 trillion red blood cells. Nearly half of the blood's volume (40% to 45%) is red blood cells.
Packed red blood cells are red blood cells that have been donated, processed, and stored in a blood bank for blood transfusion.
The vast majority of vertebrates, including mammals and humans, have red blood cells. These erythrocytes are cells present in blood to transport oxygen. The only known vertebrates without red blood cells are the crocodile icefish (family Channichthyidae); they live in very oxygen-rich cold water and transport oxygen freely dissolved in their blood. While they no longer use hemoglobin, remnants of hemoglobin genes can be found in their genome.
Vertebrate red blood cells consist mainly of hemoglobin, a complex metalloprotein containing heme groups whose iron atoms temporarily bind to oxygen molecules (O2) in the lungs or gills and release them throughout the body. Oxygen can easily diffuse through the erythrocyte's cell membrane. Hemoglobin in the red blood cells also carries some of the waste product carbon dioxide back from the tissues; most waste carbon dioxide, however, is transported back to the pulmonary capillaries of the lungs as bicarbonate (HCO3−) dissolved in the blood plasma. Myoglobin, a compound related to hemoglobin, acts to store oxygen in muscle cells.
The color of red blood cells is due to the heme group of hemoglobin. The blood plasma alone is straw-colored, but the red blood cells change color depending on the state of the hemoglobin: when combined with oxygen the resulting oxyhemoglobin is scarlet, and when oxygen has been released the resulting deoxyhemoglobin is of a dark red burgundy color. However, blood can appear bluish when seen through the vessel wall and skin. Pulse oximetry takes advantage of the hemoglobin color change to directly measure the arterial blood oxygen saturation using colorimetric techniques. Hemoglobin also has a very high affinity for carbon monoxide, forming carboxyhemoglobin which is a very bright red in color. Flushed, confused patients with a saturation reading of 100% on pulse oximetry are sometimes found to be suffering from carbon monoxide poisoning.[citation needed]
Having oxygen-carrying proteins inside specialized cells (as opposed to oxygen carriers being dissolved in body fluid) was an important step in the evolution of vertebrates as it allows for less viscous blood, higher concentrations of oxygen, and better diffusion of oxygen from the blood to the tissues. The size of red blood cells varies widely among vertebrate species; red blood cell width is on average about 25% larger than capillary diameter, and it has been hypothesized that this improves the oxygen transfer from red blood cells to tissues.