Recent from talks
Nothing was collected or created yet.
Transplant rejection
View on Wikipedia| Transplant rejection | |
|---|---|
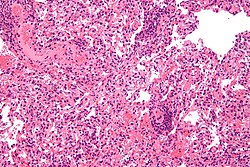 | |
| Micrograph showing lung transplant rejection. Lung biopsy. H&E stain. | |
| Specialty | Immunology |
| Treatment | Immunosuppressive drugs |
Transplant rejection occurs when transplanted tissue is rejected by the recipient's immune system, which destroys the transplanted tissue. Transplant rejection can be lessened by determining the molecular similitude between donor and recipient and by use of immunosuppressant drugs after transplant.[1]
Types
[edit]Transplant rejection can be classified into three types: hyperacute, acute, and chronic.[2] These types are differentiated by how quickly the recipient's immune system is activated and the specific aspect or aspects of immunity involved.[3]
Hyperacute rejection
[edit]Hyperacute rejection is a form of rejection that manifests itself in the minutes to hours following transplantation.[4] It is caused by the presence of pre-existing antibodies in the recipient that recognize antigens in the donor organ.[5] These antigens are located on the endothelial lining of blood vessels within the transplanted organ and, once antibodies bind, will lead to the rapid activation of the complement system.[6] Irreversible damage via thrombosis and subsequent graft necrosis is to be expected.[7] Tissue left implanted will fail to work and could lead to high fever and malaise as the immune system acts against foreign tissue.[8]
Graft failure secondary to hyperacute rejection has significantly decreased in incidence as a result of improved pre-transplant screening for antibodies to donor tissues.[4] While these preformed antibodies may result from prior transplants, prior blood transfusions, or pregnancy, hyperacute rejection is most commonly from antibodies to ABO blood group antigens.[6] Consequently, transplants between individuals with differing ABO blood types is generally avoided though may be pursued in very young children (generally under 12 months, but often as old as 24 months)[9] who do not have fully developed immune systems.[10] Shortages of organs and the morbidity and mortality associated with being on transplant waitlists has also increased interest in ABO-incompatible transplantation in older children and adults.[11]
Acute rejection
[edit]Acute rejection is a category of rejection that occurs on the timescale of weeks to months, with most episodes occurring within the first 3 months to 1 year after transplantation.[6][8] Unlike hyperacute rejection, acute rejection is thought to arise from two distinct immunological mechanisms as lymphocytes, a subset of white blood cells, begin to recognize antigens on transplanted organ/graft.[12] This recognition occurs due to the major histocompatibility complex (MHC), which are proteins on cell surface that are presented to the T-cell receptor found on T-cells.[13] In humans, this is known as the human leukocyte antigen (HLA) system[13] and over 17,000 HLA alleles or genetic variants have been described such that it is extremely uncommon for any two people to have identical alleles.[14] Other non-HLA proteins, known as minor histocompatibility antigens, do exist but generally are unable to cause acute rejection in and of themselves unless a multitude of non-HLA proteins are mismatched.[15] As such, HLA matching (in addition to matching ABO groups) is critical in preventing acute rejection.[16]
This process of recognition by T-cells can happen directly or indirectly and lead to acute cellular and acute humoral rejection respectively.[6] Direct allorecognition is a phenomenon within transplant immunology where the dendritic cells, which are the body's antigen-presenting cells (APCs), migrate from donor tissue to lymphoid tissue (lymphoid follicles and lymph nodes) in the recipient and present their MHC peptides to recipient lymphocytes.[17] In comparison, indirect allorecognition is more analogous to how foreign antigens are recognized by the immune system.[18] Dendritic cells of the recipient come across peptides from donor tissue whether in circulation, lymphoid tissue, or in donor tissue itself.[18] Since not the result of direct antigen presentation, these may not necessarily be intact MHC molecules but instead other proteins that are deemed different enough from recipient may engender a response.[18] This process leads to the priming of T-cells to respond to the peptides secondarily going forward.[2] A third semi-direct pathway has been described in which recipient APCs present fully intact donor MHCs,[17] yet its relative contribution to acute rejection is not as well understood.[15]
Acute cellular rejection occurs following direct allorecognition of mismatched donor MHC by cytotoxic T-cells that begin to secrete cytokines to recruit more lymphocytes as well as cause apoptosis or cell death directly.[4][6] The greater the difference in MHC between donor and recipient, the more cytotoxic T-cells are recruited to damage the graft,[6] which may be seen via biopsy in solid organ transplants, with increased lymphocyte infiltration indicative of more severe acute cellular rejection.[15] Acute humoral rejection is a process usually initiated by indirect allorecognition arising from recipient helper T-cells.[6] These helper T-cells have a crucial role in the development of B-cells that can create donor-specific antibodies.[4] The antibodies deposit themselves within the donor graft and lead to activation of the complement cascade alongside antibody-mediated cytotoxicity with neutrophils, a type of white blood cell separate from lymphocytes, predominantly infiltrating into tissues.[6]
Barring genetically identical twins, acute rejection is to be expected to some degree.[16] Rates of clinically significant acute rejection that could endanger transplant have decreased significantly with the development of immunosuppressive regimens. Using kidney transplants as an example, rates of acute rejection have declined from >50% in the 1970s to 10-20%.[19] Singular episodes of acute rejection, when promptly treated, should not compromise transplant; however, repeated episodes may lead to chronic rejection.[16]
Chronic rejection
[edit]
Chronic rejection is an insidious form of rejection that leads to graft destruction over months to years after tissue transplantation.[12] The mechanism for chronic rejection is yet to be fully understood, but it is known that prior acute rejection episodes are the main clinical predictor for the development of chronic rejection.[6] In particular, the incidence increases following severe or persistent acute rejection, whereas acute rejection episodes with return to function back to baseline do not have major effects on graft survival.[20][21] Chronic rejection is generally thought of as being related to either vascular damage or parenchymal damage with subsequent fibrosis.[22] While it is unknown the exact contribution of the immune system in these processes, the indirect pathway of allorecognition and the associated antibody formation seems to be especially involved.[6]
Chronic rejection has widely varied effects on different organs. At 5 years post-transplant, 80% of lung transplants, 60% of heart transplants and 50% of kidney transplants are affected, while liver transplants are only affected 10% of the time.[20] Therefore, chronic rejection explains long-term morbidity in most lung-transplant recipients,[23][24] the median survival roughly 4.7 years, about half the span versus other major organ transplants.[25] Airflow obstruction not ascribable to other cause is labeled bronchiolitis obliterans syndrome (BOS), confirmed by a persistent drop—three or more weeks—in forced expiratory volume (FEV1) by at least 20%.[26] First noted is infiltration by lymphocytes, followed by epithelial cell injury, then inflammatory lesions and recruitment of fibroblasts and myofibroblasts, which proliferate and secrete proteins forming scar tissue.[27] A similar phenomenon can be seen with liver transplant wherein fibrosis leads to jaundice secondary to the destruction of bile ducts within the liver, also known as vanishing bile duct syndrome.[28]
Rejection due to non-adherence
[edit]One principal reason for transplant rejection is non-adherence to prescribed immunosuppressant regimens. This is particularly the case with adolescent recipients,[29] with non-adherence rates near 50% in some instances.[29]
A pilot study conducted by Michael O. Killian PhD from Florida State University and Dr. Dipankar Gupta from University of Florida published in April 2022 in Pediatric Transplantation [30] studied the acceptability and feasibility of an asynchronous directly observed therapy mobile health application among adolescent heart transplant recipients. Patients in the study utilized emocha Health's digital medication adherence program which included asynchronous video messages and chat messages exchanged with a care team. Patients completing the study achieved a 90.1% adherence rate. The researchers noted that further randomized trials are required to confirm the initial findings. However, the results were very promising considering few options exist to support pediatric patients in taking their medications.[citation needed]
Rejection detection
[edit]Diagnosis of acute rejection relies on clinical data—patient signs and symptoms but also calls on laboratory data such as blood or even tissue biopsy. The laboratory pathologist generally seeks three main histological signs: (1) infiltrating T cells, perhaps accompanied by infiltrating eosinophils, plasma cells, and neutrophils, particularly in telltale ratios, (2) structural compromise of tissue anatomy, varying by tissue type transplanted, and (3) injury to blood vessels. Tissue biopsy is restricted, however, by sampling limitations and risks/complications of the invasive procedure.[31][32][33] Cellular magnetic resonance imaging (MRI) of immune cells radiolabeled in vivo might—similarly to Gene Expression Profiling (GEP)—offer noninvasive testing.[34][35]
Rejection treatment
[edit]This section needs additional citations for verification. (June 2020) |
Hyperacute rejection manifests severely and within minutes, and so treatment is immediate: removal of the tissue. Acute rejection is treated with one or several of a few strategies. Despite treatment, rejection remains a major cause of transplant failure.[36] Chronic rejection is generally considered irreversible and poorly amenable to treatment—only retransplant generally indicated if feasible—though inhaled ciclosporin is being investigated to delay or prevent chronic rejection of lung transplants.
Immunosuppressive therapy
[edit]A short course of high-dose corticosteroids can be applied, and repeated. Triple therapy adds a calcineurin inhibitor and an anti-proliferative agent. Where calcineurin inhibitors or steroids are contraindicated, mTOR inhibitors are used.
Immunosuppressive drugs:
- Corticosteroids
- Calcineurin inhibitors
- Anti-proliferatives
- mTOR inhibitors
Antibody-based treatments
[edit]Antibody specific to select immune components can be added to immunosuppressive therapy. The monoclonal anti-T cell antibody OKT3, once used to prevent rejection, and still occasionally used to treat severe acute rejection, has fallen into disfavor, as it commonly brings severe cytokine release syndrome and late post-transplant lymphoproliferative disorder. (OKT3 is available in the United Kingdom for named-patient use only.)
Antibody drugs:
- Monoclonal anti-IL-2Rα receptor antibodies
- Monoclonal anti-IL-6R receptor antibodies
- Polyclonal anti-T-cell antibodies
- Anti-thymocyte globulin (ATG)
- Anti-lymphocyte globulin (ALG)
- Monoclonal anti-CD20 antibodies
Blood transfer
[edit]Cases refractory to immunosuppressive or antibody therapy are sometimes treated with photopheresis, or extracorporeal photoimmune therapy (ECP), to remove antibody molecules specific to the transplanted tissue.
Marrow transplant
[edit]Bone marrow transplant can replace the transplant recipient's immune system with the donor's, and the recipient accepts the new organ without rejection. The marrow's hematopoietic stem cells—the reservoir of stem cells replenishing exhausted blood cells including white blood cells forming the immune system—must be of the individual who donated the organ or of an identical twin or a clone. There is a risk of graft-versus-host disease (GVHD), however, whereby mature lymphocytes entering with marrow recognize the new host tissues as foreign and destroy them.
Gene therapy
[edit]Gene therapy is another method that can be used. In this method, the genes that cause the body to reject transplants would be deactivated. Research is still being conducted, and no gene therapies are being used to date to treat patients.[37][38][39] Current research tends to focus[citation needed] on Th1 and Th17 which mediate allograft rejection via the CD4 and CD8 T cells.[40]
See also
[edit]References
[edit]- ^ Frohn C, Fricke L, Puchta JC, Kirchner H (February 2001). "The effect of HLA-C matching on acute renal transplant rejection". Nephrology, Dialysis, Transplantation. 16 (2): 355–360. doi:10.1093/ndt/16.2.355. PMID 11158412.
- ^ a b Moreau A, Varey E, Anegon I, Cuturi MC (November 2013). "Effector mechanisms of rejection". Cold Spring Harbor Perspectives in Medicine. 3 (11) a015461. doi:10.1101/cshperspect.a015461. PMC 3808773. PMID 24186491.
- ^ Moreau A, Varey E, Anegon I, Cuturi MC (November 2013). "Effector mechanisms of rejection". Cold Spring Harbor Perspectives in Medicine. 3 (11) a015461. doi:10.1101/cshperspect.a015461. PMC 3808773. PMID 24186491.
- ^ a b c d Chang YC (2021-01-01). "The surgical and immunosuppressive basis for infections in the pediatric solid organ transplant recipient". In Steinbach WJ, Green MD, Michaels MG, Danziger-Isakov LA (eds.). Pediatric Transplant and Oncology Infectious Diseases. Philadelphia: Elsevier. pp. 1–9.e3. doi:10.1016/B978-0-323-64198-2.00010-5. ISBN 978-0-323-64198-2. S2CID 228907807.
- ^ Harmon WE (2010-01-01). "Chapter 41 - Pediatric Renal Transplantation". In Himmelfarb J, Sayegh MH (eds.). Chronic Kidney Disease, Dialysis, and Transplantation (Third ed.). Philadelphia: W.B. Saunders. pp. 591–608. doi:10.1016/B978-1-4377-0987-2.00041-8. ISBN 978-1-4377-0987-2.
- ^ a b c d e f g h i j Mak TW, Saunders ME, Jett BD, eds. (2014-01-01). "Chapter 17 - Transplantation". Primer to the Immune Response (Second ed.). Boston: Academic Cell. pp. 457–486. doi:10.1016/B978-0-12-385245-8.00017-0. ISBN 978-0-12-385245-8.
- ^ Dharnidharka VR (2019-01-01). "43 - Pediatric Renal Transplantation". In Himmelfarb J, Ikizler TA (eds.). Chronic Kidney Disease, Dialysis, and Transplantation (Fourth ed.). Philadelphia: Elsevier. pp. 661–675.e7. doi:10.1016/B978-0-323-52978-5.00043-4. ISBN 978-0-323-52978-5. S2CID 81475473.
- ^ a b Vitak K (2014-01-01). "Chapter 14 - Organ Transplantation". In Paz JC, West MP (eds.). Acute Care Handbook for Physical Therapists (Fourth ed.). St. Louis: W.B. Saunders. pp. 335–356. doi:10.1016/B978-1-4557-2896-1.00014-7. ISBN 978-1-4557-2896-1.
- ^ West LJ, Pollock-Barziv SM, Dipchand AI, Lee KJ, Cardella CJ, Benson LN, Rebeyka IM, Coles JG (March 2001). "ABO-incompatible heart transplantation in infants". The New England Journal of Medicine. 344 (11): 793–800. doi:10.1056/NEJM200103153441102. PMID 11248154.
- ^ West LJ (October 2011). "ABO-incompatible hearts for infant transplantation". Current Opinion in Organ Transplantation. 16 (5): 548–554. doi:10.1097/MOT.0b013e32834a97a5. PMID 21836514. S2CID 26070409.
- ^ Subramanian V, Ramachandran S, Klein C, Wellen JR, Shenoy S, Chapman WC, Mohanakumar T (August 2012). "ABO-incompatible organ transplantation". International Journal of Immunogenetics. 39 (4): 282–290. doi:10.1111/j.1744-313x.2012.01101.x. PMID 22339811. S2CID 41947505.
- ^ a b Justiz Vaillant AA, Mohseni M (2022). "Chronic Transplantation Rejection". StatPearls. Treasure Island (FL): StatPearls Publishing. PMID 30571056. Retrieved 2022-03-16.
- ^ a b Delves PJ. "Human Leukocyte Antigen (HLA) System - Immunology; Allergic Disorders". Merck Manuals Professional Edition. Merck & Co. Retrieved 30 September 2020.
- ^ Frenet EM, Scaradavou A (2019-01-01). "Chapter 32 - Human Leukocyte Antigens". In Shaz BH, Hillyer CD, Gil MR (eds.). Transfusion Medicine and Hemostasis (PDF) (Third ed.). Elsevier. pp. 191–197. doi:10.1016/B978-0-12-813726-0.00032-5. ISBN 978-0-12-813726-0. S2CID 91582896.
- ^ a b c Menon MC, Cravedi P, El Salem F (2017-01-01). "Chapter 32 - Acute Cellular Rejection". In Orlando G, Remuzzi G, Williams DF (eds.). Kidney Transplantation, Bioengineering and Regeneration. Academic Press. pp. 461–474. doi:10.1016/B978-0-12-801734-0.00032-1. ISBN 978-0-12-801734-0.
- ^ a b c Justiz Vaillant AA, Misra S, Fitzgerald BM (2022). "Acute Transplantation Rejection". StatPearls. Treasure Island (FL): StatPearls Publishing. PMID 30571031. Retrieved 2022-03-16.
- ^ a b Boardman DA, Jacob J, Smyth LA, Lombardi G, Lechler RI (2016). "What Is Direct Allorecognition?". Current Transplantation Reports. 3 (4): 275–283. doi:10.1007/s40472-016-0115-8. PMC 5107184. PMID 27909647.
- ^ a b c Ingulli E (January 2010). "Mechanism of cellular rejection in transplantation". Pediatric Nephrology. 25 (1): 61–74. doi:10.1007/s00467-008-1020-x. PMC 2778785. PMID 21476231.
- ^ Clayton PA, McDonald SP, Russ GR, Chadban SJ (September 2019). "Long-Term Outcomes after Acute Rejection in Kidney Transplant Recipients: An ANZDATA Analysis". Journal of the American Society of Nephrology. 30 (9): 1697–1707. doi:10.1681/ASN.2018111101. PMC 6727270. PMID 31308074.
- ^ a b Gautreaux MD (2017-01-01). "Chapter 17 - Histocompatibility Testing in the Transplant Setting". In Orlando G, Remuzzi G, Williams DF (eds.). Kidney Transplantation, Bioengineering and Regeneration. Academic Press. pp. 223–234. doi:10.1016/b978-0-12-801734-0.00017-5. ISBN 978-0-12-801734-0.
- ^ Srinivas TR, Schold JD, Meier-Kriesche HU (2010-01-01). "Chapter 105 - Outcomes of Renal Transplantation". In Floege J, Johnson RJ, Feehally J (eds.). Comprehensive Clinical Nephrology (Fourth ed.). Philadelphia: Mosby. pp. 1222–1231. doi:10.1016/b978-0-323-05876-6.00105-2. ISBN 978-0-323-05876-6.
- ^ Dharnidharka VR (2019-01-01). "Chapter 43 - Pediatric Renal Transplantation". In Himmelfarb J, Ikizler TA (eds.). Chronic Kidney Disease, Dialysis, and Transplantation (Fourth ed.). Philadelphia: Elsevier. pp. 661–675.e7. doi:10.1016/B978-0-323-52978-5.00043-4. ISBN 978-0-323-52978-5. S2CID 81475473.
- ^ Jaramillo A, Fernández FG, Kuo EY, Trulock EP, Patterson GA, Mohanakumar T (February 2005). "Immune mechanisms in the pathogenesis of bronchiolitis obliterans syndrome after lung transplantation". Pediatric Transplantation. 9 (1): 84–93. doi:10.1111/j.1399-3046.2004.00270.x. PMID 15667618. S2CID 25841425.
- ^ Lau CL, Patterson GA (November 2003). "Current status of lung transplantation". The European Respiratory Journal. Supplement. 47: 57s – 64s. doi:10.1183/09031936.03.00022103. PMID 14621118.
- ^ "Organ Procurement and Transplantation Network". U.S. Department of Health & Human Services.
- ^ Lama VN, Murray S, Lonigro RJ, Toews GB, Chang A, Lau C, et al. (June 2007). "Course of FEV(1) after onset of bronchiolitis obliterans syndrome in lung transplant recipients". American Journal of Respiratory and Critical Care Medicine. 175 (11): 1192–1198. doi:10.1164/rccm.200609-1344OC. PMC 1899272. PMID 17347496.
- ^ Nicod LP (July 2006). "Mechanisms of airway obliteration after lung transplantation". Proceedings of the American Thoracic Society. 3 (5): 444–449. doi:10.1513/pats.200601-007AW. PMID 16799090.
- ^ Hübscher SG, Clouston AD (January 2012). "Chapter 15 - Transplantation pathology". In Burt AD, Portmann BC, Ferrell LD (eds.). MacSween's Pathology of the Liver (Sixth ed.). Edinburgh: Churchill Livingstone. pp. 853–933. doi:10.1016/B978-0-7020-3398-8.00015-5. ISBN 978-0-7020-3398-8.
- ^ a b Dobbels F, Hames A, Aujoulat I, Heaton N, Samyn M (February 2012). "Should we retransplant a patient who is non-adherent? A literature review and critical reflection". Pediatric Transplantation. 16 (1): 4–11. doi:10.1111/j.1399-3046.2011.01633.x. PMID 22248250. S2CID 1895827.
- ^ Killian, Michael O.; Clifford, Stephanie; Lustria, Mia Liza A.; Skivington, Gage L.; Gupta, Dipankar (2022-04-18). "Directly observed therapy to promote medication adherence in adolescent heart transplant recipients". Pediatric Transplantation. 26 (5) e14288. doi:10.1111/petr.14288. ISSN 1397-3142. PMID 35436376. S2CID 248242427.
- ^ "Kidney biopsy:risks". Johns Hopkins Medicine. The Johns Hopkins University.
- ^ Clinical trial number NCT00351559 for "IMAGE: A Comparison of AlloMap Molecular Testing and Traditional Biopsy-based Surveillance for Heart Transplant Rejection" at ClinicalTrials.gov
- ^ "Lung biopsy:risks". Johns Hopkins Medicine. The Johns Hopkins University. 8 August 2021.
- ^ Hitchens TK, Ye Q, Eytan DF, Janjic JM, Ahrens ET, Ho C (April 2011). "19F MRI detection of acute allograft rejection with in vivo perfluorocarbon labeling of immune cells". Magnetic Resonance in Medicine. 65 (4): 1144–1153. doi:10.1002/mrm.22702. PMC 3135171. PMID 21305593.
- ^ Gheith OA (2011). "Gene expression profiling in organ transplantation". International Journal of Nephrology. 2011 180201. doi:10.4061/2011/180201. PMC 3154482. PMID 21845224.
- ^ Naesens M, Kuypers DR, De Vusser K, Evenepoel P, Claes K, Bammens B, et al. (August 2014). "The histology of kidney transplant failure: a long-term follow-up study". Transplantation. 98 (4): 427–435. doi:10.1097/TP.0000000000000183. PMID 25243513. S2CID 20703626.
- ^ Yang JY, Sarwal MM (May 2017). "Transplant genetics and genomics". Nature Reviews. Genetics. 18 (5): 309–326. doi:10.1038/nrg.2017.12. PMID 28286337. S2CID 2222755.
- ^ Bagley J, Iacomini J (April 2003). "Gene therapy progress and prospects: gene therapy in organ transplantation". Gene Therapy. 10 (8): 605–611. doi:10.1038/sj.gt.3302020. PMID 12692588.
- ^ Giannoukakis N, Thomson A, Robbins P (September 1999). "Gene therapy in transplantation". Gene Therapy. 6 (9): 1499–1511. doi:10.1038/sj.gt.3300981. PMID 10490759.
- ^ Yuan X, Paez-Cortez J, Schmitt-Knosalla I, D'Addio F, Mfarrej B, Donnarumma M, et al. (December 2008). "A novel role of CD4 Th17 cells in mediating cardiac allograft rejection and vasculopathy". The Journal of Experimental Medicine. 205 (13): 3133–3144. doi:10.1084/jem.20081937. PMC 2605226. PMID 19047438.

