Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Vosilasarm AI simulator
(@Vosilasarm_simulator)
Hub AI
Vosilasarm AI simulator
(@Vosilasarm_simulator)
Vosilasarm
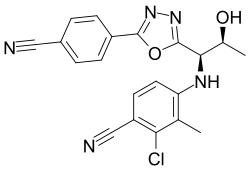
Vosilasarm, also known by the development codes RAD140 and EP0062 and by the black-market name Testolone or Testalone, is a selective androgen receptor modulator (SARM) which is under development for the treatment of hormone-sensitive breast cancer. It is specifically under development for the treatment of androgen receptor-positive, estrogen receptor-negative, HER2-negative advanced breast cancer. Vosilasarm was also previously under development for the treatment of sarcopenia (age-related muscle atrophy), osteoporosis, and weight loss due to cancer cachexia, but development for these indications was discontinued. The drug is taken by mouth.
Side effects of vosilasarm may include vomiting, dehydration, constipation, decreased appetite, weight loss, changes in sex hormone levels, elevated liver enzymes, and liver toxicity. Vosilasarm is a nonsteroidal SARM, acting as an agonist of the androgen receptor (AR), the biological target of androgens and anabolic steroids like testosterone and dihydrotestosterone (DHT). However, it shows dissociation of effect between tissues in preclinical studies, with agonistic and anabolic effects in muscle, agonistic effects in breast, and partially agonistic or antagonistic effects in the prostate gland and seminal vesicles.
Vosilasarm was developed in 2010 and was first described in the literature in 2011. It was originally developed by Radius Health and is now under development by Ellipses Pharma. The first clinical study of vosilasarm, a small (n=22) phase 1 study in women with metastatic breast cancer, was started in 2017 and completed in 2020, with results published in 2019, 2020, and 2022. As of March 2023, vosilasarm is in phase 1/2 clinical trials for the treatment of breast cancer.
Aside from its development as a potential pharmaceutical drug, vosilasarm is on the World Anti-Doping Agency list of prohibited substances and is sold for physique- and performance-enhancing purposes by black-market Internet suppliers. Vosilasarm is often used in these contexts at doses that have not been evaluated in clinical trials, with unknown effectiveness and safety. Many products sold online that are purported to be a specific SARM either contain none or contain other unrelated substances. Social media has played an important role in facilitating the widespread non-medical use of SARMs.
Vosilasarm is not approved for any medical use and is not available as a licensed pharmaceutical drug as of 2023.
Side effects of vosilasarm in preliminary clinical studies in women with metastatic breast cancer have included vomiting (27%), dehydration (27%), constipation, decreased appetite and weight loss (27%), hypophosphatemia, decreased sex hormone-binding globulin (SHBG) levels (100%), increased prostate-specific antigen (PSA) levels (80%), and abnormal liver function tests, including elevated aspartate aminotransferase (59%), elevated alanine aminotransferase (46%), and elevated total blood bilirubin (27%).
In gonadally intact male cynomolgus monkeys, vosilasarm suppressed testosterone levels by around 50% (from ~600–800 ng/dL to ~200–300 ng/dL) across three dose levels (0.01 mg/kg, 0.1 mg/kg, and 1.0 mg/kg). Changes in serum lipids, including of triglycerides, LDL cholesterol, and HDL cholesterol were also observed. Elevated liver enzymes were minimally observed in monkeys.
A number of case reports of liver toxicity with non-medical use of vosilasarm have been published. A case report of acute myocarditis with non-medical use of vosilasarm also exists.
Vosilasarm
Vosilasarm, also known by the development codes RAD140 and EP0062 and by the black-market name Testolone or Testalone, is a selective androgen receptor modulator (SARM) which is under development for the treatment of hormone-sensitive breast cancer. It is specifically under development for the treatment of androgen receptor-positive, estrogen receptor-negative, HER2-negative advanced breast cancer. Vosilasarm was also previously under development for the treatment of sarcopenia (age-related muscle atrophy), osteoporosis, and weight loss due to cancer cachexia, but development for these indications was discontinued. The drug is taken by mouth.
Side effects of vosilasarm may include vomiting, dehydration, constipation, decreased appetite, weight loss, changes in sex hormone levels, elevated liver enzymes, and liver toxicity. Vosilasarm is a nonsteroidal SARM, acting as an agonist of the androgen receptor (AR), the biological target of androgens and anabolic steroids like testosterone and dihydrotestosterone (DHT). However, it shows dissociation of effect between tissues in preclinical studies, with agonistic and anabolic effects in muscle, agonistic effects in breast, and partially agonistic or antagonistic effects in the prostate gland and seminal vesicles.
Vosilasarm was developed in 2010 and was first described in the literature in 2011. It was originally developed by Radius Health and is now under development by Ellipses Pharma. The first clinical study of vosilasarm, a small (n=22) phase 1 study in women with metastatic breast cancer, was started in 2017 and completed in 2020, with results published in 2019, 2020, and 2022. As of March 2023, vosilasarm is in phase 1/2 clinical trials for the treatment of breast cancer.
Aside from its development as a potential pharmaceutical drug, vosilasarm is on the World Anti-Doping Agency list of prohibited substances and is sold for physique- and performance-enhancing purposes by black-market Internet suppliers. Vosilasarm is often used in these contexts at doses that have not been evaluated in clinical trials, with unknown effectiveness and safety. Many products sold online that are purported to be a specific SARM either contain none or contain other unrelated substances. Social media has played an important role in facilitating the widespread non-medical use of SARMs.
Vosilasarm is not approved for any medical use and is not available as a licensed pharmaceutical drug as of 2023.
Side effects of vosilasarm in preliminary clinical studies in women with metastatic breast cancer have included vomiting (27%), dehydration (27%), constipation, decreased appetite and weight loss (27%), hypophosphatemia, decreased sex hormone-binding globulin (SHBG) levels (100%), increased prostate-specific antigen (PSA) levels (80%), and abnormal liver function tests, including elevated aspartate aminotransferase (59%), elevated alanine aminotransferase (46%), and elevated total blood bilirubin (27%).
In gonadally intact male cynomolgus monkeys, vosilasarm suppressed testosterone levels by around 50% (from ~600–800 ng/dL to ~200–300 ng/dL) across three dose levels (0.01 mg/kg, 0.1 mg/kg, and 1.0 mg/kg). Changes in serum lipids, including of triglycerides, LDL cholesterol, and HDL cholesterol were also observed. Elevated liver enzymes were minimally observed in monkeys.
A number of case reports of liver toxicity with non-medical use of vosilasarm have been published. A case report of acute myocarditis with non-medical use of vosilasarm also exists.