Recent from talks
Knowledge base stats:
Talk channels stats:
Members stats:
YWHAZ
14-3-3 protein zeta/delta (14-3-3ζ) is a protein that in humans is encoded by the YWHAZ gene on chromosome 8. The protein encoded by this gene is a member of the 14-3-3 protein family and a central hub protein for many signal transduction pathways. 14-3-3ζ is a major regulator of apoptotic pathways critical to cell survival and plays a key role in a number of cancers and neurodegenerative diseases.
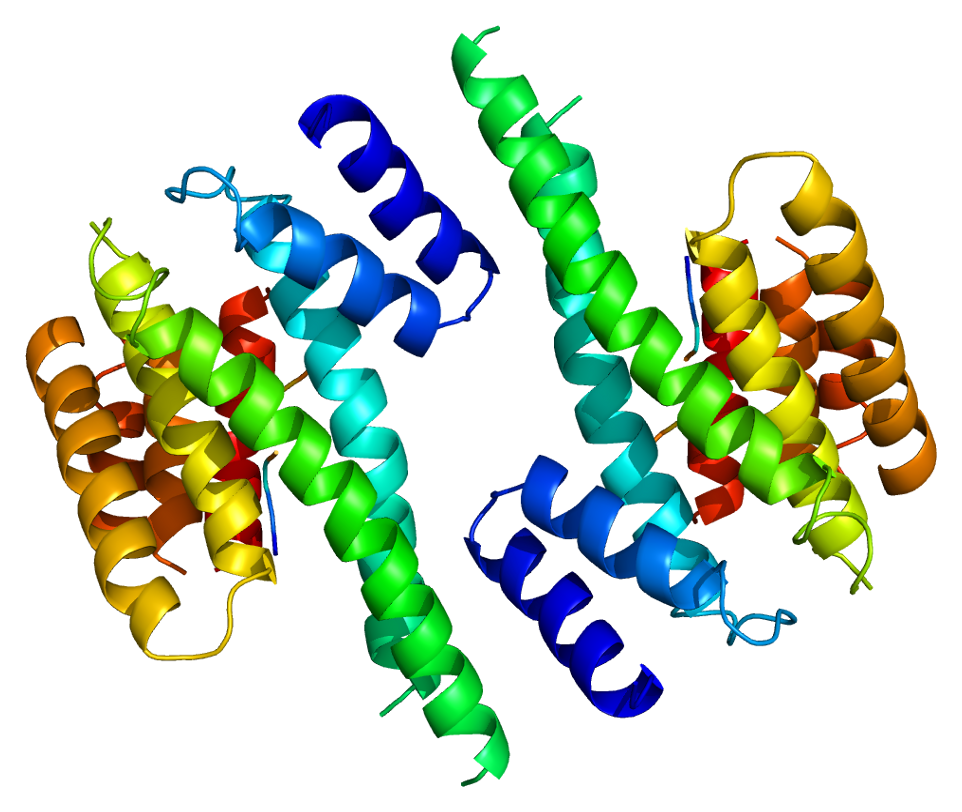
14-3-3 proteins generally form ~30 kDa-long homo- or heterodimers. Each of the monomers are composed of 9 antiparallel alpha helices. Four alpha-helices (αC, αE, αG, and αI) form an amphipathic groove that serves as the ligand binding site, which can recognize three types of consensus binding motifs: RXX(pS/pT)XP, RXXX(pS/pT)XP, and (pS/pT)X1-2-COOH (where pS/pT represents phosphorylated serine/threonine). In addition to these primary interactions, the target protein can also bind outside the groove via secondary interactions. In particular, the crystallized structure of 14-3-3ζ forms a cup-shaped dimer when complexed with CBY. The YWHAZ gene encodes two transcript variants which differ in the 5' UTR but produce the same protein.
14-3-3ζ is one of 7 members of the 14-3-3 protein family, which is ubiquitously expressed and highly conserved among plants and mammals. This protein family is known for regulating signal transduction pathways primarily through binding phosphoserine proteins, though it can also bind phosphothreonine proteins and unphosphorylated proteins. By extension, 14-3-3 proteins are involved in a wide range of biological processes, including metabolism, transcription, apoptosis, protein transport, and cell cycle regulation. This combination of dependence on phosphorylation and widespread biological impact results in dynamic regulation of multiple signalling pathways and allows for cellular adaptation to environmental changes.
In particular, 14-3-3ζ is a key player in regulating cell survival and interacts with many apoptotic proteins, including Raf kinases, BAX, BAD, NOXA, and caspase-2. For the most part, 14-3-3ζ negatively regulates apoptosis by binding and sequestering BAD and BAX in the cytoplasm, effectively preventing activation of proapoptotic Bcl-2 and Bcl-XL, as well as by preventing NOXA from inhibiting antiapoptotic MCL1. As a result, 14-3-3ζ functions to protect the cell from environmental stresses, such as chemotherapy-induced death, anoikis, growth factor deprivation, and hypoxia. As an example of its dynamic activity, 14-3-3ζ activates autophagy under hypoxic conditions by binding ATG9A, while it prevents autophagy under hyperglycemic conditions by binding Vps34. Furthermore, 14-3-3ζ may regulate glucose receptor trafficking in response to insulin levels through its interaction with IRS1.
In addition to cell survival, 14-3-3ζ regulates cell cycle progression through various ligands and processes. For instance, 14-3-3ζ controls cellular senescence by complexing with BIS to chaperone protein folding of STAT3 and activate the signaling pathway. Also, 14-3-3ζ can negatively regulate the G2-M phase checkpoint by binding and sequestering the cyclin-dependent kinases to the cytoplasm, thus inhibiting their activity. Since 14-3-3ζ is predominantly found in the cytoplasm and binds many nuclear proteins, it likely prevents nuclear import by blocking the nuclear localization signal of target proteins. Its localization to both the cytoplasm and nucleus also suggests a role in gene expression, possibly through regulation of transcription factor activity.
Emerging literature shows the increased presence of the anti-14-3-3ζ antibodies in several immune dysfunctions, including human vasculitis and cancer. The antigenic 14-3-3ζ can directly affect T cell differentiation into Th1 and Th17 cells, and thereby promotes IFN-gamma and IL-17 production. The MHC class II presentation of 14-3-3ζ antigen strongly influence IFN-gamma production. The physiological significance of its antigenic role remains unknown
Intracellular 14-3-3ζ plays a role in interleukin-17 signaling. IL-17A is a proinflammatory cytokine involved in autoimmune diseases and host defense. The presence of 14-3-3ζ creates a bias in IL-17A signaling outcomes, by promoting the production of IL-6 while suppressing CXCL1.
The14-3-3 protein zeta/delta (14-3-3ζ) is a protein (in humans encoded by the YWHAZ gene on chromosome 8) with an important apoptotic constituents. During a normal embryologic processes, or during cell injury (such as ischemia-reperfusion injury during heart attacks and strokes) or during developments and processes in cancer, an apoptotic cell undergoes structural changes including cell shrinkage, plasma membrane blebbing, nuclear condensation, and fragmentation of the DNA and nucleus. This is followed by fragmentation into apoptotic bodies that are quickly removed by phagocytes, thereby preventing an inflammatory response. It is a mode of cell death defined by characteristic morphological, biochemical and molecular changes. It was first described as a "shrinkage necrosis", and then this term was replaced by apoptosis to emphasize its role opposite mitosis in tissue kinetics. In later stages of apoptosis the entire cell becomes fragmented, forming a number of plasma membrane-bounded apoptotic bodies which contain nuclear and or cytoplasmic elements. The ultrastructural appearance of necrosis is quite different, the main features being mitochondrial swelling, plasma membrane breakdown and cellular disintegration. Apoptosis occurs in many physiological and pathological processes. It plays an important role during embryonal development as programmed cell death and accompanies a variety of normal involutional processes in which it serves as a mechanism to remove "unwanted" cells.
Hub AI
YWHAZ AI simulator
(@YWHAZ_simulator)
YWHAZ
14-3-3 protein zeta/delta (14-3-3ζ) is a protein that in humans is encoded by the YWHAZ gene on chromosome 8. The protein encoded by this gene is a member of the 14-3-3 protein family and a central hub protein for many signal transduction pathways. 14-3-3ζ is a major regulator of apoptotic pathways critical to cell survival and plays a key role in a number of cancers and neurodegenerative diseases.
14-3-3 proteins generally form ~30 kDa-long homo- or heterodimers. Each of the monomers are composed of 9 antiparallel alpha helices. Four alpha-helices (αC, αE, αG, and αI) form an amphipathic groove that serves as the ligand binding site, which can recognize three types of consensus binding motifs: RXX(pS/pT)XP, RXXX(pS/pT)XP, and (pS/pT)X1-2-COOH (where pS/pT represents phosphorylated serine/threonine). In addition to these primary interactions, the target protein can also bind outside the groove via secondary interactions. In particular, the crystallized structure of 14-3-3ζ forms a cup-shaped dimer when complexed with CBY. The YWHAZ gene encodes two transcript variants which differ in the 5' UTR but produce the same protein.
14-3-3ζ is one of 7 members of the 14-3-3 protein family, which is ubiquitously expressed and highly conserved among plants and mammals. This protein family is known for regulating signal transduction pathways primarily through binding phosphoserine proteins, though it can also bind phosphothreonine proteins and unphosphorylated proteins. By extension, 14-3-3 proteins are involved in a wide range of biological processes, including metabolism, transcription, apoptosis, protein transport, and cell cycle regulation. This combination of dependence on phosphorylation and widespread biological impact results in dynamic regulation of multiple signalling pathways and allows for cellular adaptation to environmental changes.
In particular, 14-3-3ζ is a key player in regulating cell survival and interacts with many apoptotic proteins, including Raf kinases, BAX, BAD, NOXA, and caspase-2. For the most part, 14-3-3ζ negatively regulates apoptosis by binding and sequestering BAD and BAX in the cytoplasm, effectively preventing activation of proapoptotic Bcl-2 and Bcl-XL, as well as by preventing NOXA from inhibiting antiapoptotic MCL1. As a result, 14-3-3ζ functions to protect the cell from environmental stresses, such as chemotherapy-induced death, anoikis, growth factor deprivation, and hypoxia. As an example of its dynamic activity, 14-3-3ζ activates autophagy under hypoxic conditions by binding ATG9A, while it prevents autophagy under hyperglycemic conditions by binding Vps34. Furthermore, 14-3-3ζ may regulate glucose receptor trafficking in response to insulin levels through its interaction with IRS1.
In addition to cell survival, 14-3-3ζ regulates cell cycle progression through various ligands and processes. For instance, 14-3-3ζ controls cellular senescence by complexing with BIS to chaperone protein folding of STAT3 and activate the signaling pathway. Also, 14-3-3ζ can negatively regulate the G2-M phase checkpoint by binding and sequestering the cyclin-dependent kinases to the cytoplasm, thus inhibiting their activity. Since 14-3-3ζ is predominantly found in the cytoplasm and binds many nuclear proteins, it likely prevents nuclear import by blocking the nuclear localization signal of target proteins. Its localization to both the cytoplasm and nucleus also suggests a role in gene expression, possibly through regulation of transcription factor activity.
Emerging literature shows the increased presence of the anti-14-3-3ζ antibodies in several immune dysfunctions, including human vasculitis and cancer. The antigenic 14-3-3ζ can directly affect T cell differentiation into Th1 and Th17 cells, and thereby promotes IFN-gamma and IL-17 production. The MHC class II presentation of 14-3-3ζ antigen strongly influence IFN-gamma production. The physiological significance of its antigenic role remains unknown
Intracellular 14-3-3ζ plays a role in interleukin-17 signaling. IL-17A is a proinflammatory cytokine involved in autoimmune diseases and host defense. The presence of 14-3-3ζ creates a bias in IL-17A signaling outcomes, by promoting the production of IL-6 while suppressing CXCL1.
The14-3-3 protein zeta/delta (14-3-3ζ) is a protein (in humans encoded by the YWHAZ gene on chromosome 8) with an important apoptotic constituents. During a normal embryologic processes, or during cell injury (such as ischemia-reperfusion injury during heart attacks and strokes) or during developments and processes in cancer, an apoptotic cell undergoes structural changes including cell shrinkage, plasma membrane blebbing, nuclear condensation, and fragmentation of the DNA and nucleus. This is followed by fragmentation into apoptotic bodies that are quickly removed by phagocytes, thereby preventing an inflammatory response. It is a mode of cell death defined by characteristic morphological, biochemical and molecular changes. It was first described as a "shrinkage necrosis", and then this term was replaced by apoptosis to emphasize its role opposite mitosis in tissue kinetics. In later stages of apoptosis the entire cell becomes fragmented, forming a number of plasma membrane-bounded apoptotic bodies which contain nuclear and or cytoplasmic elements. The ultrastructural appearance of necrosis is quite different, the main features being mitochondrial swelling, plasma membrane breakdown and cellular disintegration. Apoptosis occurs in many physiological and pathological processes. It plays an important role during embryonal development as programmed cell death and accompanies a variety of normal involutional processes in which it serves as a mechanism to remove "unwanted" cells.