Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
2-Naphthol AI simulator
(@2-Naphthol_simulator)
Hub AI
2-Naphthol AI simulator
(@2-Naphthol_simulator)
2-Naphthol
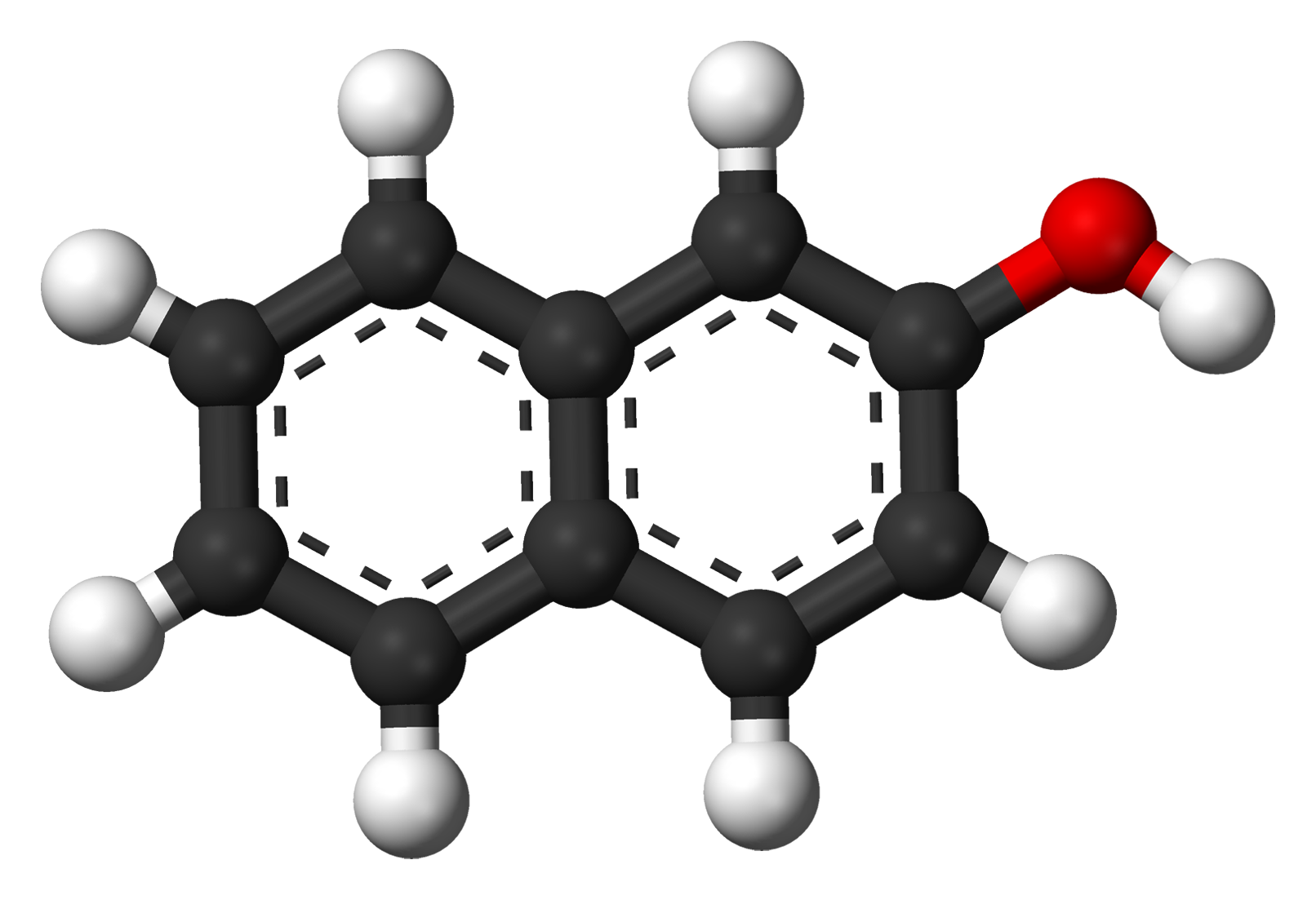
2-Naphthol, or β-naphthol, is a fluorescent colorless (or occasionally yellow) crystalline solid with the formula C10H7OH. It is an isomer of 1-naphthol, differing by the location of the hydroxyl group on the naphthalene ring. The naphthols are naphthalene homologues of phenol, but more reactive. Both isomers are soluble in simple alcohols, ethers, and chloroform. 2-Naphthol is a widely used intermediate for the production of dyes and other compounds.
Traditionally, 2-naphthol is produced by a two-step process that begins with the sulfonation of naphthalene in sulfuric acid:
The sulfonic acid group is then cleaved in molten sodium hydroxide:
Neutralization of the product with acid gives 2-naphthol.
2-Naphthol can also be produced by a method analogous to the cumene process.
The Sudan dyes are popular dyes noted for being soluble in organic solvents. Several of the Sudan dyes are derived from 2-naphthol by coupling with diazonium salts. Sudan dyes I–IV and Sudan Red G consist of arylazo-substituted naphthols.
Some reactions of 2-naphthol are explicable with reference to its tautomerism, which produces a small amount of the keto tautomer.
One consequence of this tautomerism is the Bucherer reaction, the ammonolysis of 2-naphthol to give 2-aminonaphthalene.
2-Naphthol
2-Naphthol, or β-naphthol, is a fluorescent colorless (or occasionally yellow) crystalline solid with the formula C10H7OH. It is an isomer of 1-naphthol, differing by the location of the hydroxyl group on the naphthalene ring. The naphthols are naphthalene homologues of phenol, but more reactive. Both isomers are soluble in simple alcohols, ethers, and chloroform. 2-Naphthol is a widely used intermediate for the production of dyes and other compounds.
Traditionally, 2-naphthol is produced by a two-step process that begins with the sulfonation of naphthalene in sulfuric acid:
The sulfonic acid group is then cleaved in molten sodium hydroxide:
Neutralization of the product with acid gives 2-naphthol.
2-Naphthol can also be produced by a method analogous to the cumene process.
The Sudan dyes are popular dyes noted for being soluble in organic solvents. Several of the Sudan dyes are derived from 2-naphthol by coupling with diazonium salts. Sudan dyes I–IV and Sudan Red G consist of arylazo-substituted naphthols.
Some reactions of 2-naphthol are explicable with reference to its tautomerism, which produces a small amount of the keto tautomer.
One consequence of this tautomerism is the Bucherer reaction, the ammonolysis of 2-naphthol to give 2-aminonaphthalene.