Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
2C-Ph AI simulator
(@2C-Ph_simulator)
Hub AI
2C-Ph AI simulator
(@2C-Ph_simulator)
2C-Ph
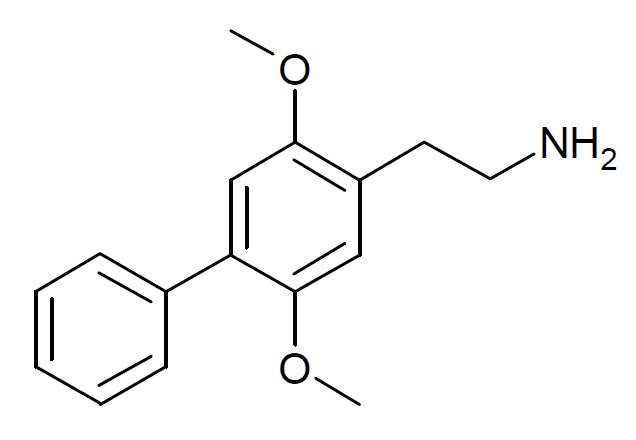
2C-Ph, or 2C-Phenyl, also known as 2C-BI-1 or as 2,5-dimethoxy-4-phenylphenethylamine, is a serotonin receptor modulator of the phenethylamine and 2C families that was developed by Daniel Trachsel and David E. Nichols and colleagues.
The drug's affinity (Ki) for the rat serotonin 5-HT2A receptor was 778 nM. It was said to be an antagonist of this receptor. In a subsequent study, 2C-Ph was a weak partial agonist of the human serotonin 5-HT2A receptor (Ki = 630 nM, EC50 = 1,596 nM, Emax = 23%). The drug also shows affinity for the serotonin 5-HT1A, 5-HT2B, and 5-HT2C receptors, but did not activate the serotonin 5-HT2B receptor. In addition, it interacted with other monoamine receptors, with the monoamine transporters, and was a potent and high-efficacy partial agonist of the human trace amine-associated receptor 1 (TAAR1) (EC50 = 580 nM, Emax = 82%).
Besides 2C-Ph itself, a variety of derivatives of 2C-Ph with substituents on the 4-position phenyl ring have been synthesized and studied by Trachsel and colleagues. These drugs, inclusive of 2C-Ph, have been denoted 2C-BI-1 to 2C-BI-12. 2C-BI-4 (the 2′-trifluoromethyl derivative), 2C-BI-8 (the 4′-methoxy derivative), and 2C-BI-12 (the 3′,4′-dimethoxy derivative) are agonists of the human serotonin 5-HT2A receptor with higher efficacy than 2C-Ph (EC50 = 37–2,408 nM, Emax = 38–44%). The effects of 2C-Ph and its derivatives in humans are unknown. However, 2C-BI-8 and 2C-BI-12, the most potent agonists, in particular might have the potential for psychedelic effects.
2C-Ph was first described in the scientific literature, by Trachsel and Nichols and colleagues, in 2009.
2C-Ph
2C-Ph, or 2C-Phenyl, also known as 2C-BI-1 or as 2,5-dimethoxy-4-phenylphenethylamine, is a serotonin receptor modulator of the phenethylamine and 2C families that was developed by Daniel Trachsel and David E. Nichols and colleagues.
The drug's affinity (Ki) for the rat serotonin 5-HT2A receptor was 778 nM. It was said to be an antagonist of this receptor. In a subsequent study, 2C-Ph was a weak partial agonist of the human serotonin 5-HT2A receptor (Ki = 630 nM, EC50 = 1,596 nM, Emax = 23%). The drug also shows affinity for the serotonin 5-HT1A, 5-HT2B, and 5-HT2C receptors, but did not activate the serotonin 5-HT2B receptor. In addition, it interacted with other monoamine receptors, with the monoamine transporters, and was a potent and high-efficacy partial agonist of the human trace amine-associated receptor 1 (TAAR1) (EC50 = 580 nM, Emax = 82%).
Besides 2C-Ph itself, a variety of derivatives of 2C-Ph with substituents on the 4-position phenyl ring have been synthesized and studied by Trachsel and colleagues. These drugs, inclusive of 2C-Ph, have been denoted 2C-BI-1 to 2C-BI-12. 2C-BI-4 (the 2′-trifluoromethyl derivative), 2C-BI-8 (the 4′-methoxy derivative), and 2C-BI-12 (the 3′,4′-dimethoxy derivative) are agonists of the human serotonin 5-HT2A receptor with higher efficacy than 2C-Ph (EC50 = 37–2,408 nM, Emax = 38–44%). The effects of 2C-Ph and its derivatives in humans are unknown. However, 2C-BI-8 and 2C-BI-12, the most potent agonists, in particular might have the potential for psychedelic effects.
2C-Ph was first described in the scientific literature, by Trachsel and Nichols and colleagues, in 2009.