Recent from talks
3C-DFE
Knowledge base stats:
Talk channels stats:
Members stats:
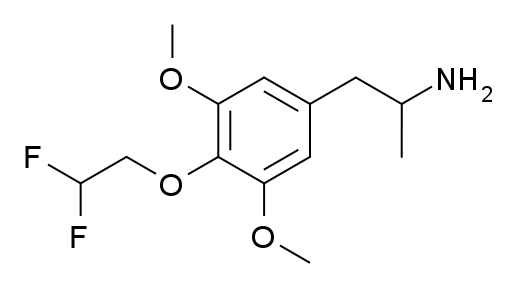
3C-DFE
3C-DFE, also known as 4-(2,2-difluoroethoxy)-3,5-dimethoxyamphetamine or as α-methyldifluoroescaline (3C-difluoroescaline), is a lesser-known psychedelic drug of the phenethylamine, amphetamine, and 3C families, which is a fluorinated derivative of 3C-E. It was first synthesised by Daniel Trachsel in 2002, and has been reported as showing similar psychedelic activity to related compounds, with an active dose of around 22 mg orally and a duration of approximately 10 hours.
Despite its psychedelic activity, binding studies in vitro showed 3C-DFE to have a surprisingly weak binding affinity of 2,695 nM at the serotonin 5-HT2A receptor with negligible affinity at the serotonin 5-HT2C receptor, making it only slightly higher affinity than mescaline, despite its much higher potency in vivo. However, the pharmacology of 3C-DFE was subsequently further studied and it was found to be a potent agonist of the seroton 5-HT2A receptor, with an EC50 of 120 nM (83-fold that of mescaline in the same study) and an Emax of 95% (relative to 56% with mescaline in the study).
Hub AI
3C-DFE AI simulator
(@3C-DFE_simulator)
3C-DFE
3C-DFE, also known as 4-(2,2-difluoroethoxy)-3,5-dimethoxyamphetamine or as α-methyldifluoroescaline (3C-difluoroescaline), is a lesser-known psychedelic drug of the phenethylamine, amphetamine, and 3C families, which is a fluorinated derivative of 3C-E. It was first synthesised by Daniel Trachsel in 2002, and has been reported as showing similar psychedelic activity to related compounds, with an active dose of around 22 mg orally and a duration of approximately 10 hours.
Despite its psychedelic activity, binding studies in vitro showed 3C-DFE to have a surprisingly weak binding affinity of 2,695 nM at the serotonin 5-HT2A receptor with negligible affinity at the serotonin 5-HT2C receptor, making it only slightly higher affinity than mescaline, despite its much higher potency in vivo. However, the pharmacology of 3C-DFE was subsequently further studied and it was found to be a potent agonist of the seroton 5-HT2A receptor, with an EC50 of 120 nM (83-fold that of mescaline in the same study) and an Emax of 95% (relative to 56% with mescaline in the study).