Recent from talks
All channels
Be the first to start a discussion here.
Be the first to start a discussion here.
Be the first to start a discussion here.
Be the first to start a discussion here.
Welcome to the community hub built to collect knowledge and have discussions related to Blue nevus.
Nothing was collected or created yet.
Blue nevus
View on Wikipediafrom Wikipedia
| Blue nevus | |
|---|---|
| Other names | Blue neuronevus, dermal melanocytoma, nevus coeruleus, nevus bleu[1] |
 | |
| Blue nevus | |
| Specialty | Dermatology |
| Symptoms | Single well-defined blue-black bump[2] |
| Complications | Rarely malignant transformation[3] |
| Types | Dendritic, cellular[2] |
| Causes | Unclear[3] |
| Diagnostic method | Visualisation, dermoscopy[4] |
| Differential diagnosis | Dermatofibroma, melanoma[3][5] |
| Treatment | Monitoring, excision[3] |
| Prognosis | Good[3] |
| Frequency | Female>male[2] |
A blue nevus is a type of coloured mole, typically a single well-defined blue-black bump.[1][2]
The blue colour is caused by the pigment being deep in the skin.[4]
Diagnosis is by visualisation and dermoscopy.[4] A biopsy is sometimes performed, or the whole lesion surgically removed.[3] The outcome is generally good but there is a small chance of cancerous transformation.[3] Differential diagnosis includes dermatofibroma and melanoma.[3]
Blue nevi are more common in females than males.[2] It was first studied in 1906 by Tièche, a student of Josef Jadassohn.[6]
Classification
[edit]Blue nevi may be divided into the following types:[7]: 701
- A patch blue nevus (also known as an "acquired dermal melanocytosis", and "dermal melanocyte hamartoma") is a cutaneous condition characterized by a diffusely gray-blue area that may have superimposed darker macules.[1]
- A blue nevus of Jadassohn–Tièche (also known as a "common blue nevus", and "nevus ceruleus") is a cutaneous condition characterized by a steel-blue papule or nodule.[7]: 701
- A cellular blue nevus is a cutaneous condition characterized by large, firm, blue or blue-black nodules.[7]: 701
- An epithelioid blue nevus is a cutaneous condition most commonly seen in patients with the Carney complex.[7]: 701
- A deep penetrating nevus is a type of benign melanocytic skin tumor characterized, as its name suggests, by penetration into the deep dermis and/or subcutis. Smudged chromatic is a typical finding. In some cases mitotic figures or atypical melanocytic cytology are seen, potentially mimicking a malignant melanoma. Evaluation by an expert skin pathologist is advisable in some cases to help differentiate from invasive melanoma.[7]: 701
- An amelanotic blue nevus (also known as a "hypomelanotic blue nevus") is a cutaneous condition characterized by mild atypia and pleomorphism.[7]: 701
- A malignant blue nevus is a cutaneous condition characterized by a sheet-like growth pattern, mitoses, necrosis, and cellular atypia.[1][7]: 701
-
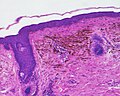
Micrograph of a blue nevus showing the characteristic pigmented melanocytes between bundles of collagen. H&E stain
-
Blue nevus
-
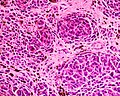
Cellular blue nevus
-
Epithelioid blue nevus
-
Malignant blue nevus
See also
[edit]References
[edit]- ^ a b c d Rapini, Ronald P.; Bolognia, Jean L.; Jorizzo, Joseph L. (2007). Dermatology: 2-Volume Set. St. Louis: Mosby. p. 1722. ISBN 978-1-4160-2999-1.
- ^ a b c d e Johnstone, Ronald B. (2017). "32. Lentigines and melanomas". Weedon's Skin Pathology Essentials (2nd ed.). Elsevier. p. 545. ISBN 978-0-7020-6830-0.
- ^ a b c d e f g h Austad, Steve S.; Athalye, Leela (2021). "Blue Nevus". StatPearls. StatPearls Publishing. PMID 31747181.
- ^ a b c "Blue naevus". dermnetnz.org. Retrieved 21 October 2021.
- ^ Blue+Nevi at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- ^ Sreeremya, S. (17 April 2018). "Blue Nevus". International Journal of Molecular Biotechnology. 4 (1): 1–4. doi:10.37628/ijmb.v4i1.255 (inactive 11 July 2025).
{{cite journal}}: CS1 maint: DOI inactive as of July 2025 (link) - ^ a b c d e f g James, William D.; Berger, Timothy G.; et al. (2006). Andrews' Diseases of the Skin: clinical Dermatology. Saunders Elsevier. ISBN 0-7216-2921-0.
External links
[edit]Blue nevus
View on Grokipediafrom Grokipedia
Clinical Features
Appearance and Location
Blue nevi typically present as well-defined, solitary nodules, papules, or macules with a smooth, firm surface, ranging from 2 to 10 mm in diameter for the common type. The characteristic blue-to-black coloration arises from the Tyndall effect, where deep dermal melanin scatters shorter wavelengths of visible light, preferentially reflecting blue hues while longer wavelengths penetrate deeper. These lesions are usually asymptomatic and stable over time, though confluence of multiple nodules can occasionally occur.[1][3][4] They most commonly appear on the scalp, face, neck, dorsal aspects of the hands and feet, buttocks, and sacrococcygeal region, with solitary lesions predominating in these areas. While the majority are isolated, multiple blue nevi can develop, particularly in eruptive forms or associated with certain syndromes. Eruptive blue nevi are a rare variant characterized by the sudden appearance of multiple blue-gray lesions. Reported triggers and associations include trauma, severe sunburns, oral contraceptives, vesiculobullous dermatoses (blistering disorders), herpes zoster, cutaneous injury, immunosuppression, pregnancy, puberty, tanning-bed use, and in isolated cases, medications like isotretinoin. However, many cases are idiopathic with no identifiable trigger. The distribution reflects the lesion's dermal origin, with no strong predilection for sun-exposed sites.[1][4][5][3][6] Morphological variations exist between subtypes: the common blue nevus is often flat to slightly elevated with a gray-blue hue, whereas the cellular blue nevus tends to be dome-shaped, more raised, and larger, reaching up to 3 cm in size with a deeper blue-black tone. These differences aid in clinical recognition but require histopathological confirmation for precise classification.[4][3][1]Symptoms
Blue nevi are typically asymptomatic, manifesting as stable, unchanging lesions that do not cause pain, itching, or bleeding.[4][1] These benign dermal melanocytic proliferations remain consistent in size and appearance over time, rarely leading to any sensory disturbances.[7] In rare instances, tenderness or irritation may arise if the lesion is situated in pressure-prone areas such as the feet or buttocks, where friction or trauma can affect the skin.[8] No systemic symptoms are associated with isolated blue nevi, though they may occur in the context of rare genetic syndromes like LAMB syndrome (lentigines, atrial myxoma, mucocutaneous myxomas, and blue nevi) or Carney complex.[1][7] Any perceived growth or color change in a blue nevus often prompts medical attention due to concerns over potential malignancy, but these alterations are not inherent symptoms of the lesion itself.[9]Classification
Common Blue Nevus
The common blue nevus, also known as the Jadassohn-Tièche type, is defined as a benign dermal proliferation of dendritic or spindle-shaped melanocytes within a fibrotic stroma.[1][10] It typically measures 1 to 5 mm in diameter and presents as a solitary, well-demarcated, blue-gray papule or nodule that is flat to mildly elevated.[11] This subtype most commonly appears in young adults during the second decade of life, with a peak incidence in individuals under 40 years of age.[1][10] It frequently occurs on the face, scalp, or extremities, though it can arise on other acral sites such as the dorsal hands or feet.[1][11] The characteristic blue-gray coloration results from the Tyndall effect, where deep dermal melanin scatters shorter wavelengths of light.[1] Histologically, the common blue nevus consists of slender, pigmented dendritic melanocytes interspersed with spindle cells and melanophages in a densely sclerotic dermis, forming a wedge-shaped or bulbous pattern without epidermal involvement.[1][10] It is considered entirely benign, exhibiting no malignant potential and remaining stable throughout life unless subjected to trauma, which may rarely lead to pigmentation changes or satellitosis.[1][10]Cellular Blue Nevus
The cellular blue nevus represents a less common subtype of blue nevus distinguished by its increased cellularity and larger size, often exhibiting more prominent growth patterns that can extend deeper into tissues. These lesions typically measure 1-3 cm in diameter and appear as firm, dome-shaped, bluish-gray nodules, most frequently arising on the buttocks or in the sacrococcygeal region. Unlike the smaller, more superficial common blue nevus, the cellular variant shows a biphasic composition with areas of densely packed cells and surrounding pigmented dendritic melanocytes.[13][10] Histologically, cellular blue nevi are characterized by plump epithelioid or spindle-shaped melanocytes with pale cytoplasm, arranged in nests, fascicles, and storiform patterns amid a collagenous stroma, accompanied by melanophages; this contrasts with the lower cellularity of common blue nevi while sharing similar spindle cell morphology. These tumors often demonstrate a wedge-shaped architecture with vertical orientation, infiltrating into the subcutaneous adipose tissue in many cases, which contributes to their deeper dermal and subcuticular extension. The lesions occur primarily in adults, with a peak incidence in those under 40 years (though reported across a broad age range from 6 to 85 years), showing a female predominance at a ratio of approximately 2:1 to 2.2:1.[10][14] Although benign in the vast majority of cases, cellular blue nevi exhibit a higher potential for local recurrence compared to the common variant, particularly following incomplete surgical excision, with rates reported as low but notable (e.g., ~2% in one series of 45 cases); rare persistence or regrowth may occur following incomplete removal. Complete excision with clear margins is recommended to minimize this risk, as the tumors generally follow an indolent course without metastatic potential.[15][16]Other Variants
Epithelioid blue nevus represents a rare subtype of blue nevus characterized by intradermal proliferation of polygonal epithelioid melanocytes heavily laden with melanin; it is considered part of the spectrum of pigmented epithelioid melanocytoma (PEM), encompassing lesions with benign to low-grade malignant potential.[17] These lesions often present as solitary, darkly pigmented, domed papules or nodules less than 1 cm in diameter, primarily on the extremities or trunk, though multiplicity can occur, particularly in syndromic contexts.[18][19] Histologically, they feature poorly circumscribed dermal nests of epithelioid cells with vesicular nuclei, prominent nucleoli, and variable pigmentation, lacking the fibrosis typical of common blue nevi and showing no maturation toward the base.[18] While generally benign with no reported recurrence or metastasis after excision, this variant holds clinical significance due to its strong association with Carney complex, a genetic disorder involving myxomas, spotty pigmentation, and endocrine overactivity, where multiple epithelioid blue nevi may serve as cutaneous markers.[19] Sclerosing blue nevus is an uncommon variant marked by prominent dermal fibrosis surrounding the melanocytic proliferation, potentially simulating melanoma clinically and dermoscopically with features like asymmetric pigmentation, polychromasia, and irregular vessels.[20] Amelanotic blue nevus, comprising about 2.7% of blue nevi, deviates from the typical pigmented presentation by exhibiting minimal or absent melanin, appearing as hypopigmented or skin-colored papules that may mimic dermatofibroma, neurofibroma, or amelanotic melanoma; histologically, it retains spindle-shaped melanocytes in a fibrous stroma with scant pigmentation confirmed by Fontana-Masson stain.[21] A further rare form is the sclerosing mucinous blue nevus, distinguished by abundant mucin deposition in the sclerotic stroma, which can clinically resemble dermatofibroma but requires differentiation from desmoplastic melanoma.[22] Combined blue nevus, often with Spitz nevus components, constitutes a histologic variant where blue nevus elements—such as dendritic or spindle melanocytes—intermingle with epithelioid or spindled cells typical of Spitz lesions, comprising a subset of the approximately 511 reported combined nevi cases.[23] These hybrid lesions frequently appear as pigmented papules or nodules and may show atypia in nearly half of Spitz-involved cases, though long-term follow-up reveals no malignant progression.[23] Blue nevi, particularly epithelioid variants, are syndromically linked to Carney complex (formerly encompassing LAMB syndrome—lentigines, atrial myxoma, blue nevi—and NAME syndrome—nevi, atrial myxoma, myxoid neurofibromas, ephelides), where multiple lesions serve as cutaneous markers alongside cardiac myxomas and endocrine tumors.[24] Congenital blue nevi remain exceptionally rare, often presenting as pauci-melanotic or giant forms with a benign course but potential for diagnostic confusion with melanoma mimics.[25]Pathophysiology
Etiology
Blue nevi are primarily acquired lesions that develop postnatally, often appearing in the second or third decade of life, though their pathogenesis is thought to stem from aberrant migration of neural crest-derived melanocytes during embryogenesis, where these cells become arrested in the dermis instead of reaching the epidermis.[4][1] Genetic alterations play a central role in their development, with somatic hotspot mutations in the GNAQ gene identified in up to 83% of cases, leading to constitutive activation of the MAPK signaling pathway and promoting melanocyte proliferation.[26] Mutations in BRAF are rare in blue nevi, occurring at much lower frequencies compared to other melanocytic nevi.[4] Unlike superficial melanocytic lesions, typical blue nevi show no strong association with environmental triggers such as ultraviolet radiation exposure.[27] However, rare eruptive blue nevi, characterized by the sudden appearance of multiple blue-gray lesions, have been associated with various triggers and conditions including trauma, severe sunburns, oral contraceptives, vesiculobullous dermatoses, herpes zoster, cutaneous injury, immunosuppression, pregnancy, puberty, tanning-bed use, and in isolated cases medications such as isotretinoin, although many cases remain idiopathic.[6][28] Congenital blue nevi are rare and typically occur in the context of syndromic conditions, such as LAMB syndrome (characterized by lentigines, atrial myxomas, blue nevi, and mucocutaneous myxomas), rather than isolated familial inheritance patterns.[24] No established hereditary transmission has been identified for nonsyndromic forms.[4]Histological Characteristics
Blue nevi are characterized histologically by a proliferation of melanocytes confined to the dermis, without any junctional or epidermal involvement. These melanocytes are typically spindle-shaped or dendritic, producing abundant melanin that is distributed within the cells and surrounding melanophages, which are pigment-laden macrophages. The lesion is embedded in a sclerotic, fibrotic stroma that lacks vascularity and inflammatory infiltrate. This deep dermal localization of heavily pigmented cells accounts for the clinical blue appearance through the Tyndall scattering phenomenon, where longer wavelengths of light penetrate deeper while shorter blue wavelengths are scattered back to the observer.[1][29] In the common blue nevus, the histology reveals a relatively superficial, wedge-shaped collection of elongated dendritic melanocytes in the upper to mid-dermis, with fine, dusty melanin pigment finely dispersed throughout the cytoplasm, often obscuring nuclear details. Melanophages are prominent, contributing to the heavy pigmentation, while the surrounding stroma shows collagen bundling and sclerosis without nesting or junctional activity. Benign cases demonstrate no cytologic atypia, such as nuclear pleomorphism, and absence of mitotic figures, distinguishing them from malignant counterparts.[1][30] The cellular blue nevus displays a more biphasic cellular composition, featuring a mix of dendritic melanocytes peripherally and central nests of ovoid to spindle-shaped melanocytes with paler, less pigmented cytoplasm compared to the common type. These lesions often extend deeper into the reticular dermis or subcutis as well-defined, expansile nodules up to several centimeters in size, accompanied by melanophages and a variably sclerotic stroma. Despite the increased cellularity, benign cellular blue nevi lack significant atypia, with mitotic activity typically absent or limited to fewer than 1-2 mitoses per mm², and no evidence of necrosis or infiltrative growth.[1][30][29]Diagnosis
Clinical Assessment
Clinical assessment of a suspected blue nevus begins with a detailed patient history to evaluate the lesion's onset, stability, and potential syndromic associations. Most blue nevi are acquired during adolescence or early adulthood, with congenital presentations being uncommon and often linked to genetic syndromes such as LAMB (lentigines, atrial myxomas, blue nevi, and mucocutaneous myxomas) or Carney complex. Lesions that have remained stable in size, color, and morphology over time are typically benign and require no immediate intervention beyond monitoring. In contrast, a history of rapid growth, color change, or recent onset in adulthood may indicate atypia or malignant transformation. Family history is crucial, as hereditary patterns occur in conditions like Carney complex, where multiple blue nevi may signal increased risk for associated tumors.[1] Visual inspection employs established criteria to identify concerning features without invasive procedures. The ABCDE rule—asymmetry of shape, irregular or notched borders, variegated color (beyond uniform blue), diameter exceeding 6 mm, and evolving characteristics (such as growth or bleeding)—serves as a primary screening tool for melanocytic lesions, including blue nevi, to detect potential melanoma mimics. Complementing this is the "ugly duckling" sign, which highlights any lesion that deviates significantly from an individual's other nevi in size, shape, or color, thereby flagging outliers for closer scrutiny. These methods are particularly useful for blue nevi, which classically appear as small, dome-shaped blue nodules on the scalp, face, or extremities, but any deviation from this symmetric, stable profile warrants heightened suspicion. Combining ABCDE with the ugly duckling sign improves diagnostic sensitivity for atypical lesions.[1][31] Dermoscopy provides a non-invasive magnification of subsurface structures, aiding differentiation from malignant entities like blue nevus-like melanoma. Typical findings in benign blue nevi include homogeneous blue-gray structureless pigmentation due to the Tyndall scattering effect of dermal melanin, lacking pigment networks, globules, or streaks. Variants may show subtle blue globules, dots, or pepper-like gray-blue granules representing melanophages and pigment incontinence. These uniform patterns contrast with melanoma's irregular pigmentation, atypical vascular structures, or polymorphic features, enhancing specificity in clinical decision-making.[32][33]Biopsy and Histopathology
Biopsy is indicated for blue nevi exhibiting atypical clinical features, such as rapid changes in size, shape, or color, asymmetry, diameter greater than 6 mm, or patient concerns, to confirm the diagnosis and exclude malignancy like melanoma.[1] In cellular blue nevi, biopsy is particularly warranted due to their larger size and potential for atypical presentations, ensuring evaluation of margins for completeness.[14] Excisional biopsy is the preferred method, as it allows for complete removal of the lesion and comprehensive histopathological assessment, minimizing the risk of sampling error that can occur with partial techniques.[1] Histopathological examination reveals characteristic features of blue nevi, including dermal aggregates of spindle-shaped or dendritic melanocytes with abundant melanin pigment, accompanied by melanophages and a fibrotic stroma, often with a grenz zone separating the lesion from the epidermis.[14] In cellular variants, the lesion shows increased cellularity with ovoid or epithelioid cells extending into deeper dermal or subcutaneous tissues, but without significant cytologic atypia, high mitotic activity (typically less than 1-2 mitoses per mm²), or necrosis in benign cases.[1] To rule out atypia or malignant transformation, full margins are evaluated to confirm the absence of infiltrative growth or residual tumor cells.[14] Immunohistochemical staining supports the diagnosis by confirming melanocytic origin and distinguishing blue nevi from other spindle cell lesions or melanoma. Blue nevi typically show strong positivity for S-100 protein, HMB-45, and Melan-A (MART-1), with variable expression in deeper portions of the lesion; the presence of these markers without aberrant patterns helps exclude malignancy.[1] In cases with heavy pigmentation, using a red chromogen for immunohistochemistry enhances interpretation by reducing interference from melanin.[14]Treatment
Observation
Observation is the preferred management strategy for stable, asymptomatic blue nevi that display classic clinical features, such as small, dome-shaped, blue-gray nodules less than 1 cm in diameter without recent onset or alteration.[1] This conservative approach is particularly appropriate for common blue nevi, which are typically benign and remain unchanged over time in the majority of cases, thereby preventing unnecessary excision and the potential for scarring.[34][35] Effective monitoring involves regular self-examinations performed monthly to assess for any evolving characteristics, supplemented by annual visits to a dermatologist for professional evaluation.[36] Patients are encouraged to photograph lesions at baseline and periodically thereafter to facilitate comparison and early detection of changes.[37] Key features to track include those outlined by the ABCDE criteria: asymmetry, irregular borders, varied colors, diameter exceeding 6 mm, and any evolution in size, shape, or symptoms.[37] To minimize irritation or potential aggravation, patients should be educated on consistent sun protection practices, including the daily application of broad-spectrum sunscreen with SPF 30 or higher and protective clothing when outdoors.[38] Given their generally benign prognosis, this watchful waiting strategy supports long-term stability without invasive measures.[39]Excision
Excision is the primary surgical intervention for blue nevi when indicated, serving as both a diagnostic and therapeutic measure to ensure complete removal and histopathological confirmation.[1] Indications for excision typically arise from cosmetic concerns, particularly for visible lesions on the face or other prominent areas, or when the nevus causes symptoms such as irritation or pruritus due to its location.[34] Atypical features, including rapid growth, changes in color or size, irregular borders, or ulceration, warrant excision to rule out malignant transformation, especially in cellular blue nevi which carry a slightly higher risk of local aggressiveness.[1] Additionally, cellular blue nevi are often excised prophylactically to prevent potential recurrence, given their deeper dermal involvement and larger size compared to common blue nevi.[34] The procedure involves a full-thickness excisional biopsy under local anesthesia, encompassing the entire lesion to achieve clear margins and avoid transecting the deep dermal melanocytes characteristic of blue nevi.[10] Margins of 2-3 mm are generally sufficient for benign or atypical lesions, ensuring complete removal while minimizing cosmetic defects, though wider margins (up to 1 cm) may be considered for larger or cellular variants to reduce recurrence risk.[10] Shave excision is discouraged due to the risk of incomplete removal, as blue nevi extend deeply into the dermis, potentially leading to persistent pigmentation or false-negative histopathology.[34] Similarly, laser ablation or electrodessication is not recommended, as these methods often fail to eradicate the deep melanocytic component, increasing the likelihood of incomplete treatment and recurrence.[34] Postoperative care focuses on standard wound management to promote healing and minimize complications, including gentle cleansing with mild soap and water once or twice daily, application of a non-adherent dressing, and avoidance of submersion in water until sutures are removed (typically 7-14 days).[1] Scarring is usually minimal, particularly on the face where lesions are common, due to the small size of most excisions and the use of elliptical incisions aligned with skin tension lines.[34] Recurrence is rare following complete excision, though incompletely excised cellular blue nevi may recur with satellite lesions, often presenting at the prior site and necessitating re-excision if confirmed histologically.[40] Regular follow-up is advised to monitor for any changes, though the procedure itself confers a low risk of complications when performed by a dermatologic surgeon.[1]Prognosis
Benign Nature
Blue nevi are benign melanocytic lesions that typically remain stable throughout an individual's lifetime, exhibiting no growth, ulceration, or metastatic potential in the vast majority of cases.[1] This inherent stability ensures that these lesions exert no adverse impact on quality of life, often requiring only periodic clinical monitoring rather than intervention unless changes are observed.[41] The cellular composition of common and cellular blue nevi contributes to this predictable, non-progressive behavior.[1] Surgical excision of blue nevi, when performed, results in a complete cure provided that clear margins are achieved, with no necessity for adjuvant therapies such as radiation or chemotherapy.[1] Recurrence is infrequent following adequate removal, and the procedure is generally straightforward due to the lesion's well-defined borders.[1] Extracutaneous blue nevi, including those arising in the oral mucosa, gastrointestinal tract, or conjunctivae, mirror the benign pattern observed in cutaneous variants, maintaining stability without progression or complications in most instances.[1][42]Risk of Malignancy
The malignant transformation of a blue nevus into melanoma, termed malignant blue nevus or blue nevus-like melanoma, is an exceedingly rare event, with over 100 cases reported in the medical literature as of the 2010s.[1][43][44] This transformation primarily occurs in cellular blue nevi and, less commonly, in common or other variants. The resulting melanoma is aggressive, often presenting with deep invasion and a high propensity for metastasis, particularly to lymph nodes, liver, and lungs.[1][43][44] Clinical warning signs that may indicate malignant change include rapid enlargement of a longstanding lesion, ulceration, development of satellite lesions, and lesions larger than 1 cm in diameter. The risk is further elevated in recurrent lesions, such as those from incompletely excised cellular blue nevi, where residual melanocytic proliferation may harbor atypical cells. These features warrant prompt evaluation, as tumor thickness greater than 5 mm is associated with poorer outcomes, including recurrence (locoregional or distant) in approximately 43% of cases and mortality from disease in 39% in one series.[1][43][44] Upon suspicion of malignancy, management involves wide local excision with clear margins, typically 1-2 cm, to address the deep dermal location of blue nevi. If histopathological confirmation of melanoma is obtained, sentinel lymph node biopsy is recommended for staging, particularly in cases with tumor thickness exceeding 1 mm or ulceration, to assess for regional metastasis. Prognosis remains guarded, with 5-year survival rates reported at approximately 68% in a recent analysis (as of 2024), comparable to conventional melanomas of similar depth but influenced by early detection and complete resection.[1][43][44][45]Epidemiology
Prevalence
Blue nevi occur in approximately 0.5% to 4% of white adults and 3% to 5% of Asian populations, reflecting an overall prevalence of 0.5% to 3% in the general population depending on ethnic background. Overall incidence is estimated at about 1%.[7][3][46] The common blue nevus is the most frequent subtype, significantly more common than the cellular variant (e.g., ratios up to ≈50:1 in some series), as evidenced by histopathological studies where common types constitute the majority of cases.[47] Prevalence shows international variation, with higher rates reported in Asian populations (3-5%) than in Caucasians (1-2%), potentially linked to fair-skinned individuals in Fitzpatrick skin types I-III where lesions are more readily visible.[3][48] In contrast, blue nevi are underreported in darker-skinned populations, such as African Americans, due to camouflage by melanin pigmentation, leading to lower documented incidence; they are rarely seen in such groups.[48] Most blue nevi appear during childhood or adolescence, though they can develop at any age into adulthood; congenital cases are rare.[4][1]Demographics
Blue nevi, including common and cellular subtypes, can occur across all age groups but are most frequently diagnosed in young adults for the common variant and in middle-aged individuals for the cellular variant.[1] Lesions may be congenital, appearing at birth or in early childhood, or acquired later in adolescence and adulthood, with rare cases noted in older patients.[49][4] A female predominance is observed, with an overall ratio of approximately 3:2 women to men; this skew is more pronounced in cellular blue nevi, where the female-to-male ratio exceeds 2:1.[4][1] Blue nevi show racial variation in prevalence, occurring more frequently in Asian populations (3–5%) than in Caucasians (1–2%), though data on other ethnic groups remain limited.[48] No strong racial predilection is reported beyond these observations, and the lesions are documented across diverse populations without evidence of geographic clustering.[4]References
- https://www.dermatologyadvisor.com/home/decision-support-in-medicine/[dermatology](/page/Dermatology)/blue-nevus-common-dendritic-cellular-blue-nevus/