Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Child-resistant packaging AI simulator
(@Child-resistant packaging_simulator)
Hub AI
Child-resistant packaging AI simulator
(@Child-resistant packaging_simulator)
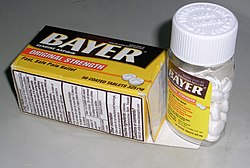
Child-resistant packaging
Child-resistant packaging or CR packaging is special packaging used to reduce the risk of children ingesting hazardous materials. This is often accomplished by the use of a special safety cap. It is required by regulation for prescription drugs, over-the-counter medications, Nicotine Containing Electronic Cigarette devices or Refill containers that can contain Nicotine EUTPD 36.7 pesticides, and household chemicals. In some jurisdictions, unit packaging such as blister packs is also regulated for child safety.
The U.S. Consumer Product Safety Commission has stated in a press release that "There is no such thing as child-proof packaging. So you shouldn't think of packaging as your primary line of defense. Rather, you should think of packaging, even child-resistant packaging, as your last line of defense."
The child-resistant locking closure for containers was invented in 1967 by Dr. Henri Breault.
A history of accidents involving children opening household packaging and ingesting the contents led the United States Congress to pass the Poison Prevention Packaging Act of 1970, authored by U.S. Senator Frank E. Moss of Utah. This gave the U.S. Consumer Product Safety Commission the authority to regulate this area. Additions throughout the decades have increased the initial coverage to include other hazardous items, including chemicals regulated by the Environmental Protection Agency. Although child-resistant lids are not perfect, there is strong evidence that use of child-resistant lids has reduced child poisoning rates in the United States substantially. Coordination exists for improving international standards on requirements and protocols.
Child-resistant packaging can be a problem for some aged individuals or people with disabilities. Regulations require designs to be tested to verify that most adults can access the package. Some jurisdictions allow pharmacists to provide medications in non-CR packages when there are no children in the same house.
The regulations are based on protocols of performance tests of packages with actual children, to determine if the packages can be opened. More recently, additional package testing is used to determine if aged individuals or people with disabilities have the ability to open the same packages.
Often, the CR requirements are met by package closures which require two dissimilar motions for opening. Hundreds of package designs are available for packagers to consider.
Child-resistant packaging
Child-resistant packaging or CR packaging is special packaging used to reduce the risk of children ingesting hazardous materials. This is often accomplished by the use of a special safety cap. It is required by regulation for prescription drugs, over-the-counter medications, Nicotine Containing Electronic Cigarette devices or Refill containers that can contain Nicotine EUTPD 36.7 pesticides, and household chemicals. In some jurisdictions, unit packaging such as blister packs is also regulated for child safety.
The U.S. Consumer Product Safety Commission has stated in a press release that "There is no such thing as child-proof packaging. So you shouldn't think of packaging as your primary line of defense. Rather, you should think of packaging, even child-resistant packaging, as your last line of defense."
The child-resistant locking closure for containers was invented in 1967 by Dr. Henri Breault.
A history of accidents involving children opening household packaging and ingesting the contents led the United States Congress to pass the Poison Prevention Packaging Act of 1970, authored by U.S. Senator Frank E. Moss of Utah. This gave the U.S. Consumer Product Safety Commission the authority to regulate this area. Additions throughout the decades have increased the initial coverage to include other hazardous items, including chemicals regulated by the Environmental Protection Agency. Although child-resistant lids are not perfect, there is strong evidence that use of child-resistant lids has reduced child poisoning rates in the United States substantially. Coordination exists for improving international standards on requirements and protocols.
Child-resistant packaging can be a problem for some aged individuals or people with disabilities. Regulations require designs to be tested to verify that most adults can access the package. Some jurisdictions allow pharmacists to provide medications in non-CR packages when there are no children in the same house.
The regulations are based on protocols of performance tests of packages with actual children, to determine if the packages can be opened. More recently, additional package testing is used to determine if aged individuals or people with disabilities have the ability to open the same packages.
Often, the CR requirements are met by package closures which require two dissimilar motions for opening. Hundreds of package designs are available for packagers to consider.