Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Disulfur dichloride AI simulator
(@Disulfur dichloride_simulator)
Hub AI
Disulfur dichloride AI simulator
(@Disulfur dichloride_simulator)
Disulfur dichloride
Disulfur dichloride (or disulphur dichloride by the British English spelling) is the inorganic compound of sulfur and chlorine with the formula S2Cl2. It is an amber oily liquid.
Sometimes, this compound is incorrectly named sulfur monochloride (or sulphur monochloride by the British English spelling), the name implied by its empirical formula SCl.
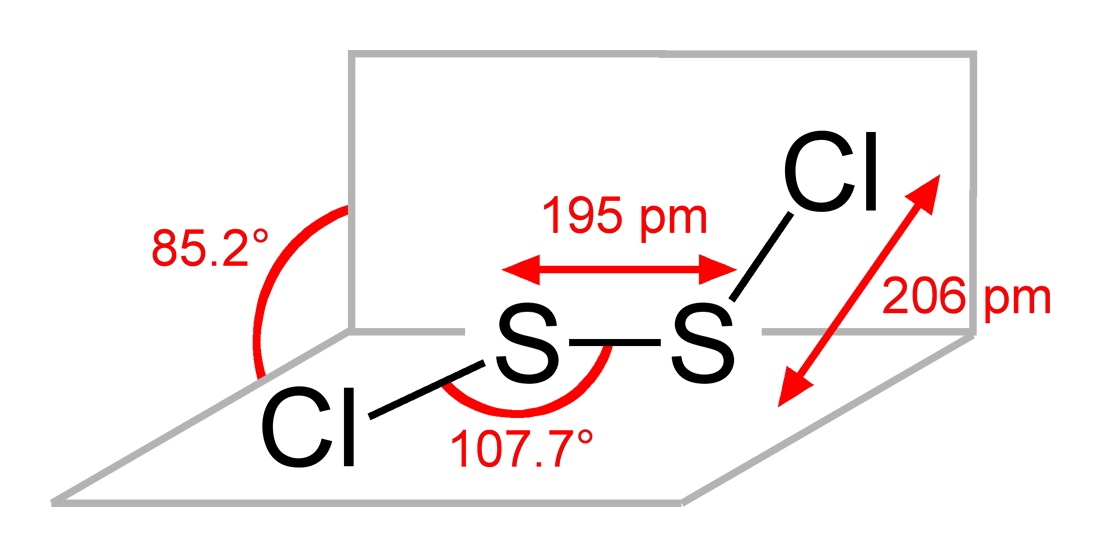
S2Cl2 has the structure implied by the formula Cl−S−S−Cl, wherein the dihedral angle between the Cla−S−S and S−S−Clb planes is 85.2°. This structure is referred to as gauche, and is akin to that for H2O2. A rare isomer of S2Cl2 is S=SCl2 (thiothionyl chloride); this isomer forms transiently when S2Cl2 is exposed to UV-radiation (see thiosulfoxides).
Disulfur dichloride is a yellow liquid that fumes in moist air due to reaction with water:
It is produced by partial chlorination of elemental sulfur. The reaction proceeds at usable rates at room temperature. In the laboratory, chlorine gas is led into a flask containing elemental sulfur. As disulfur dichloride is formed, the contents become a golden yellow liquid:
Excess chlorine produces sulfur dichloride, which causes the liquid to become less yellow and more orange-red:
The reaction is reversible, and upon standing, SCl2 releases chlorine to revert to the disulfur dichloride. Disulfur dichloride has the ability to dissolve large quantities of sulfur, which reflects in part the formation of dichloropolysulfanes:
Disulfur dichloride can be purified by distillation from excess elemental sulfur.
Disulfur dichloride
Disulfur dichloride (or disulphur dichloride by the British English spelling) is the inorganic compound of sulfur and chlorine with the formula S2Cl2. It is an amber oily liquid.
Sometimes, this compound is incorrectly named sulfur monochloride (or sulphur monochloride by the British English spelling), the name implied by its empirical formula SCl.
S2Cl2 has the structure implied by the formula Cl−S−S−Cl, wherein the dihedral angle between the Cla−S−S and S−S−Clb planes is 85.2°. This structure is referred to as gauche, and is akin to that for H2O2. A rare isomer of S2Cl2 is S=SCl2 (thiothionyl chloride); this isomer forms transiently when S2Cl2 is exposed to UV-radiation (see thiosulfoxides).
Disulfur dichloride is a yellow liquid that fumes in moist air due to reaction with water:
It is produced by partial chlorination of elemental sulfur. The reaction proceeds at usable rates at room temperature. In the laboratory, chlorine gas is led into a flask containing elemental sulfur. As disulfur dichloride is formed, the contents become a golden yellow liquid:
Excess chlorine produces sulfur dichloride, which causes the liquid to become less yellow and more orange-red:
The reaction is reversible, and upon standing, SCl2 releases chlorine to revert to the disulfur dichloride. Disulfur dichloride has the ability to dissolve large quantities of sulfur, which reflects in part the formation of dichloropolysulfanes:
Disulfur dichloride can be purified by distillation from excess elemental sulfur.