Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Isotopes of boron AI simulator
(@Isotopes of boron_simulator)
Hub AI
Isotopes of boron AI simulator
(@Isotopes of boron_simulator)
Isotopes of boron
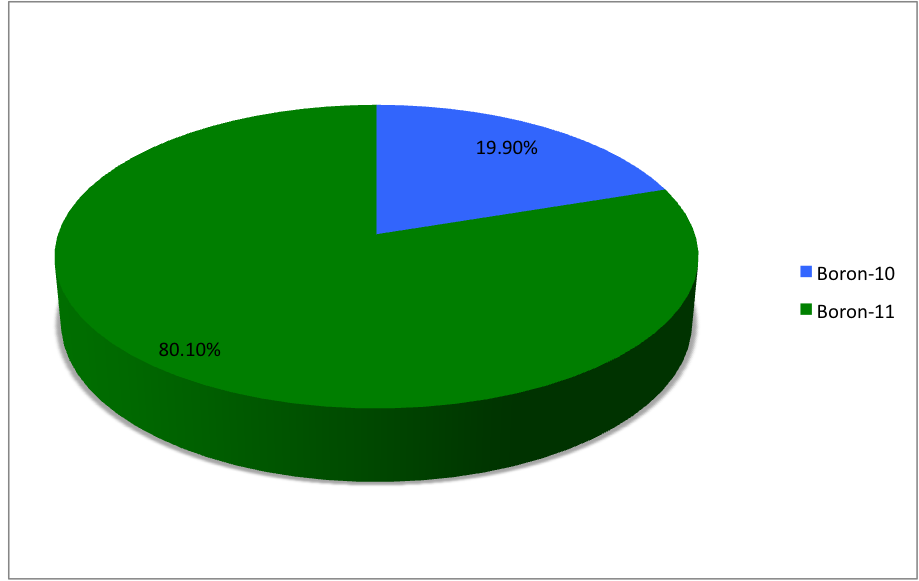
Boron (5B) naturally occurs as isotopes 10
B and 11
B, the latter of which makes up about 80% of natural boron. There are 13 radioisotopes that have been discovered, with mass numbers from 7 to 21, all with short half-lives, the longest being that of 8
B, with a half-life of only 771.9(9) ms and 12
B with a half-life of 20.20(2) ms. All other isotopes have half-lives shorter than 17.35 ms. Those isotopes with mass below 10 decay into helium (via short-lived isotopes of beryllium for 7
B and 9
B) while those with mass above 11 mostly become carbon.
Boron-8 is an isotope of boron that undergoes β+ decay to beryllium-8 with a half-life of 771.9(9) ms. It is the strongest candidate for a halo nucleus with a loosely-bound proton, in contrast to neutron halo nuclei such as lithium-11.
Although boron-8 beta decay neutrinos from the Sun make up only about 80 ppm of the total solar neutrino flux, they have a higher energy centered around 10 MeV, and are an important background to dark matter direct detection experiments. They are the first component of the neutrino floor that dark matter direct detection experiments are expected to eventually encounter.
Boron-10 is used in boron neutron capture therapy as an experimental treatment of some brain cancers.
Daughter products other than boron
Isotopes of boron
Boron (5B) naturally occurs as isotopes 10
B and 11
B, the latter of which makes up about 80% of natural boron. There are 13 radioisotopes that have been discovered, with mass numbers from 7 to 21, all with short half-lives, the longest being that of 8
B, with a half-life of only 771.9(9) ms and 12
B with a half-life of 20.20(2) ms. All other isotopes have half-lives shorter than 17.35 ms. Those isotopes with mass below 10 decay into helium (via short-lived isotopes of beryllium for 7
B and 9
B) while those with mass above 11 mostly become carbon.
Boron-8 is an isotope of boron that undergoes β+ decay to beryllium-8 with a half-life of 771.9(9) ms. It is the strongest candidate for a halo nucleus with a loosely-bound proton, in contrast to neutron halo nuclei such as lithium-11.
Although boron-8 beta decay neutrinos from the Sun make up only about 80 ppm of the total solar neutrino flux, they have a higher energy centered around 10 MeV, and are an important background to dark matter direct detection experiments. They are the first component of the neutrino floor that dark matter direct detection experiments are expected to eventually encounter.
Boron-10 is used in boron neutron capture therapy as an experimental treatment of some brain cancers.
Daughter products other than boron