Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Lactoperoxidase AI simulator
(@Lactoperoxidase_simulator)
Hub AI
Lactoperoxidase AI simulator
(@Lactoperoxidase_simulator)
Lactoperoxidase
Lactoperoxidase (LPO, EC 1.11.1.7) is a peroxidase enzyme secreted from mammary, salivary, tears and other mucosal glands including the lungs, bronchii and nose that function as a natural, first line of defense against bacteria and viral agents. Lactoperoxidase is a member of the heme peroxidase family of enzymes. In humans, lactoperoxidase is encoded by the LPO gene.
Lactoperoxidase catalyzes the oxidation of several inorganic and many organic substrates by hydrogen peroxide. Lactoperoxidase rapidly oxidizes iodide and slowly oxidizes bromide and is designated a haloperoxidase. Another important substrate is the pseudo-halide thiocyanate. The oxidized products display potent, non-specific bactericidal and antiviral activities, including destruction of the influenza virus. Lactoperoxidase together with its inorganic ion substrates, hydrogen peroxide, DUOX1 and DUOX2 and products are termed the lactoperoxidase system. Hence LPO is considered a very important defense against invasive bacteria and viral agents such as influenza and the SARS-CoV-2 virus when sufficient iodine is provided.
The LPO system plays an important role in the innate immune system by destroying bacteria in milk and mucosal (linings of mostly endodermal origin, covered in epithelium involved in absorption and secretion) secretions. Hence augmentation of the lactoperoxidase system may have therapeutic applications and applications for controlling bacteria in food and consumer health care products. This system does not vigorously attack DNA and is not mutagenic. However, under certain conditions, the LPO system may contribute to oxidative stress though recent evidence indicates LPO to be protective. LPO may contribute to the initiation of breast cancer, through its ability to oxidize estrogenic hormones producing free radical intermediates.
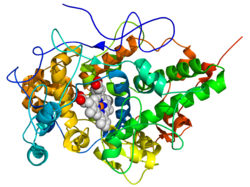
The structure of lactoperoxidase consists mainly of alpha-helices plus two short antiparallel beta-strands. Lactoperoxidase belongs to the heme peroxidase family of mammalian enzymes that also includes myeloperoxidase (MPO), eosinophil peroxidase (EPO), thyroid peroxidase (TPO), and prostaglandin H synthase (PGHS). A heme cofactor is covalently bound near the center of the protein.
Lactoperoxidase catalyzes the hydrogen peroxide (H2O2) oxidation of several acceptor molecules:
Specific examples include:
Depending on conditions the turnovers with SCN− or I− are roughly similar. The turnover with Br− is about 10−4 smaller than the other two anions. In mammals the source of hydrogen peroxide is one of the duox1 or duox2 enzymes which reduce dioxygen to H2O2 by oxidizing NADPH. In the laboratory the source of the hydrogen peroxide (H2O2) usually is the reaction of glucose with oxygen in the presence of the enzyme glucose oxidase (EC 1.1.3.4) that also takes place in saliva. Glucose, in turn, can be formed from starch in the presence of the saliva enzyme amyloglucosidase (EC 3.2.1.3).
These relatively short lived oxidized intermediates have potent bactericidal effects, hence lactoperoxidase is part of the antimicrobial defense system in tissues that express lactoperoxidase. The lactoperoxidase system is effective in killing a range of aerobic and certain anaerobic microorganisms. Research (1984): "The effect of lactoperoxidase-thiocyanate-hydrogen peroxide mixtures on bacteria is dependent on experimental conditions. If the bacteria are cultured after the exposure to lactoperoxidase-thiocyanate-hydrogen peroxide on nutrient agar under aerobic conditions, they may not grow, whereas they grow readily on blood agar under anaerobic conditions." In its antimicrobial capacity, lactoperoxidase appears to acts synergistically with lactoferrin and lysozyme.
Lactoperoxidase
Lactoperoxidase (LPO, EC 1.11.1.7) is a peroxidase enzyme secreted from mammary, salivary, tears and other mucosal glands including the lungs, bronchii and nose that function as a natural, first line of defense against bacteria and viral agents. Lactoperoxidase is a member of the heme peroxidase family of enzymes. In humans, lactoperoxidase is encoded by the LPO gene.
Lactoperoxidase catalyzes the oxidation of several inorganic and many organic substrates by hydrogen peroxide. Lactoperoxidase rapidly oxidizes iodide and slowly oxidizes bromide and is designated a haloperoxidase. Another important substrate is the pseudo-halide thiocyanate. The oxidized products display potent, non-specific bactericidal and antiviral activities, including destruction of the influenza virus. Lactoperoxidase together with its inorganic ion substrates, hydrogen peroxide, DUOX1 and DUOX2 and products are termed the lactoperoxidase system. Hence LPO is considered a very important defense against invasive bacteria and viral agents such as influenza and the SARS-CoV-2 virus when sufficient iodine is provided.
The LPO system plays an important role in the innate immune system by destroying bacteria in milk and mucosal (linings of mostly endodermal origin, covered in epithelium involved in absorption and secretion) secretions. Hence augmentation of the lactoperoxidase system may have therapeutic applications and applications for controlling bacteria in food and consumer health care products. This system does not vigorously attack DNA and is not mutagenic. However, under certain conditions, the LPO system may contribute to oxidative stress though recent evidence indicates LPO to be protective. LPO may contribute to the initiation of breast cancer, through its ability to oxidize estrogenic hormones producing free radical intermediates.
The structure of lactoperoxidase consists mainly of alpha-helices plus two short antiparallel beta-strands. Lactoperoxidase belongs to the heme peroxidase family of mammalian enzymes that also includes myeloperoxidase (MPO), eosinophil peroxidase (EPO), thyroid peroxidase (TPO), and prostaglandin H synthase (PGHS). A heme cofactor is covalently bound near the center of the protein.
Lactoperoxidase catalyzes the hydrogen peroxide (H2O2) oxidation of several acceptor molecules:
Specific examples include:
Depending on conditions the turnovers with SCN− or I− are roughly similar. The turnover with Br− is about 10−4 smaller than the other two anions. In mammals the source of hydrogen peroxide is one of the duox1 or duox2 enzymes which reduce dioxygen to H2O2 by oxidizing NADPH. In the laboratory the source of the hydrogen peroxide (H2O2) usually is the reaction of glucose with oxygen in the presence of the enzyme glucose oxidase (EC 1.1.3.4) that also takes place in saliva. Glucose, in turn, can be formed from starch in the presence of the saliva enzyme amyloglucosidase (EC 3.2.1.3).
These relatively short lived oxidized intermediates have potent bactericidal effects, hence lactoperoxidase is part of the antimicrobial defense system in tissues that express lactoperoxidase. The lactoperoxidase system is effective in killing a range of aerobic and certain anaerobic microorganisms. Research (1984): "The effect of lactoperoxidase-thiocyanate-hydrogen peroxide mixtures on bacteria is dependent on experimental conditions. If the bacteria are cultured after the exposure to lactoperoxidase-thiocyanate-hydrogen peroxide on nutrient agar under aerobic conditions, they may not grow, whereas they grow readily on blood agar under anaerobic conditions." In its antimicrobial capacity, lactoperoxidase appears to acts synergistically with lactoferrin and lysozyme.