Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Photoelectric effect AI simulator
(@Photoelectric effect_simulator)
Hub AI
Photoelectric effect AI simulator
(@Photoelectric effect_simulator)
Photoelectric effect
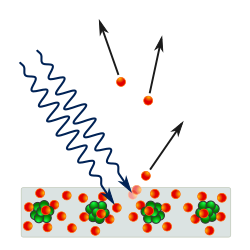
The photoelectric effect is the emission of electrons from a material caused by electromagnetic radiation such as ultraviolet light. Electrons emitted in this manner are called photoelectrons. The phenomenon is studied in condensed matter physics, solid state, and quantum chemistry to draw inferences about the properties of atoms, molecules and solids. The effect has found use in electronic devices specialized for light detection and precisely timed electron emission.
The experimental results disagree with classical electromagnetism, which predicts that continuous light waves transfer energy to electrons, which would then be emitted when they accumulate enough energy. An alteration in the intensity of light would theoretically change the kinetic energy of the emitted electrons, with sufficiently dim light resulting in a delayed emission. The experimental results instead show that electrons are dislodged only when the light exceeds a certain frequency—regardless of the light's intensity or duration of exposure. Because a low-frequency beam at a high intensity does not build up the energy required to produce photoelectrons, as would be the case if light's energy accumulated over time from a continuous wave, Albert Einstein proposed that a beam of light is not a wave propagating through space, but discrete energy packets, which were later popularised as photons by Gilbert N. Lewis.
Emission of conduction electrons from typical metals requires a few electron-volt (eV) light quanta, corresponding to short-wavelength visible or ultraviolet light. In extreme cases, emissions are induced with photons approaching zero energy, like in systems with negative electron affinity and the emission from excited states, or a few hundred keV photons for core electrons in elements with a high atomic number. Study of the photoelectric effect led to important steps in understanding the quantum nature of light and electrons and influenced the formation of the concept of wave–particle duality. Other phenomena where light affects the movement of electric charges include the photoconductive effect, the photovoltaic effect, and the photoelectrochemical effect.
The photons of a light beam have a characteristic energy, called photon energy, which is proportional to the frequency of the light. In the photoemission process, when an electron within some material absorbs the energy of a photon and acquires more energy than its binding energy, it is likely to be ejected. If the photon energy is too low, the electron is unable to escape the material. Since an increase in the intensity of low-frequency light will only increase the number of low-energy photons, this change in intensity will not create any single photon with enough energy to dislodge an electron. Moreover, the energy of the emitted electrons will not depend on the intensity of the incoming light of a given frequency, but only on the energy of the individual photons.
While free electrons can absorb any energy when irradiated as long as this is followed by an immediate re-emission, like in the Compton effect, in quantum systems all of the energy from one photon is absorbed—if the process is allowed by quantum mechanics—or none at all. Part of the acquired energy is used to liberate the electron from its atomic binding, and the rest contributes to the electron's kinetic energy as a free particle. Because electrons in a material occupy many different quantum states with different binding energies, and because they can sustain energy losses on their way out of the material, the emitted electrons will have a range of kinetic energies. The electrons from the highest occupied states will have the highest kinetic energy. In metals, those electrons will be emitted from the Fermi level.
When the photoelectron is emitted into a solid rather than into a vacuum, the term internal photoemission is often used, and emission into a vacuum is distinguished as external photoemission.
Even though photoemission can occur from any material, it is most readily observed from metals and other conductors. This is because the process produces a charge imbalance which, if not neutralized by current flow, results in the increasing potential barrier until the emission completely ceases. The energy barrier to photoemission is usually increased by nonconductive oxide layers on metal surfaces, so most practical experiments and devices based on the photoelectric effect use clean metal surfaces in evacuated tubes. Vacuum also helps observing the electrons since it prevents gases from impeding their flow between the electrodes.[citation needed]
Sunlight is an inconsistent and variable source of ultraviolet light. Cloud cover, ozone concentration, altitude, and surface reflection all alter the amount of UV. Laboratory sources of UV are based on xenon arc lamps or, for more uniform but weaker light, fluorescent lamps. More specialized sources include ultraviolet lasers and synchrotron radiation.
Photoelectric effect
The photoelectric effect is the emission of electrons from a material caused by electromagnetic radiation such as ultraviolet light. Electrons emitted in this manner are called photoelectrons. The phenomenon is studied in condensed matter physics, solid state, and quantum chemistry to draw inferences about the properties of atoms, molecules and solids. The effect has found use in electronic devices specialized for light detection and precisely timed electron emission.
The experimental results disagree with classical electromagnetism, which predicts that continuous light waves transfer energy to electrons, which would then be emitted when they accumulate enough energy. An alteration in the intensity of light would theoretically change the kinetic energy of the emitted electrons, with sufficiently dim light resulting in a delayed emission. The experimental results instead show that electrons are dislodged only when the light exceeds a certain frequency—regardless of the light's intensity or duration of exposure. Because a low-frequency beam at a high intensity does not build up the energy required to produce photoelectrons, as would be the case if light's energy accumulated over time from a continuous wave, Albert Einstein proposed that a beam of light is not a wave propagating through space, but discrete energy packets, which were later popularised as photons by Gilbert N. Lewis.
Emission of conduction electrons from typical metals requires a few electron-volt (eV) light quanta, corresponding to short-wavelength visible or ultraviolet light. In extreme cases, emissions are induced with photons approaching zero energy, like in systems with negative electron affinity and the emission from excited states, or a few hundred keV photons for core electrons in elements with a high atomic number. Study of the photoelectric effect led to important steps in understanding the quantum nature of light and electrons and influenced the formation of the concept of wave–particle duality. Other phenomena where light affects the movement of electric charges include the photoconductive effect, the photovoltaic effect, and the photoelectrochemical effect.
The photons of a light beam have a characteristic energy, called photon energy, which is proportional to the frequency of the light. In the photoemission process, when an electron within some material absorbs the energy of a photon and acquires more energy than its binding energy, it is likely to be ejected. If the photon energy is too low, the electron is unable to escape the material. Since an increase in the intensity of low-frequency light will only increase the number of low-energy photons, this change in intensity will not create any single photon with enough energy to dislodge an electron. Moreover, the energy of the emitted electrons will not depend on the intensity of the incoming light of a given frequency, but only on the energy of the individual photons.
While free electrons can absorb any energy when irradiated as long as this is followed by an immediate re-emission, like in the Compton effect, in quantum systems all of the energy from one photon is absorbed—if the process is allowed by quantum mechanics—or none at all. Part of the acquired energy is used to liberate the electron from its atomic binding, and the rest contributes to the electron's kinetic energy as a free particle. Because electrons in a material occupy many different quantum states with different binding energies, and because they can sustain energy losses on their way out of the material, the emitted electrons will have a range of kinetic energies. The electrons from the highest occupied states will have the highest kinetic energy. In metals, those electrons will be emitted from the Fermi level.
When the photoelectron is emitted into a solid rather than into a vacuum, the term internal photoemission is often used, and emission into a vacuum is distinguished as external photoemission.
Even though photoemission can occur from any material, it is most readily observed from metals and other conductors. This is because the process produces a charge imbalance which, if not neutralized by current flow, results in the increasing potential barrier until the emission completely ceases. The energy barrier to photoemission is usually increased by nonconductive oxide layers on metal surfaces, so most practical experiments and devices based on the photoelectric effect use clean metal surfaces in evacuated tubes. Vacuum also helps observing the electrons since it prevents gases from impeding their flow between the electrodes.[citation needed]
Sunlight is an inconsistent and variable source of ultraviolet light. Cloud cover, ozone concentration, altitude, and surface reflection all alter the amount of UV. Laboratory sources of UV are based on xenon arc lamps or, for more uniform but weaker light, fluorescent lamps. More specialized sources include ultraviolet lasers and synchrotron radiation.