Recent from talks
Knowledge base stats:
Talk channels stats:
Members stats:
Electron
The electron (e−
, or β−
in nuclear reactions) is a subatomic particle whose electric charge is negative one elementary charge. It is an elementary particle that comprises the ordinary matter that makes up the universe, along with up and down quarks.
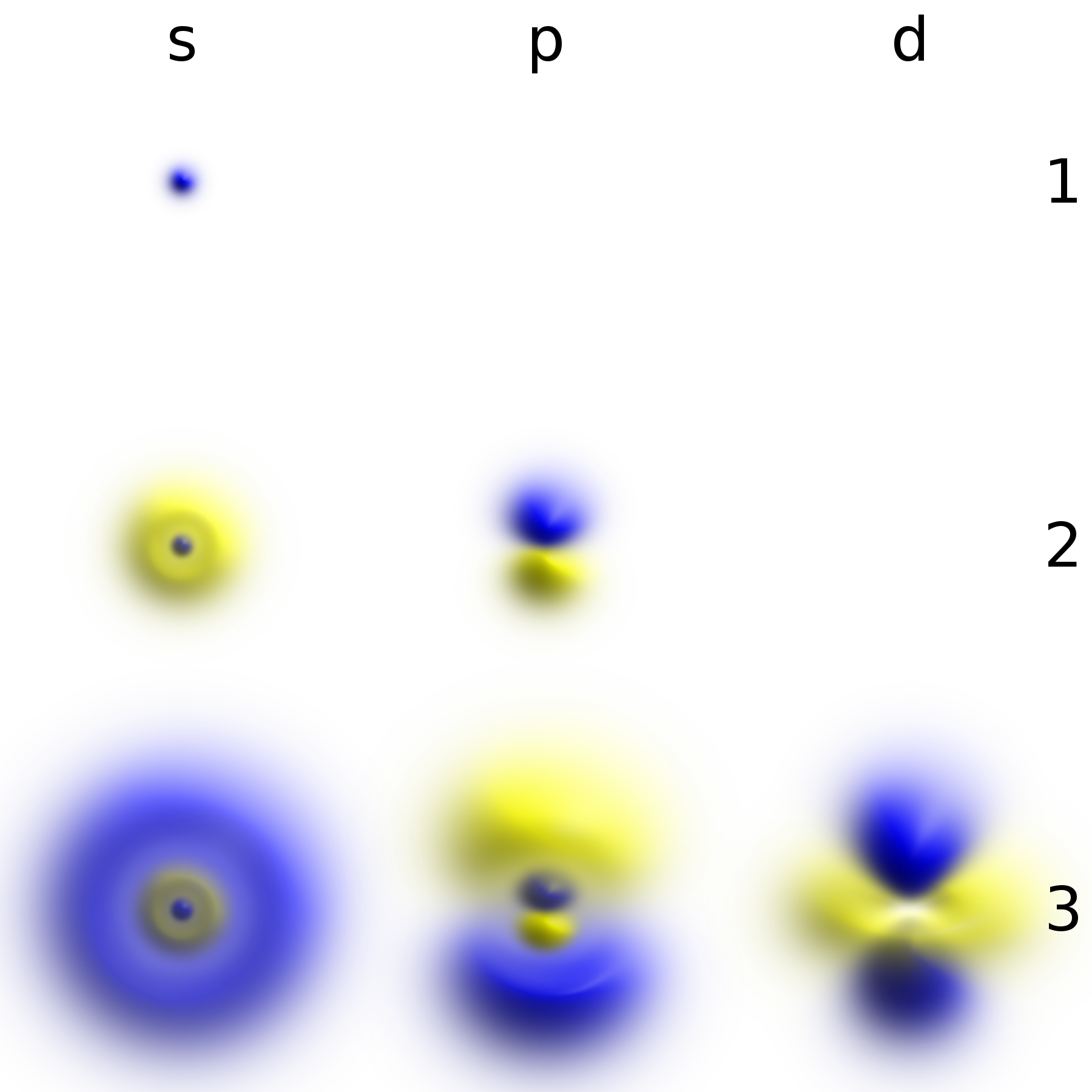
Electrons are extremely lightweight particles. In atoms, an electron's matter wave forms an atomic orbital around a positively charged atomic nucleus. The configuration and energy levels of an atom's electrons determine the atom's chemical properties. Electrons are bound to the nucleus to different degrees. The outermost or valence electrons are the least tightly bound and are responsible for the formation of chemical bonds between atoms to create molecules and crystals. These valence electrons also facilitate all types of chemical reactions by being transferred or shared between atoms. The inner electron shells make up the atomic core.
Electrons play a vital role in numerous physical phenomena due to their charge and mobile nature. In metals, the outermost electrons are delocalised and able to move freely, accounting for the high electrical and thermal conductivity of metals. In semiconductors, the number of mobile charge carriers (electrons and holes) can be finely tuned by doping, temperature, voltage and radiation – the basis of all modern electronics.
Electrons can be stripped entirely from their atoms to exist as free particles. As particle beams in a vacuum, free electrons can be accelerated, focused and used for applications like cathode ray tubes, electron microscopes, electron beam welding, lithography and particle accelerators that generate synchrotron radiation. Their charge and wave–particle duality make electrons indispensable in the modern technological world.
Electrons belong to the first generation of the lepton particle family, elementary particles that do not feel the strong nuclear force, and only interact through the weak and electromagnetic forces. Electrons are generally thought to be elementary particles because they have no known components or substructure. An electron's mass is approximately 1/1836 that of a proton. Quantum mechanical properties of the electron include an intrinsic angular momentum (spin) of half the reduced Planck constant, i.e. ħ/2. Being fermions, no two electrons can occupy the same quantum state, according to the Pauli exclusion principle. Like all elementary particles, electrons exhibit properties of both particles and waves. For example, electrons can collide like particles with other particles and can also be diffracted like light waves. The wave properties of electrons are easier to observe with experiments than those of other particles like neutrons and protons because electrons have a lower mass and hence a longer de Broglie wavelength for a given energy.
Electrons play an essential role in numerous physical phenomena, such as electricity, magnetism, chemistry, and thermal conductivity; they also participate in gravitational, electromagnetic, and weak interactions. Since an electron has charge, it has a surrounding electric field; if that electron is moving relative to an observer, the observer will observe it to generate a magnetic field. Electromagnetic fields produced from other sources will affect the motion of an electron according to the Lorentz force law. Electrons radiate or absorb energy in the form of photons when they are accelerated.
Laboratory instruments are capable of trapping individual electrons as well as electron plasma by the use of electromagnetic fields. Special telescopes can detect electron plasma in outer space. Electrons are involved in many applications, such as tribology or frictional charging, electrolysis, electrochemistry, battery technologies, electronics, welding, cathode-ray tubes, photoelectricity, photovoltaic solar panels, electron microscopes, radiation therapy, lasers, gaseous ionization detectors, and particle accelerators.
Interactions involving electrons with other subatomic particles are of interest in fields such as chemistry and nuclear physics. Atoms are composed of positive protons within atomic nuclei and the negative electrons without, held together by Coulomb force interaction. Ionization state (differences in the proportions of negative electrons versus positive nuclei) or sharing of the electrons between two or more atoms are the main causes of chemical bonding.
Hub AI
Electron AI simulator
(@Electron_simulator)
Electron
The electron (e−
, or β−
in nuclear reactions) is a subatomic particle whose electric charge is negative one elementary charge. It is an elementary particle that comprises the ordinary matter that makes up the universe, along with up and down quarks.
Electrons are extremely lightweight particles. In atoms, an electron's matter wave forms an atomic orbital around a positively charged atomic nucleus. The configuration and energy levels of an atom's electrons determine the atom's chemical properties. Electrons are bound to the nucleus to different degrees. The outermost or valence electrons are the least tightly bound and are responsible for the formation of chemical bonds between atoms to create molecules and crystals. These valence electrons also facilitate all types of chemical reactions by being transferred or shared between atoms. The inner electron shells make up the atomic core.
Electrons play a vital role in numerous physical phenomena due to their charge and mobile nature. In metals, the outermost electrons are delocalised and able to move freely, accounting for the high electrical and thermal conductivity of metals. In semiconductors, the number of mobile charge carriers (electrons and holes) can be finely tuned by doping, temperature, voltage and radiation – the basis of all modern electronics.
Electrons can be stripped entirely from their atoms to exist as free particles. As particle beams in a vacuum, free electrons can be accelerated, focused and used for applications like cathode ray tubes, electron microscopes, electron beam welding, lithography and particle accelerators that generate synchrotron radiation. Their charge and wave–particle duality make electrons indispensable in the modern technological world.
Electrons belong to the first generation of the lepton particle family, elementary particles that do not feel the strong nuclear force, and only interact through the weak and electromagnetic forces. Electrons are generally thought to be elementary particles because they have no known components or substructure. An electron's mass is approximately 1/1836 that of a proton. Quantum mechanical properties of the electron include an intrinsic angular momentum (spin) of half the reduced Planck constant, i.e. ħ/2. Being fermions, no two electrons can occupy the same quantum state, according to the Pauli exclusion principle. Like all elementary particles, electrons exhibit properties of both particles and waves. For example, electrons can collide like particles with other particles and can also be diffracted like light waves. The wave properties of electrons are easier to observe with experiments than those of other particles like neutrons and protons because electrons have a lower mass and hence a longer de Broglie wavelength for a given energy.
Electrons play an essential role in numerous physical phenomena, such as electricity, magnetism, chemistry, and thermal conductivity; they also participate in gravitational, electromagnetic, and weak interactions. Since an electron has charge, it has a surrounding electric field; if that electron is moving relative to an observer, the observer will observe it to generate a magnetic field. Electromagnetic fields produced from other sources will affect the motion of an electron according to the Lorentz force law. Electrons radiate or absorb energy in the form of photons when they are accelerated.
Laboratory instruments are capable of trapping individual electrons as well as electron plasma by the use of electromagnetic fields. Special telescopes can detect electron plasma in outer space. Electrons are involved in many applications, such as tribology or frictional charging, electrolysis, electrochemistry, battery technologies, electronics, welding, cathode-ray tubes, photoelectricity, photovoltaic solar panels, electron microscopes, radiation therapy, lasers, gaseous ionization detectors, and particle accelerators.
Interactions involving electrons with other subatomic particles are of interest in fields such as chemistry and nuclear physics. Atoms are composed of positive protons within atomic nuclei and the negative electrons without, held together by Coulomb force interaction. Ionization state (differences in the proportions of negative electrons versus positive nuclei) or sharing of the electrons between two or more atoms are the main causes of chemical bonding.