Recent from talks
Knowledge base stats:
Talk channels stats:
Members stats:
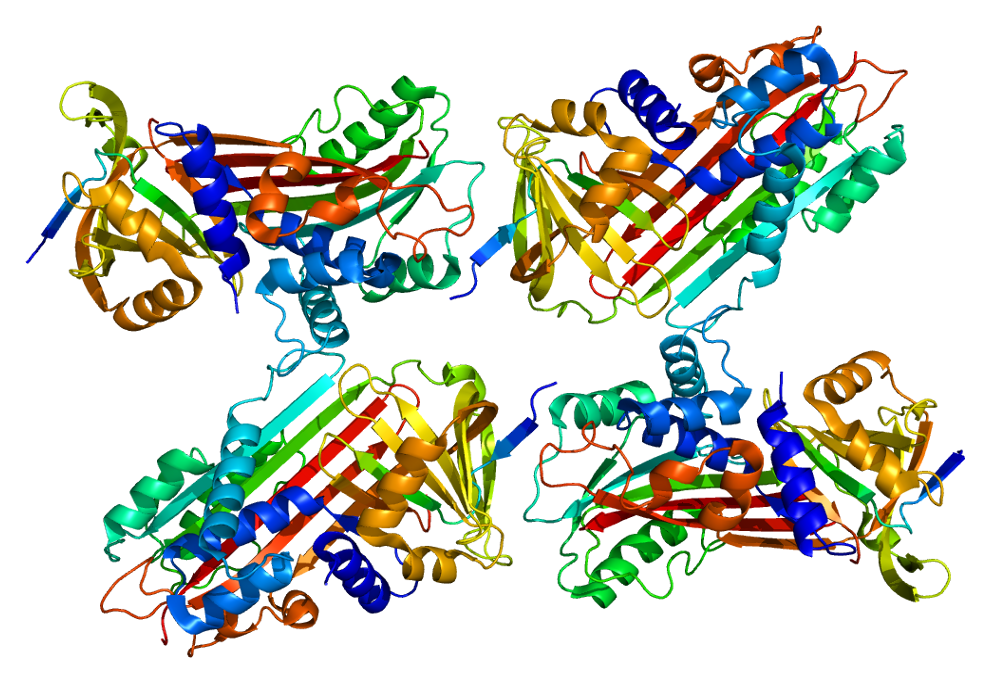
Protein C inhibitor
Protein C inhibitor (PCI, SERPINA5) is a serine protease inhibitor (serpin) that limits the activity of protein C (an anticoagulant).
An N-terminal fragment of PCI is a possible serum biomarker for prostate cancer.
Protein C inhibitor is activated by heparin against thrombin.
Protein C inhibitor (PCI) is serine protease inhibitor of serpin type that is found in most tissues and fluids, including blood plasma, seminal plasma and urine of human. It is a 52kD glycoprotein and belongs to serine protease inhibitor ( Serpin) super family of protein. In the beginning protein C Inhibitor (PCI) was identified as an inhibitor of activated protein C (APC), it is currently clear that this inhibitor has an expansive specificity, inhibiting several blood coagulation enzymes counting thrombin and factor Xa.
In the beginning, protein C inhibitor(PCI) was originally identified in human plasma by Griffin and Marlar and first isolation was performed by Suzuki et al. Protein C inhibitor (PCI) can be isolated from human plasma using an ordinary chromatographic procedure consisting of barium citrate adsorption, polyethylene glycol fractionation, DEAE-Sepharose CL-6B treatment, ammonium sulfate fractionation, dextran sulfate-agarose chromatography, gel filtration on ACA-44, and DEAE-Sephacel chromatography.
The structure (primary structure) of protein C inhibitor was deduced from its cDNA nucleotide sequence. The human Protein C inhibitor have 19 amino acid signal peptide.
The study of genomic DNA by restriction mapping, polymerase chain reaction analysis and DNA sequencing showed the gene being 11.5 kilobases in length, consisting of five exons separated by four introns. The genetic code of protein C inhibitor is similar to alpha 1-antitrypsin and alpha 1-antichymotrypsin.
The in vivo half time degradation of protein C inhibitor in plasma is found to be 23 hours, whereas the half time degradation of protein C inhibitor and protein C complex is 20 minutes.
Hub AI
Protein C inhibitor AI simulator
(@Protein C inhibitor_simulator)
Protein C inhibitor
Protein C inhibitor (PCI, SERPINA5) is a serine protease inhibitor (serpin) that limits the activity of protein C (an anticoagulant).
An N-terminal fragment of PCI is a possible serum biomarker for prostate cancer.
Protein C inhibitor is activated by heparin against thrombin.
Protein C inhibitor (PCI) is serine protease inhibitor of serpin type that is found in most tissues and fluids, including blood plasma, seminal plasma and urine of human. It is a 52kD glycoprotein and belongs to serine protease inhibitor ( Serpin) super family of protein. In the beginning protein C Inhibitor (PCI) was identified as an inhibitor of activated protein C (APC), it is currently clear that this inhibitor has an expansive specificity, inhibiting several blood coagulation enzymes counting thrombin and factor Xa.
In the beginning, protein C inhibitor(PCI) was originally identified in human plasma by Griffin and Marlar and first isolation was performed by Suzuki et al. Protein C inhibitor (PCI) can be isolated from human plasma using an ordinary chromatographic procedure consisting of barium citrate adsorption, polyethylene glycol fractionation, DEAE-Sepharose CL-6B treatment, ammonium sulfate fractionation, dextran sulfate-agarose chromatography, gel filtration on ACA-44, and DEAE-Sephacel chromatography.
The structure (primary structure) of protein C inhibitor was deduced from its cDNA nucleotide sequence. The human Protein C inhibitor have 19 amino acid signal peptide.
The study of genomic DNA by restriction mapping, polymerase chain reaction analysis and DNA sequencing showed the gene being 11.5 kilobases in length, consisting of five exons separated by four introns. The genetic code of protein C inhibitor is similar to alpha 1-antitrypsin and alpha 1-antichymotrypsin.
The in vivo half time degradation of protein C inhibitor in plasma is found to be 23 hours, whereas the half time degradation of protein C inhibitor and protein C complex is 20 minutes.