Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
R-process AI simulator
(@R-process_simulator)
Hub AI
R-process AI simulator
(@R-process_simulator)
R-process
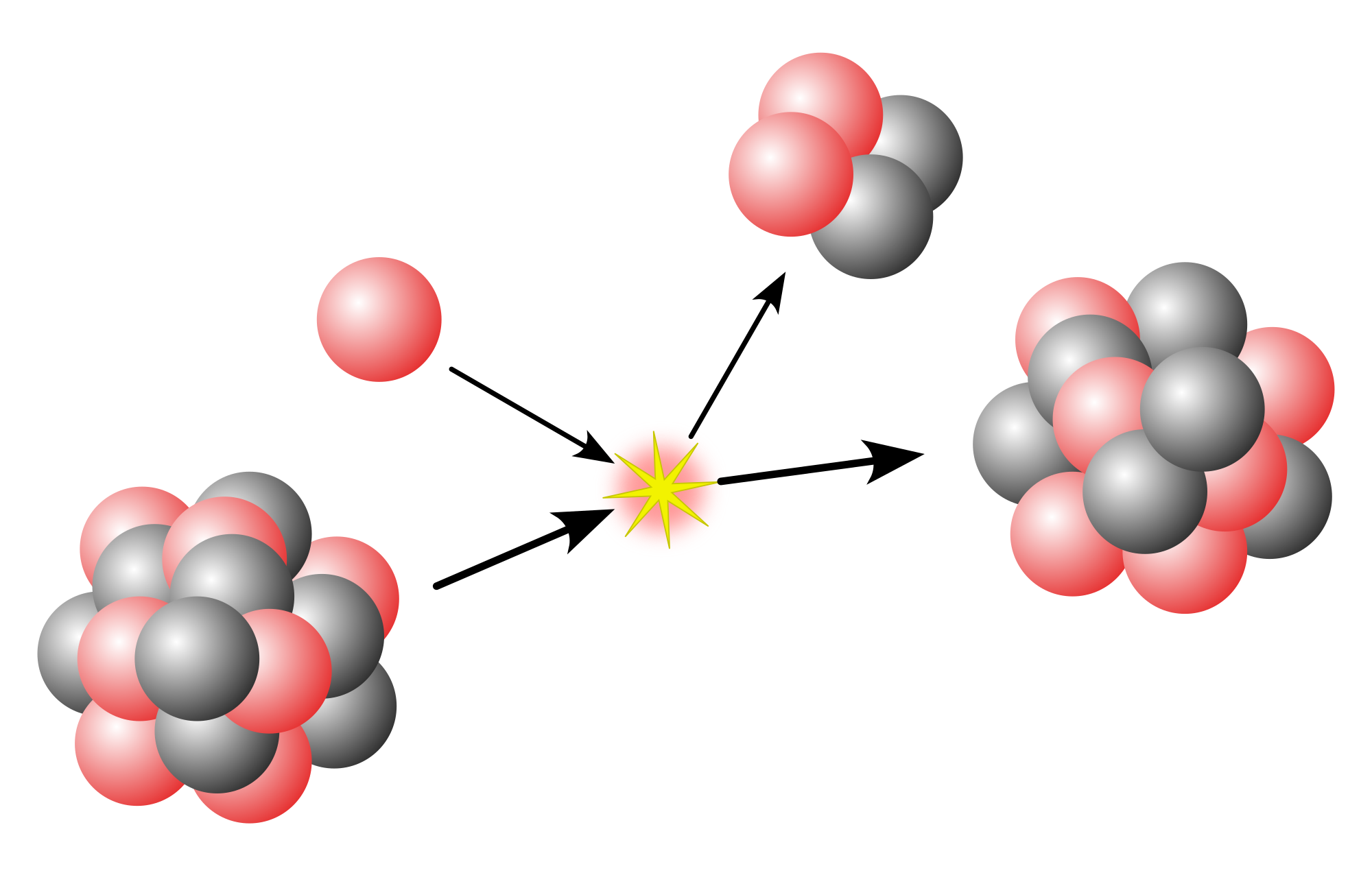
In nuclear astrophysics, the rapid neutron-capture process, also known as the r-process, is a set of nuclear reactions that is responsible for the creation of approximately half of the atomic nuclei heavier than iron, the "heavy elements", with the other half produced largely by the s-process. The r-process synthesizes the more neutron-rich of the stable isotopes of even elements, and those separated from the beta-stable isotopes by those that are not often have very low s-process yields and are considered r-only nuclei; the heaviest isotopes of most even elements from zinc to mercury fall into this category. Abundance peaks for the r-process occur near mass numbers A = 82 (elements Se, Br, and Kr), A = 130 (elements Te, I, and Xe) and A = 196 (elements Os, Ir, and Pt). Further, all the elements heavier than bismuth, including natural thorium and uranium (and other actinides) must ultimately originate in an r-process nucleus.
The r-process entails a succession of rapid neutron captures (hence the name) by one or more heavy seed nuclei, typically beginning with nuclei in the abundance peak centered on 56Fe. The captures must be rapid in the sense that the nuclei must not have time to undergo radioactive decay (typically via β− decay) before another neutron arrives to be captured. This sequence can continue up to the limit of stability of the increasingly neutron-rich nuclei (the neutron drip line) to physically retain neutrons as governed by the short range nuclear force. The r-process therefore must occur in locations where there exists a high density of free neutrons. At some time following the neutron captures, the nucleus beta-decays back to the line of stability (just as with fission products) resulting in a stable isotope of the same mass number A, and normally the most neutron-rich of those.
Early studies theorized that 1024 free neutrons per cm3 would be required, for temperatures of about 1 GK, in order to match the waiting points, at which no more neutrons can be captured, with the mass numbers of the abundance peaks for r-process nuclei. This amounts to almost a gram of free neutrons in every cubic centimeter, an astonishing number requiring extreme locations. Traditionally this suggested the material ejected from the re-expanded core of a core-collapse supernova, as part of supernova nucleosynthesis, or decompression of neutron star matter thrown off by a binary neutron star merger in a kilonova. The relative contribution of each of these sources to the astrophysical abundance of r-process elements is a matter of ongoing research as of 2018[update].
An r-process-like series of neutron captures (on uranium-238 normally) occurs to a minor extent in thermonuclear weapon explosions, and can be enhanced by purposeful design. The elements einsteinium (element 99) and fermium (element 100) in nuclear weapon fallout, and in general this neutron capture results in isotopes as heavy as A = 257.
The r-process contrasts with the s-process, the other predominant mechanism for the production of heavy elements, which is nucleosynthesis by means of slow captures of neutrons. In general, isotopes involved in the s-process have half-lives long enough to enable their study in laboratory experiments, but this is not typically true for isotopes involved in the r-process. The s-process primarily occurs within ordinary stars, particularly AGB stars, where the neutron flux is sufficient to cause neutron captures to recur every 10–100 years, much too slow for the r-process, which requires up to 100 captures per second. The s-process is secondary, meaning that it requires pre-existing heavy isotopes as seed nuclei to be converted into other heavy nuclei by a slow sequence of captures of free neutrons. The r-process scenarios create their own seed nuclei, so they might proceed in massive stars that contain no heavy seed nuclei. Taken together, the r- and s-processes account for almost the entire abundance of chemical elements heavier than iron. The historical challenge has been to locate physical settings appropriate to their time scales.
Following pioneering research into the Big Bang and the formation of helium in stars, an unknown process responsible for producing heavier elements found on Earth from hydrogen and helium was suspected to exist. One early attempt at explanation came from Subrahmanyan Chandrasekhar and Louis R. Henrich who postulated that elements were produced at temperatures between 6 billion and 8 billion K. Their theory accounted for elements up to chlorine, though there was no explanation for elements of atomic weight heavier than 40 amu at non-negligible abundances. This became the foundation of a study by Fred Hoyle, who hypothesized that conditions in the core of collapsing stars would enable nucleosynthesis of the remainder of the elements via rapid capture of densely packed free neutrons. However, there remained unanswered questions about equilibrium in stars that was required to balance beta-decays and precisely account for abundances of elements that would be formed in such conditions.
The need for a physical setting providing rapid neutron capture, which was known to almost certainly have a role in element formation, was also seen in a table of abundances of isotopes of heavy elements by Hans Suess and Harold Urey in 1956. Their abundance table revealed larger than average abundances of natural isotopes containing magic numbers of neutrons as well as abundance peaks about 10 amu lighter than stable nuclei containing magic numbers of neutrons which were also in abundance, suggesting that radioactive neutron-rich nuclei having the magic neutron numbers but roughly ten fewer protons were formed. These observations also implied that rapid neutron capture occurred faster than beta decay, and the resulting abundance peaks were caused by so-called waiting points at magic numbers. This process, rapid neutron capture by neutron-rich isotopes, became known as the r-process, whereas the s-process was named for its characteristic slow neutron capture. A table apportioning the heavy isotopes between the s-process and the r-process was published in 1957 in the B2FH review paper, which named the r-process and outlined the physics that guides it. Alastair G. W. Cameron also published a smaller study about the r-process in the same year.
The stationary r-process as described by the B2FH paper was first demonstrated in a time-dependent calculation at Caltech by Phillip A. Seeger, William A. Fowler and Donald D. Clayton, who found that no single temporal snapshot matched the solar r-process abundances, but, that when superposed, did achieve a successful characterization of the r-process abundance distribution. Shorter-time distributions emphasize abundances at atomic weights less than A = 140, whereas longer-time distributions emphasized those at atomic weights greater than A = 140. Subsequent treatments of the r-process reinforced those temporal features. Seeger et al. were also able to construct more quantitative apportionment between s-process and r-process of the abundance table of heavy isotopes, thereby establishing a more reliable abundance curve for the r-process isotopes than B2FH had been able to define. Today, the r-process abundances are determined using their technique of subtracting the more reliable s-process isotopic abundances from the total isotopic abundances and attributing the remainder to r-process nucleosynthesis. That r-process abundance curve (vs. atomic weight) has provided for many decades the target for theoretical computations of abundances synthesized by the physical r-process.
R-process
In nuclear astrophysics, the rapid neutron-capture process, also known as the r-process, is a set of nuclear reactions that is responsible for the creation of approximately half of the atomic nuclei heavier than iron, the "heavy elements", with the other half produced largely by the s-process. The r-process synthesizes the more neutron-rich of the stable isotopes of even elements, and those separated from the beta-stable isotopes by those that are not often have very low s-process yields and are considered r-only nuclei; the heaviest isotopes of most even elements from zinc to mercury fall into this category. Abundance peaks for the r-process occur near mass numbers A = 82 (elements Se, Br, and Kr), A = 130 (elements Te, I, and Xe) and A = 196 (elements Os, Ir, and Pt). Further, all the elements heavier than bismuth, including natural thorium and uranium (and other actinides) must ultimately originate in an r-process nucleus.
The r-process entails a succession of rapid neutron captures (hence the name) by one or more heavy seed nuclei, typically beginning with nuclei in the abundance peak centered on 56Fe. The captures must be rapid in the sense that the nuclei must not have time to undergo radioactive decay (typically via β− decay) before another neutron arrives to be captured. This sequence can continue up to the limit of stability of the increasingly neutron-rich nuclei (the neutron drip line) to physically retain neutrons as governed by the short range nuclear force. The r-process therefore must occur in locations where there exists a high density of free neutrons. At some time following the neutron captures, the nucleus beta-decays back to the line of stability (just as with fission products) resulting in a stable isotope of the same mass number A, and normally the most neutron-rich of those.
Early studies theorized that 1024 free neutrons per cm3 would be required, for temperatures of about 1 GK, in order to match the waiting points, at which no more neutrons can be captured, with the mass numbers of the abundance peaks for r-process nuclei. This amounts to almost a gram of free neutrons in every cubic centimeter, an astonishing number requiring extreme locations. Traditionally this suggested the material ejected from the re-expanded core of a core-collapse supernova, as part of supernova nucleosynthesis, or decompression of neutron star matter thrown off by a binary neutron star merger in a kilonova. The relative contribution of each of these sources to the astrophysical abundance of r-process elements is a matter of ongoing research as of 2018[update].
An r-process-like series of neutron captures (on uranium-238 normally) occurs to a minor extent in thermonuclear weapon explosions, and can be enhanced by purposeful design. The elements einsteinium (element 99) and fermium (element 100) in nuclear weapon fallout, and in general this neutron capture results in isotopes as heavy as A = 257.
The r-process contrasts with the s-process, the other predominant mechanism for the production of heavy elements, which is nucleosynthesis by means of slow captures of neutrons. In general, isotopes involved in the s-process have half-lives long enough to enable their study in laboratory experiments, but this is not typically true for isotopes involved in the r-process. The s-process primarily occurs within ordinary stars, particularly AGB stars, where the neutron flux is sufficient to cause neutron captures to recur every 10–100 years, much too slow for the r-process, which requires up to 100 captures per second. The s-process is secondary, meaning that it requires pre-existing heavy isotopes as seed nuclei to be converted into other heavy nuclei by a slow sequence of captures of free neutrons. The r-process scenarios create their own seed nuclei, so they might proceed in massive stars that contain no heavy seed nuclei. Taken together, the r- and s-processes account for almost the entire abundance of chemical elements heavier than iron. The historical challenge has been to locate physical settings appropriate to their time scales.
Following pioneering research into the Big Bang and the formation of helium in stars, an unknown process responsible for producing heavier elements found on Earth from hydrogen and helium was suspected to exist. One early attempt at explanation came from Subrahmanyan Chandrasekhar and Louis R. Henrich who postulated that elements were produced at temperatures between 6 billion and 8 billion K. Their theory accounted for elements up to chlorine, though there was no explanation for elements of atomic weight heavier than 40 amu at non-negligible abundances. This became the foundation of a study by Fred Hoyle, who hypothesized that conditions in the core of collapsing stars would enable nucleosynthesis of the remainder of the elements via rapid capture of densely packed free neutrons. However, there remained unanswered questions about equilibrium in stars that was required to balance beta-decays and precisely account for abundances of elements that would be formed in such conditions.
The need for a physical setting providing rapid neutron capture, which was known to almost certainly have a role in element formation, was also seen in a table of abundances of isotopes of heavy elements by Hans Suess and Harold Urey in 1956. Their abundance table revealed larger than average abundances of natural isotopes containing magic numbers of neutrons as well as abundance peaks about 10 amu lighter than stable nuclei containing magic numbers of neutrons which were also in abundance, suggesting that radioactive neutron-rich nuclei having the magic neutron numbers but roughly ten fewer protons were formed. These observations also implied that rapid neutron capture occurred faster than beta decay, and the resulting abundance peaks were caused by so-called waiting points at magic numbers. This process, rapid neutron capture by neutron-rich isotopes, became known as the r-process, whereas the s-process was named for its characteristic slow neutron capture. A table apportioning the heavy isotopes between the s-process and the r-process was published in 1957 in the B2FH review paper, which named the r-process and outlined the physics that guides it. Alastair G. W. Cameron also published a smaller study about the r-process in the same year.
The stationary r-process as described by the B2FH paper was first demonstrated in a time-dependent calculation at Caltech by Phillip A. Seeger, William A. Fowler and Donald D. Clayton, who found that no single temporal snapshot matched the solar r-process abundances, but, that when superposed, did achieve a successful characterization of the r-process abundance distribution. Shorter-time distributions emphasize abundances at atomic weights less than A = 140, whereas longer-time distributions emphasized those at atomic weights greater than A = 140. Subsequent treatments of the r-process reinforced those temporal features. Seeger et al. were also able to construct more quantitative apportionment between s-process and r-process of the abundance table of heavy isotopes, thereby establishing a more reliable abundance curve for the r-process isotopes than B2FH had been able to define. Today, the r-process abundances are determined using their technique of subtracting the more reliable s-process isotopic abundances from the total isotopic abundances and attributing the remainder to r-process nucleosynthesis. That r-process abundance curve (vs. atomic weight) has provided for many decades the target for theoretical computations of abundances synthesized by the physical r-process.