Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Ractopamine AI simulator
(@Ractopamine_simulator)
Hub AI
Ractopamine AI simulator
(@Ractopamine_simulator)
Ractopamine
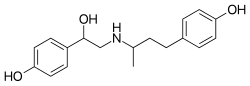
Ractopamine (/rækˈtɒpəmaɪn, -miːn/) is an animal feed additive used to promote leanness and increase food conversion efficiency in farmed animals in few countries, banned in most. Pharmacologically, it is a phenol-based TAAR1 agonist and β adrenoreceptor agonist that stimulates β1 and β2 adrenergic receptors. It is most commonly administered to animals for meat production as ractopamine hydrochloride. It is the active ingredient in products marketed in the US as Paylean for swine, Optaflexx for cattle, and Topmax for turkeys. It was developed by Elanco Animal Health, a former division of Eli Lilly and Company.
As of 2025, the use of ractopamine "is banned or its use restricted in 168 nations", including the European Union, China and Russia, while it is legal in some other countries, with differing maximum residual limits (MRLs) in meat. Examples include the United States (50 ppb in pork, 30 ppb in beef); Japan, Taiwan and South Korea (10 ppb, in accordance with the Codex Alimentarius Commission); and New Zealand (0.1 ppb).
Commercial ractopamine is a mixture of all four possible stereoisomers. It is also a positional isomer of dobutamine, a related drug.
When used as a food additive, ractopamine added to feed can be distributed by the blood to the muscle tissues, where it serves as a full agonist to murine (mouse or rat) TAAR1, a receptor protein (not necessarily in humans). It is also an agonist to beta-adrenergic receptors and a dopamine releasing agent. A cascade of events will then be initiated to increase protein synthesis, which results in increased muscle fiber size. Ractopamine is known to increase the rate of weight gain, improve feed efficiency, and increase carcass leanness in finishing swine. Its use in finishing swine yields about 3 kg (6.6 lb) of additional lean pork per animal, and improves feed efficiency by 10%. In cattle on 28 January 2004 Elanco Animal Health made Optaflexx commercially available in the US. Optaflexx is a "medicated feed additive that is labeled only for use in steers or market heifers (not breeding heifers or bulls) during the last 28–42 days on feed."
On 6 July 2012, the international reference standard Codex Alimentarius Commission narrowly approved the adoption of a maximum residue limit (MRL) of 10 parts per billion (ppb) for muscle cuts of beef and pork. Setting any limit was a controversial move. Countries with major meat export markets had been lobbying for the establishment of such a standard for several years to use it as leverage to erode individual national-level bans in World Trade Organization disputes. Consumers International, a world federation of consumer groups that represents 220 consumer organizations in 115 countries, strongly opposed the move.
A 2020 petition to the Food and Drug Administration to reconsider the approval of ractopamine stated that 168 other nations had banned or restricted its use. In 2025 and in response to an unreasonable delay lawsuit, the FDA responded acknowledging the number had increased from 160 in 2012, and questioned the relevance of the figure.
Ractopamine has not been allowed in the European Union, based on the 2009 EU's Food Safety Authority's opinion on its safety evaluation, which concluded that available data were insufficient to derive a maximum residue limit as a 'safe residue level for human consumption'. The uncertainty was particularly great for people who might be thought to be more susceptible than most to an increase in β adrenergic stimulation from consuming the additive, such as children or people with cardiovascular disease, and that simply increasing the "uncertainty factor" built into the calculation as a safety factor would rapidly become arbitrary.
Ractopamine use as a feed additive is authorized in the United States, Canada, and Mexico. In the US, ractopamine is allowed to be used at a feed concentration of 5–20 mg/kg feed for finishing pigs and in dosages of 5–10 mg/kg feed for finishing pigs heavier than 109 kg. The maximum residue limit for ractopamine for meat in the US is 50 parts per billion (ppb), or five times the standard set by the Codex Alimentarius. Ractopamine was approved by the FDA in 1999 for use in swine, in 2003 for use in cattle, and in 2008 for use in turkeys.
Ractopamine
Ractopamine (/rækˈtɒpəmaɪn, -miːn/) is an animal feed additive used to promote leanness and increase food conversion efficiency in farmed animals in few countries, banned in most. Pharmacologically, it is a phenol-based TAAR1 agonist and β adrenoreceptor agonist that stimulates β1 and β2 adrenergic receptors. It is most commonly administered to animals for meat production as ractopamine hydrochloride. It is the active ingredient in products marketed in the US as Paylean for swine, Optaflexx for cattle, and Topmax for turkeys. It was developed by Elanco Animal Health, a former division of Eli Lilly and Company.
As of 2025, the use of ractopamine "is banned or its use restricted in 168 nations", including the European Union, China and Russia, while it is legal in some other countries, with differing maximum residual limits (MRLs) in meat. Examples include the United States (50 ppb in pork, 30 ppb in beef); Japan, Taiwan and South Korea (10 ppb, in accordance with the Codex Alimentarius Commission); and New Zealand (0.1 ppb).
Commercial ractopamine is a mixture of all four possible stereoisomers. It is also a positional isomer of dobutamine, a related drug.
When used as a food additive, ractopamine added to feed can be distributed by the blood to the muscle tissues, where it serves as a full agonist to murine (mouse or rat) TAAR1, a receptor protein (not necessarily in humans). It is also an agonist to beta-adrenergic receptors and a dopamine releasing agent. A cascade of events will then be initiated to increase protein synthesis, which results in increased muscle fiber size. Ractopamine is known to increase the rate of weight gain, improve feed efficiency, and increase carcass leanness in finishing swine. Its use in finishing swine yields about 3 kg (6.6 lb) of additional lean pork per animal, and improves feed efficiency by 10%. In cattle on 28 January 2004 Elanco Animal Health made Optaflexx commercially available in the US. Optaflexx is a "medicated feed additive that is labeled only for use in steers or market heifers (not breeding heifers or bulls) during the last 28–42 days on feed."
On 6 July 2012, the international reference standard Codex Alimentarius Commission narrowly approved the adoption of a maximum residue limit (MRL) of 10 parts per billion (ppb) for muscle cuts of beef and pork. Setting any limit was a controversial move. Countries with major meat export markets had been lobbying for the establishment of such a standard for several years to use it as leverage to erode individual national-level bans in World Trade Organization disputes. Consumers International, a world federation of consumer groups that represents 220 consumer organizations in 115 countries, strongly opposed the move.
A 2020 petition to the Food and Drug Administration to reconsider the approval of ractopamine stated that 168 other nations had banned or restricted its use. In 2025 and in response to an unreasonable delay lawsuit, the FDA responded acknowledging the number had increased from 160 in 2012, and questioned the relevance of the figure.
Ractopamine has not been allowed in the European Union, based on the 2009 EU's Food Safety Authority's opinion on its safety evaluation, which concluded that available data were insufficient to derive a maximum residue limit as a 'safe residue level for human consumption'. The uncertainty was particularly great for people who might be thought to be more susceptible than most to an increase in β adrenergic stimulation from consuming the additive, such as children or people with cardiovascular disease, and that simply increasing the "uncertainty factor" built into the calculation as a safety factor would rapidly become arbitrary.
Ractopamine use as a feed additive is authorized in the United States, Canada, and Mexico. In the US, ractopamine is allowed to be used at a feed concentration of 5–20 mg/kg feed for finishing pigs and in dosages of 5–10 mg/kg feed for finishing pigs heavier than 109 kg. The maximum residue limit for ractopamine for meat in the US is 50 parts per billion (ppb), or five times the standard set by the Codex Alimentarius. Ractopamine was approved by the FDA in 1999 for use in swine, in 2003 for use in cattle, and in 2008 for use in turkeys.