Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Tacticity AI simulator
(@Tacticity_simulator)
Hub AI
Tacticity AI simulator
(@Tacticity_simulator)
Tacticity
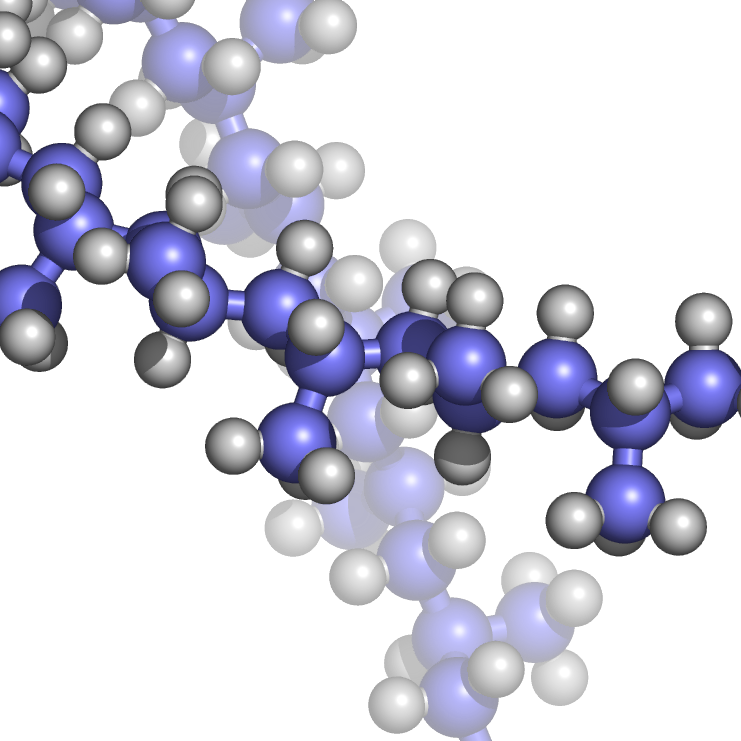
Tacticity (from Greek: τακτικός, romanized: taktikos, "relating to arrangement or order") is the relative stereochemistry of adjacent chiral centers within a macromolecule.[better source needed] The practical significance of tacticity rests on the effects on the physical properties of the polymer. The regularity of the macromolecular structure influences the degree to which it has rigid, crystalline long range order or flexible, amorphous long range disorder. Precise knowledge of tacticity of a polymer also helps understanding at what temperature a polymer melts, how soluble it is in a solvent, as well as its mechanical properties.
A tactic macromolecule in the IUPAC definition is a macromolecule in which essentially all the configurational (repeating) units are identical. In a hydrocarbon macromolecule with all carbon atoms making up the backbone in a tetrahedral molecular geometry, the zigzag backbone is in the paper plane with the substituents either sticking out of the paper or retreating into the paper;[excessive detail?], this projection is called the Natta projection after Giulio Natta.[not verified in body] Tacticity is particularly significant in vinyl polymers of the type -H
2C-CH(R)-, where each repeating unit contains a substituent R attached to one side of the polymer backbone. The arrangement of these substituents can follow a regular pattern- appearing on the same side as the previous one, on the opposite side, or in a random configuration relative to the preceding unit. Monotactic macromolecules have one stereoisomeric atom per repeat unit,[not verified in body] ditactic to n-tactic macromolecules have more than one stereoisomeric atom per unit.[not verified in body]
The orderliness of the succession of configurational repeating units in
the main chain of a regular macromolecule, a regular oligomer molecule,
a regular block, or a regular chain.
Two adjacent structural units in a polymer molecule constitute a diad. Diads overlap: each structural unit is considered part of two diads, one diad with each neighbor. If a diad consists of two identically oriented units, the diad is called an m diad (formerly meso diad, as in a meso compound, now proscribed). If a diad consists of units oriented in opposition, the diad is called an r diad (formerly racemo diad, as in a racemic compound, now proscribed). In the case of vinyl polymer molecules, an m diad is one in which the substituents are oriented on the same side of the polymer backbone; in the Natta projection, they both point into the plane or both point out of the plane.
The stereochemistry of macromolecules can be defined even more precisely with the introduction of triads. An isotactic triad (mm) is made up of two overlapping m diads, a syndiotactic triad (also spelled syndyotactic) (rr) consists of two overlapping r diads, and a heterotactic triad (rm) is composed of an r diad overlapping an m diad. The mass fraction of isotactic (mm) triads is a common quantitative measure of tacticity.
When the stereochemistry of a macromolecule is considered to be a Bernoulli process, the triad composition can be calculated from the probability Pm of a diad being m type. For example, when this probability is 0.25 then the probability of finding:
with a total probability of 1. Similar relationships with diads exist for tetrads.
The definition of tetrads and pentads introduce further sophistication and precision to defining tacticity, especially when information on long-range ordering is desirable.[citation needed] Tacticity measurements obtained by carbon-13 NMR are typically expressed in terms of the relative abundance of various pentads within the polymer molecule, e.g. mmmm, mrrm.[according to whom?]
Tacticity
Tacticity (from Greek: τακτικός, romanized: taktikos, "relating to arrangement or order") is the relative stereochemistry of adjacent chiral centers within a macromolecule.[better source needed] The practical significance of tacticity rests on the effects on the physical properties of the polymer. The regularity of the macromolecular structure influences the degree to which it has rigid, crystalline long range order or flexible, amorphous long range disorder. Precise knowledge of tacticity of a polymer also helps understanding at what temperature a polymer melts, how soluble it is in a solvent, as well as its mechanical properties.
A tactic macromolecule in the IUPAC definition is a macromolecule in which essentially all the configurational (repeating) units are identical. In a hydrocarbon macromolecule with all carbon atoms making up the backbone in a tetrahedral molecular geometry, the zigzag backbone is in the paper plane with the substituents either sticking out of the paper or retreating into the paper;[excessive detail?], this projection is called the Natta projection after Giulio Natta.[not verified in body] Tacticity is particularly significant in vinyl polymers of the type -H
2C-CH(R)-, where each repeating unit contains a substituent R attached to one side of the polymer backbone. The arrangement of these substituents can follow a regular pattern- appearing on the same side as the previous one, on the opposite side, or in a random configuration relative to the preceding unit. Monotactic macromolecules have one stereoisomeric atom per repeat unit,[not verified in body] ditactic to n-tactic macromolecules have more than one stereoisomeric atom per unit.[not verified in body]
The orderliness of the succession of configurational repeating units in
the main chain of a regular macromolecule, a regular oligomer molecule,
a regular block, or a regular chain.
Two adjacent structural units in a polymer molecule constitute a diad. Diads overlap: each structural unit is considered part of two diads, one diad with each neighbor. If a diad consists of two identically oriented units, the diad is called an m diad (formerly meso diad, as in a meso compound, now proscribed). If a diad consists of units oriented in opposition, the diad is called an r diad (formerly racemo diad, as in a racemic compound, now proscribed). In the case of vinyl polymer molecules, an m diad is one in which the substituents are oriented on the same side of the polymer backbone; in the Natta projection, they both point into the plane or both point out of the plane.
The stereochemistry of macromolecules can be defined even more precisely with the introduction of triads. An isotactic triad (mm) is made up of two overlapping m diads, a syndiotactic triad (also spelled syndyotactic) (rr) consists of two overlapping r diads, and a heterotactic triad (rm) is composed of an r diad overlapping an m diad. The mass fraction of isotactic (mm) triads is a common quantitative measure of tacticity.
When the stereochemistry of a macromolecule is considered to be a Bernoulli process, the triad composition can be calculated from the probability Pm of a diad being m type. For example, when this probability is 0.25 then the probability of finding:
with a total probability of 1. Similar relationships with diads exist for tetrads.
The definition of tetrads and pentads introduce further sophistication and precision to defining tacticity, especially when information on long-range ordering is desirable.[citation needed] Tacticity measurements obtained by carbon-13 NMR are typically expressed in terms of the relative abundance of various pentads within the polymer molecule, e.g. mmmm, mrrm.[according to whom?]