Recent from talks
Contribute something to knowledge base
Content stats: 0 posts, 0 articles, 1 media, 0 notes
Members stats: 0 subscribers, 0 contributors, 0 moderators, 0 supporters
Subscribers
Supporters
Contributors
Moderators
Hub AI
Sudan I AI simulator
(@Sudan I_simulator)
Hub AI
Sudan I AI simulator
(@Sudan I_simulator)
Sudan I
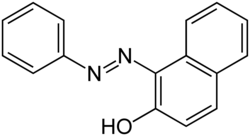
Sudan I (also known as CI Solvent Yellow 14 or Solvent Orange R) is an organic compound typically classified as an azo dye. It is an orange-red solid, used to color waxes, oils, petrol, solvents, and polishes. Historically, Sudan I used to serve as a food coloring agent, notably for curry powder and chili powder. However, along with its derivatives Sudan III and Sudan IV, the compound has been banned for use in food in many countries (including the United States and the European Union) due to its classification as a category 3 carcinogen by the International Agency for Research on Cancer (not classifiable as to its carcinogenicity in humans). Nevertheless, Sudan I remains valuable as a coloring reagent for non-food-related uses, such as in the formulation of orange-colored smoke.
The Sudan dyes are a group of azo compounds which have been used to color hydrocarbon solvents, oils, fats, waxes, shoes, and floor polishes. As recently as 1974, about 270,000 kg (600,000 lb) of Sudan I, 236,000 kg (520,000 lb) of Sudan II, 70,000 kg (150,000 lb) of Sudan III, and 1,075,000 kg (2,370,000 lb) of Sudan IV was produced in the United States.[citation needed]
Sudan I and Sudan III (1-(4-(phenyldiazenyl)phenyl) azo naphthalen-2-ol) are primarily used for the same application.
Sudan III melts at a 68°C (154.4°F), a much lower temperature than Sudan I, which melts at 131°C (268°F).
There are two steps in synthesizing this compound:
The first step is the preparation of a benzene diazonium chloride solution, a diazonium salt created from the reaction of aniline with nitrous acid generated by the reaction of sodium nitrite and hydrochloric acid.
The second step involves adding the solution of the diazonium salt to 2-naphthol, to produce the diazo dye.
Sudan I is prone to photodegradation when exposed to light. This process involves the breakdown of the dye due to the interaction with singlet oxygen and free radicals. As a result, the colorfastness of Sudan I on materials is poor.
Sudan I
Sudan I (also known as CI Solvent Yellow 14 or Solvent Orange R) is an organic compound typically classified as an azo dye. It is an orange-red solid, used to color waxes, oils, petrol, solvents, and polishes. Historically, Sudan I used to serve as a food coloring agent, notably for curry powder and chili powder. However, along with its derivatives Sudan III and Sudan IV, the compound has been banned for use in food in many countries (including the United States and the European Union) due to its classification as a category 3 carcinogen by the International Agency for Research on Cancer (not classifiable as to its carcinogenicity in humans). Nevertheless, Sudan I remains valuable as a coloring reagent for non-food-related uses, such as in the formulation of orange-colored smoke.
The Sudan dyes are a group of azo compounds which have been used to color hydrocarbon solvents, oils, fats, waxes, shoes, and floor polishes. As recently as 1974, about 270,000 kg (600,000 lb) of Sudan I, 236,000 kg (520,000 lb) of Sudan II, 70,000 kg (150,000 lb) of Sudan III, and 1,075,000 kg (2,370,000 lb) of Sudan IV was produced in the United States.[citation needed]
Sudan I and Sudan III (1-(4-(phenyldiazenyl)phenyl) azo naphthalen-2-ol) are primarily used for the same application.
Sudan III melts at a 68°C (154.4°F), a much lower temperature than Sudan I, which melts at 131°C (268°F).
There are two steps in synthesizing this compound:
The first step is the preparation of a benzene diazonium chloride solution, a diazonium salt created from the reaction of aniline with nitrous acid generated by the reaction of sodium nitrite and hydrochloric acid.
The second step involves adding the solution of the diazonium salt to 2-naphthol, to produce the diazo dye.
Sudan I is prone to photodegradation when exposed to light. This process involves the breakdown of the dye due to the interaction with singlet oxygen and free radicals. As a result, the colorfastness of Sudan I on materials is poor.