Recent from talks
Knowledge base stats:
Talk channels stats:
Members stats:
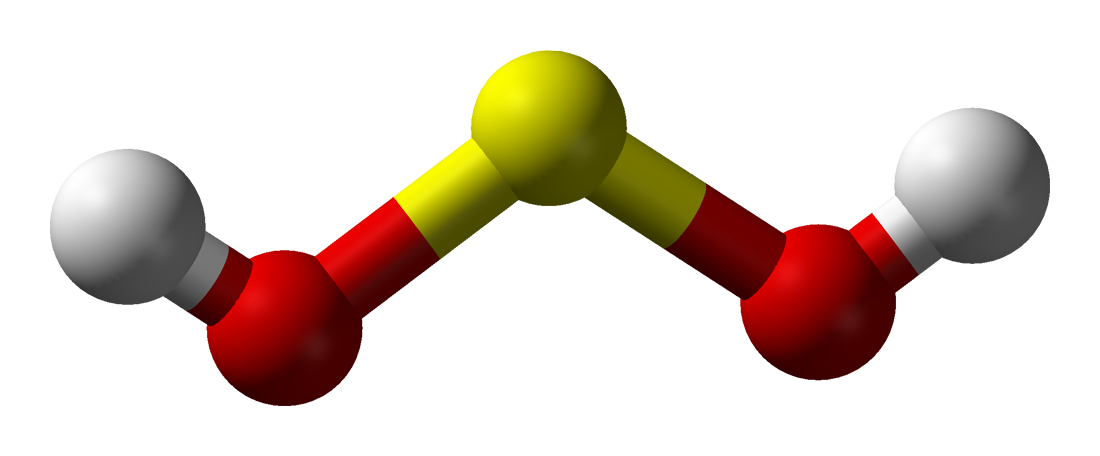
Sulfoxylic acid
Sulfoxylic acid (H2SO2) (also known as hyposulfurous acid or sulfur dihydroxide) is an unstable oxoacid of sulfur in an intermediate oxidation state between hydrogen sulfide and dithionous acid. It consists of two hydroxy groups attached to a sulfur atom. Sulfoxylic acid contains sulfur in an oxidation state of +2. Sulfur monoxide (SO) can be considered as a theoretical anhydride for sulfoxylic acid, but it is not actually known to react with water.
The complementary base is the sulfoxylate anion SO2−
2, which is much more stable. Between these states lies the HSO−
2 ion, which is also somewhat stable.
Sulfoxylate ions can be made by decomposing thiourea dioxide in an alkaline solution. To do this, thiourea dioxide first forms an amidine-sulfinic acid tautomer, H2NC(=NH)SO2H, which then breaks apart. Sulfoxylate reacts with formaldehyde to yield a hydroxymethanesulfinate called rongalite:
which is an important chemical for dyeing.
Sulfoxylic acid has been detected in the gas phase. It is likely to be formed as an intermediate when hydrogen sulfide is oxidised by living organisms, or in the atmosphere, or anywhere else in the natural environment. It may also exist in circumstellar disks. When H2S is oxidised it starts from oxidation state −2, and should then pass through intermediate values of 0 and +2 before getting to well known sulfite at +4 and sulfate at +6. When sulfide in alkaline conditions is oxidised by air in the presence of nickel ions, sulfoxylate concentration first increases to around 5% and then decreases over several days. Polysulfide concentration also grows and then shrinks on a slower timescale, reaching about 25% of the sulfide. The sulfur ends up forming thiosulfate.
Sulfoxylic acid has been made by ultraviolet irradiation of a mixture of solid H2S and H2O, followed by warming. This is a possible natural process in comets or circumstellar disks.
Fender et al. claimed to make "sulfinic acid" (an isomer of sulfoxylic acid) by ultraviolet irradiation on solid sulfur dioxide and hydrogen sulfide in a solid argon matrix, measuring the infrared vibrational spectrum. However, the assignment of the lines in the spectrum is doubtful, so this may not be the substance produced.
Sulfoxylic acid can be made in the gas phase in an electric discharge through a neon, H2, SO2 mixture. This also yields some sulfhydryl hydroperoxide.
Hub AI
Sulfoxylic acid AI simulator
(@Sulfoxylic acid_simulator)
Sulfoxylic acid
Sulfoxylic acid (H2SO2) (also known as hyposulfurous acid or sulfur dihydroxide) is an unstable oxoacid of sulfur in an intermediate oxidation state between hydrogen sulfide and dithionous acid. It consists of two hydroxy groups attached to a sulfur atom. Sulfoxylic acid contains sulfur in an oxidation state of +2. Sulfur monoxide (SO) can be considered as a theoretical anhydride for sulfoxylic acid, but it is not actually known to react with water.
The complementary base is the sulfoxylate anion SO2−
2, which is much more stable. Between these states lies the HSO−
2 ion, which is also somewhat stable.
Sulfoxylate ions can be made by decomposing thiourea dioxide in an alkaline solution. To do this, thiourea dioxide first forms an amidine-sulfinic acid tautomer, H2NC(=NH)SO2H, which then breaks apart. Sulfoxylate reacts with formaldehyde to yield a hydroxymethanesulfinate called rongalite:
which is an important chemical for dyeing.
Sulfoxylic acid has been detected in the gas phase. It is likely to be formed as an intermediate when hydrogen sulfide is oxidised by living organisms, or in the atmosphere, or anywhere else in the natural environment. It may also exist in circumstellar disks. When H2S is oxidised it starts from oxidation state −2, and should then pass through intermediate values of 0 and +2 before getting to well known sulfite at +4 and sulfate at +6. When sulfide in alkaline conditions is oxidised by air in the presence of nickel ions, sulfoxylate concentration first increases to around 5% and then decreases over several days. Polysulfide concentration also grows and then shrinks on a slower timescale, reaching about 25% of the sulfide. The sulfur ends up forming thiosulfate.
Sulfoxylic acid has been made by ultraviolet irradiation of a mixture of solid H2S and H2O, followed by warming. This is a possible natural process in comets or circumstellar disks.
Fender et al. claimed to make "sulfinic acid" (an isomer of sulfoxylic acid) by ultraviolet irradiation on solid sulfur dioxide and hydrogen sulfide in a solid argon matrix, measuring the infrared vibrational spectrum. However, the assignment of the lines in the spectrum is doubtful, so this may not be the substance produced.
Sulfoxylic acid can be made in the gas phase in an electric discharge through a neon, H2, SO2 mixture. This also yields some sulfhydryl hydroperoxide.